- Methylenetetrahydrofolate reductase
-
methylene tetrahydrofolate reductase [NAD(P)H] 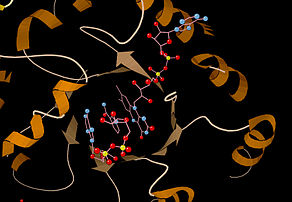
Ribbon diagram of the active site of E. coli MTHFR. The flavin cofactor (top) is shown interacting with the bound substrate NADH.[1] Identifiers EC number 1.5.1.20 CAS number 9028-69-7 Databases IntEnz IntEnz view BRENDA BRENDA entry ExPASy NiceZyme view KEGG KEGG entry MetaCyc metabolic pathway PRIAM profile PDB structures RCSB PDB PDBe PDBsum Gene Ontology AmiGO / EGO Search PMC articles PubMed articles Methylenetetrahydrofolate reductase (NAD(P)H) Identifiers Symbols MTHFR; External IDs OMIM: 607093 MGI: 106639 HomoloGene: 4349 GeneCards: MTHFR Gene EC number 1.5.1.20 Gene Ontology Molecular function • methylenetetrahydrofolate reductase (NADPH) activity
• protein binding
• oxidoreductase activityCellular component • cytosol Biological process • cellular amino acid metabolic process
• vitamin metabolic process
• water-soluble vitamin metabolic process
• blood circulation
• methionine biosynthetic process
• folic acid metabolic processSources: Amigo / QuickGO RNA expression pattern 
More reference expression data Orthologs Species Human Mouse Entrez 4524 17769 Ensembl ENSG00000177000 ENSMUSG00000029009 UniProt P42898 Q3V399 RefSeq (mRNA) NM_005957.4 NM_010840 RefSeq (protein) NP_005948.3 NP_034970 Location (UCSC) Chr 1:
11.85 – 11.87 MbChr 4:
147.41 – 147.43 MbPubMed search [1] [2] Methylenetetrahydrofolate reductase (MTHFR) is an enzyme that in humans is encoded by the MTHFR gene.[2] Methylenetetrahydrofolate reductase catalyzes the conversion of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, a cosubstrate for homocysteine remethylation to methionine. Genetic variation in this gene influences susceptibility to occlusive vascular disease, neural tube defects, colon cancer and acute leukemia, and mutations in this gene are associated with methylenetetrahydrofolate reductase deficiency.[3][4]
Contents
Biochemistry
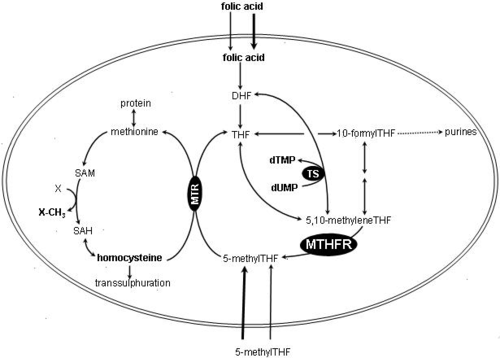
MTHFR irreversibly reduces 5,10-methylenetetrahydrofolate (substrate) to 5-methyltetrahydrofolate (product).
- 5,10-methylenetetrahydrofolate is used to convert dUMP to dTMP for de novo thymidine synthesis.
- 5-Methyltetrahydrofolate is used to convert homocysteine (a potentially toxic amino acid) to methionine by the enzyme methionine synthase. (Note that homocysteine can also be converted to methionine by the folate-independent enzyme betaine-homocysteine methyltransferase (BHMT))
MTHFR contains a bound flavin cofactor and uses NAD(P)H as the reducing agent.
Structure
Mammalian MTHFR is composed of an N-terminal catalytic domain and a C-terminal regulatory domain. MTHFR has at least two promoters and two isoforms (70 kDa and 77 kDa).[5]
Regulation
MTHFR activity may be inhibited by binding of dihydrofolate (DHF)[6] and S-adenosylmethionine (SAM, or AdoMet).[7] MTHFR can also be phosphorylated - this decreases its activity by ~20% and allows it to be more easily inhibited by SAM.[8]
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective Wikipedia articles. [9]
Genetics
The enzyme is coded by the gene with the symbol MTHFR on chromosome 1 location p36.3 in humans.[10] There are DNA sequence variants (genetic polymorphisms) associated with this gene. In 2000 a report brought the number of polymorphisms up to 24.[11] Two of the most investigated are C677T (rs1801133) and A1298C (rs1801131) single nucleotide polymorphisms (SNP).
C677T SNP (Ala222Val)
The MTHFR nucleotide at position 677 in the gene has two possibilities: C (cytosine) or T (thymine). C at position 677 (leading to an alanine at amino acid 222) is the normal allele. The 677T allele (leading to a valine substitution at amino acid 222) encodes a thermolabile enzyme with reduced activity.
Individual with two copies of 677C (677CC) have the "normal" or "wildtype" genotype. 677TT individuals (homozygous) are said to have mild MTHFR deficiency. 677CT individuals (heterozygotes) are almost the same as normal individuals because the normal MTHFR can make up for the thermolabile MTHFR. About ten percent of the North American population are T-homozygous for this polymorphism. There is ethnic variability in the frequency of the T allele – frequency in Mediterranean/Hispanics is greater than the frequency in Caucasians which, in turn, is greater than in Africans/African-Americans.[12]
The degree of enzyme thermolability (assessed as residual activity after heat inactivation) is much greater in 677TT individuals (18-22%) compared with 677CT (56%) and 677CC (66-67%).[13] Individuals of 677TT are predisposed to mild hyperhomocysteinemia (high blood homocysteine levels), because they have less active MTHFR available to produce 5-methyltetrahydrofolate (which is used to decrease homocysteine). Low dietary intake of the vitamin folic acid can also cause mild hyperhomocysteinemia.
Low folate intake affects individuals with the 677TT genotype to a greater extent than those with the 677CC/CT genotypes. 677TT (but not 677CC/CT) individuals with lower plasma folate levels are at risk for elevated plasma homocysteine levels.[14] In studies of human recombinant MTHFR, the protein encoded by 677T loses its FAD cofactor three times faster than the wild-type protein.[15] 5-Methyl-THF slows the rate of FAD release in both the wild-type and mutant enzymes, although it is to a much greater extent in the mutant enzyme.[16] 677TT individuals are at a decreased risk for certain leukemias[17] and colon cancer.[18]
Mutations in the MTHFR gene could be one of the factors leading to increased risk of developing schizophrenia.[19] Schizophrenic patients having the risk allele (T\T) show more deficiencies in executive function tasks.[20]
A1298C SNP (Glu429Ala)
At nucleotide 1298 of the MTHFR, there are two possibilities: A or C. 1298A (leading to a Glu at amino acid 429) is the most common while 1298C (leading to an Ala substitution at amino acid 429) is less common. 1298AA is the "normal" homozygous, 1298AC the heterozygous, and 1298CC the homozygous for the "variant". In studies of human recombinant MTHFR, the protein encoded by 1298C cannot be distinguished from 1298A in terms of activity, thermolability, FAD release, or the protective effect of 5-methyl-THF.[15] The C mutation does not appear to affect the MTHFR protein. It does not result in thermolabile MTHFR and does not appear to affect homocysteine levels.
Compound Heterozygotes
Mutations at 677 and 1298 are different locations; however, they are both in the 'same' gene: MTHFR. Some studies have shown that the MTHFR protein in people with the genotype 677CT 1298AC does its job a bit less well than the normal MTHFR.
Severe MTHFR deficiency
Severe MTHFR deficiency is rare (about 50 cases worldwide) and caused by mutations resulting in 0-20% residual enzyme activity.[11] Patients exhibit developmental delay, motor and gait dysfunction, seizures, and neurological impairment and have extremely high levels of homocysteine in their plasma and urine as well as low to normal plasma methionine levels.
Reaction schematic and folate pathway
MTHFR = methylenetetrahydrofolate reductase DHF = dihydrofolate THF = tetrahydrofolate 5,10-methylene-THF =
5,10-methylenetetrahydrofolate5-methyl-THF =
5-methyltetrahydrofolateMTR = methionine synthase SAH = S-adenosylhomocysteine NADPH = reduced form of Nicotinamide adenine dinucleotide phosphate NADP+ = oxidized form of Nicotinamide adenine dinucleotide phosphate SAM = S-Adenosyl methionine TS = thymidylate synthase As a drug target
Inhibitors of MTHFR or antisense knockdown of the expression of the enzyme has been proposed as a treatment for cancer.[21]
References
- ^ PDB 1ZPT; Pejchal R, Sargeant R, Ludwig ML (2005). "Structures of NADH and CH3-H4folate complexes of Escherichia coli methylenetetrahydrofolate reductase reveal a spartan strategy for a ping-pong reaction". Biochemistry 44 (34): 11447–57. doi:10.1021/bi050533q. PMID 16114881.
- ^ Goyette P, Sumner JS, Milos R, Duncan AM, Rosenblatt DS, Matthews RG, Rozen R (June 1994). "Human methylenetetrahydrofolate reductase: isolation of cDNA, mapping and mutation identification". Nat. Genet. 7 (2): 195–200. doi:10.1038/ng0694-195. PMID 7920641.
- ^ "Entrez Gene: MTHFR methylenetetrahydrofolate reductase (NAD(P)H)". http://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=4524.
- ^ Födinger M, Hörl WH, Sunder-Plassmann G (2000). "Molecular biology of 5,10-methylenetetrahydrofolate reductase.". J Nephrol 13 (1): 20–33. PMID 10720211.
- ^ Tran P, Leclerc D, Chan M, et al. (September 2002). "Multiple transcription start sites and alternative splicing in the methylenetetrahydrofolate reductase gene result in two enzyme isoforms". Mamm. Genome 13 (9): 483–92. doi:10.1007/s00335-002-2167-6. PMID 12370778.
- ^ Matthews RG, Daubner SC (1982). "Modulation of methylenetetrahydrofolate reductase activity by S-adenosylmethionine and by dihydrofolate and its polyglutamate analogues". Adv. Enzyme Regul. 20: 123–31. doi:10.1016/0065-2571(82)90012-7. PMID 7051769.
- ^ Jencks DA, Mathews RG (February 1987). "Allosteric inhibition of methylenetetrahydrofolate reductase by adenosylmethionine. Effects of adenosylmethionine and NADPH on the equilibrium between active and inactive forms of the enzyme and on the kinetics of approach to equilibrium". J. Biol. Chem. 262 (6): 2485–93. PMID 3818603. http://www.jbc.org/cgi/pmidlookup?view=long&pmid=3818603.
- ^ Yamada K, Strahler JR, Andrews PC, Matthews RG (July 2005). "Regulation of human methylenetetrahydrofolate reductase by phosphorylation". Proc. Natl. Acad. Sci. U.S.A. 102 (30): 10454–9. doi:10.1073/pnas.0504786102. PMC 1180802. PMID 16024724. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1180802.
- ^ The interactive pathway map can be edited at WikiPathways: "FluoropyrimidineActivity_WP1601". http://www.wikipathways.org/index.php/Pathway:WP1601.
- ^ Goyette P, Sumner JS, Milos R, et al. (August 1994). "Human methylenetetrahydrofolate reductase: isolation of cDNA mapping and mutation identification". Nat. Genet. 7 (4): 551. doi:10.1038/ng0894-551a. PMID 7951330.
- ^ a b Sibani S, Christensen B, O'Ferrall E, Saadi I, Hiou-Tim F, Rosenblatt DS, Rozen R (2000). "Characterization of six novel mutations in the methylenetetrahydrofolate reductase (MTHFR) gene in patients with homocystinuria". Hum. Mutat. 15 (3): 280–7. doi:10.1002/(SICI)1098-1004(200003)15:3<280::AID-HUMU9>3.0.CO;2-I. PMID 10679944.
- ^ Schneider JA, Rees DC, Liu YT, Clegg JB (May 1998). "Worldwide distribution of a common methylenetetrahydrofolate reductase mutation". Am. J. Hum. Genet. 62 (5): 1258–60. doi:10.1086/301836. PMC 1377093. PMID 9545406. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1377093.
- ^ Frosst P, Blom HJ, Milos R, Goyette P, Sheppard CA, Matthews RG, Boers GJ, den Heijer M, Kluijtmans LA, van den Heuvel LP et al. (May 1995). "A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase". Nat. Genet. 10 (1): 111–3. doi:10.1038/ng0595-111. PMID 7647779.
- ^ Jacques PF, Bostom AG, Williams RR, Ellison RC, Eckfeldt JH, Rosenberg IH, Selhub J, Rozen R (January 1996). "Relation between folate status, a common mutation in methylenetetrahydrofolate reductase, and plasma homocysteine concentrations". Circulation 93 (1): 7–9. PMID 8616944. http://circ.ahajournals.org/cgi/content/abstract/93/1/7.
- ^ a b Yamada K, Chen Z, Rozen R, Matthews RG (December 2001). "Effects of common polymorphisms on the properties of recombinant human methylenetetrahydrofolate reductase". Proc. Natl. Acad. Sci. U.S.A. 98 (26): 14853–8. doi:10.1073/pnas.261469998. PMC 64948. PMID 11742092. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=64948.
- ^ Schwahn B, Rozen R (2001). "Polymorphisms in the methylenetetrahydrofolate reductase gene: clinical consequences". Am J Pharmacogenomics 1 (3): 189–201. doi:10.2165/00129785-200101030-00004. PMID 12083967.
- ^ Skibola CF, Smith MT, Kane E, Roman E, Rollinson S, Cartwright RA, Morgan G' (October 1999). "Polymorphisms in the methylenetetrahydrofolate reductase gene are associated with susceptibility to acute leukemia in adults". Proc. Natl. Acad. Sci. U.S.A. 96 (22): 12810–5. doi:10.1073/pnas.96.22.12810. PMC 23109. PMID 10536004. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=23109.
- ^ Ma J, Stampfer MJ, Giovannucci E, Artigas C, Hunter DJ, Fuchs C, Willett WC, Selhub J, Hennekens CH, Rozen R (15 March 1997). "Methylenetetrahydrofolate reductase polymorphism, dietary interactions, and risk of colorectal cancer". Cancer Res. 57 (6): 1098–102. PMID 9067278. http://cancerres.aacrjournals.org/cgi/content/abstract/57/6/1098.
- ^ "Meta-Analysis of All Published Schizophrenia-Association Studies (Case-Control Only) for rs1801133 (C677T) polymorphism, MTHFR gene". Schizophrenia Research Forum. http://www.schizophreniaforum.org/res/sczgene/meta.asp?geneID=4. Retrieved 2007-03-11.
- ^ Roffman JL, Weiss AP, Deckersbach T, Freudenreich O, Henderson DC, Purcell S, Wong DH, Halsted CH, Goff DC (May 2007). "Effects of the methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism on executive function in schizophrenia". Schizophr. Res. 92 (1–3): 181–8. doi:10.1016/j.schres.2007.01.003. PMID 17344026.
- ^ Stankova J, Lawrance AK, Rozen R (2008). "Methylenetetrahydrofolate reductase (MTHFR): a novel target for cancer therapy". Curr Pharm Des 14 (11): 1143–50. doi:10.2174/138161208784246171. PMID 18473861.
Further reading
- Matthews RG (2003). "Methylenetetrahydrofolate reductase: a common human polymorphism and its biochemical implications". Chemical record 2 (1): 4–12. doi:10.1002/tcr.10006. PMID 11933257.
- Schwahn B, Rozen R (2002). "Polymorphisms in the methylenetetrahydrofolate reductase gene: clinical consequences". American journal of pharmacogenomics: genomics-related research in drug development and clinical practice 1 (3): 189–201. doi:10.2165/00129785-200101030-00004. PMID 12083967.
- Iqbal MP, Frossard PM (2003). "Methylene tetrahydrofolate reductase gene and coronary artery disease". JPMA. the Journal of the Pakistan Medical Association 53 (1): 33–6. PMID 12666851.
- Bailey LB (2003). "Folate, methyl-related nutrients, alcohol, and the MTHFR 677C-->T polymorphism affect cancer risk: intake recommendations". J. Nutr. 133 (11 Suppl 1): 3748S–3753S. PMID 14608109.
- Wiwanitkit V (2005). "Roles of methylenetetrahydrofolate reductase C677T polymorphism in repeated pregnancy loss". Clin. Appl. Thromb. Hemost. 11 (3): 343–5. doi:10.1177/107602960501100315. PMID 16015422.
- Muntjewerff JW, Kahn RS, Blom HJ, den Heijer M (2006). "Homocysteine, methylenetetrahydrofolate reductase and risk of schizophrenia: a meta-analysis". Mol. Psychiatry 11 (2): 143–9. doi:10.1038/sj.mp.4001746. PMID 16172608.
- Lewis SJ, Lawlor DA, Davey Smith G, et al. (2006). "The thermolabile variant of MTHFR is associated with depression in the British Women's Heart and Health Study and a meta-analysis". Mol. Psychiatry 11 (4): 352–60. doi:10.1038/sj.mp.4001790. PMID 16402130.
- Pereira TV, Rudnicki M, Pereira AC, et al. (2007). "5,10-Methylenetetrahydrofolate reductase polymorphisms and acute lymphoblastic leukemia risk: a meta-analysis". Cancer Epidemiol. Biomarkers Prev. 15 (10): 1956–63. doi:10.1158/1055-9965.EPI-06-0334. PMID 17035405.
- Leclerc D, Rozen R (2007). "[Molecular genetics of MTHFR: polymorphisms are not all benign]". Med Sci (Paris) 23 (3): 297–302. doi:10.1051/medsci/2007233297. PMID 17349292.
External links
- Smith DO (2007-02-08). "MTHFR and homocysteine". Ask Dr. Stephan Moll / Factor V Leiden / Thrombophilia Support Page. Thrombophilia Awareness Project. http://www.fvleiden.org/ask/51.html. Retrieved 2008-08-02.
Acetolactate synthase - Acyl CoA dehydrogenase - Apoptosis-inducing factor - Butyryl CoA dehydrogenase - Cryptochrome - Cytochrome b5 reductase - Dihydrolipoamide dehydrogenase - Flavodoxin - Methemoglobin reductase - Methylenetetrahydrofolate reductase - NADH dehydrogenase - NADPH oxidase - Nitrate reductase - Sarcosine oxidase - Thioredoxin reductaseOxidoreductases: CH-NH (EC 1.5) 1.5.1: NAD or NADP acceptor 1.5.3: oxygen acceptor 1.5.5: quinone acceptor 1.5.99 B enzm: 1.1/2/3/4/5/6/7/8/10/11/13/14/15-18, 2.1/2/3/4/5/6/7/8, 2.7.10, 2.7.11-12, 3.1/2/3/4/5/6/7, 3.1.3.48, 3.4.21/22/23/24, 4.1/2/3/4/5/6, 5.1/2/3/4/99, 6.1-3/4/5-6 Metabolism of vitamins, coenzymes, and cofactors Fat soluble vitamins Alpha-tocopherol transfer proteinliver (Sterol 27-hydroxylase or CYP27A1) · renal (25-Hydroxyvitamin D3 1-alpha-hydroxylase or CYP27B1) · degradation (1,25-Dihydroxyvitamin D3 24-hydroxylase or CYP24A1)Water soluble vitamins Dihydropteroate synthase · Dihydrofolate reductase · Serine hydroxymethyltransferase
Methylenetetrahydrofolate reductaseNonvitamin cofactors M: NUT
cof, enz, met
noco, nuvi, sysi/epon, met
drug(A8/11/12)
Categories:- Human proteins
- EC 1.5.1
Wikimedia Foundation. 2010.