- History of malaria
-
The history of malaria predates humanity, as this ancient disease evolved before humans did. Malaria, a widespread and potentially lethal infectious disease, has afflicted people for much of human history, and has affected settlement patterns. The prevention and treatment of the disease have been investigated in science and medicine for hundreds of years, and, since the discovery of the parasite which causes it, attention has focused on its biology. These studies have continued up to the present day, since no effective Malaria vaccine has yet been developed and many of the older antimalarial drugs are losing effectiveness as the parasite evolves high levels of drug resistance. As malaria remains a major public health problem, causing 250 million cases of fever and approximately one million deaths annually, understanding its history is key.
Contents
Origin and early history
Human malaria likely originated in Africa and has coevolved along with its hosts, mosquitoes and non-human primates. The first evidence of malaria parasites was found in mosquitoes preserved in amber from the Palaeogene period that are approximately 30 million years old.[1] Malaria may have been a human pathogen for the entire history of the species.[2][3] Humans may have originally caught Plasmodium falciparum from gorillas.[4] About 10,000 years ago malaria started having a major impact on human survival which coincides with the start of agriculture (Neolithic revolution);[5] a consequence was natural selection for sickle-cell disease, thalassaemias, glucose-6-phosphate dehydrogenase deficiency, ovalocytosis, elliptocytosis and loss of the Gerbich antigen (glycophorin C) and the Duffy antigen on the erythrocytes because such blood disorders confer a selective advantage against malaria infection (balancing selection).[6] The three major types of inherited genetic resistance (sickle-cell disease, thalassaemias, and glucose-6-phosphate dehydrogenase deficiency) were present in the Mediterranean world by the time of the Roman Empire, about 2000 years ago.[7]
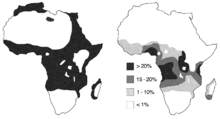 Left: regions in Africa where Plasmodium falciparum malaria was transmitted before control was introduced. Right: frequencies of sickle-cell heterozygotes in the indigenous African population.[8]
Left: regions in Africa where Plasmodium falciparum malaria was transmitted before control was introduced. Right: frequencies of sickle-cell heterozygotes in the indigenous African population.[8]
References to the unique periodic fevers of malaria are found throughout recorded history. According to legend, the Chinese emperor Huang Di (Yellow Emperor, 2697–2590 BCE) ordered the compilation of a canon of internal medicine. The Chinese Huangdi Neijing (The Inner Canon of the Yellow Emperor) apparently refers to repeated paroxysmal fevers associated with enlarged spleens and a tendency to epidemic occurrence – the earliest written report of malaria.[9] In the Sushruta Samhita, a Sanskrit medical treatise (6th century BCE), the symptoms of malarial fever were described and attributed to the bites of certain insects.[10]
The term 'miasma' was coined by Hippocrates of Kos who used it to describe dangerous fumes from the ground that are transported by winds and can cause serious illnesses.[11] The name malaria, derived from ‘mal’aria’ (bad air in Medieval Italian) was probably first used by Leonardo Bruni in a publication of 1476.[12] This idea came from the Ancient Romans who thought that this disease came from the horrible fumes from the swamps. The idea that the disease came from the foul gasses released from soil, water and air persisted throughout the nineteenth century.[13]
Malaria was once common in most of Europe and North America, where it is now for all purposes non-existent. The coastal plains of southern Italy, for example, fell from international prominence (the Crusaders going by sea to the Holy Land took ship at Bari) when malaria expanded its reach in the sixteenth century. At roughly the same time, in the coastal marshes of England, mortality from "marsh fever" or "tertian ague" ("the ague" from Latin "febris acuta") was comparable to that in sub-Saharan Africa today.[14][15] William Shakespeare was born at the start of the especially cold period that climatologists call the "Little Ice Age", yet he was aware enough of the ravages of the disease to mention it in eight of his plays.[16] Throughout history the most critical factors in the spread or eradication of disease have been human behavior (shifting population centers, changing farming methods and the like) and living standards. Precise statistics do not exist because many cases occur in rural areas where people do not have access to hospitals or the means to afford health care. As a consequence, the majority of cases are undocumented.[17] Poverty has been and remains a reason for the disease to remain while it has undergone a decline in other locations.[18] Climate change is likely to affect future trends in malaria transmission, but the severity and geographic distribution of such effects is currently uncertain, though attracting increasing scientific attention.[19]
Early research and treatment
The introduction of molecular methods confirmed the high prevalence of P.falciparum malaria in ancient Egypt.[21][22] The historian Herodotus (484–425 BCE) wrote that the builders of the Egyptian pyramids were given large amount of garlic,[23] likely to protect them against malaria. Sneferu, the founder of the Fourth dynasty of Egypt, who reigned from around 2613 - 2589 BCE, used bed-nets as protection against mosquitoes, Cleopatra VII, the last Pharaoh of Ancient Egypt, also slept under a mosquito net.[24] Malaria became widely recognized in ancient Greece by the 4th century BCE, and is implicated in the decline of many city-state populations. Hippocrates (460–370 BCE), the "father of medicine", related the presence of intermittent fevers with climatic and environmental conditions and classified the fever according to periodicity: tritaios pyretos / febris tertiana, and tetrataios pyretos / febris quartana (every fourth day).[25][26]
For thousands of years, traditional herbal remedies have been used to treat malaria.[27] Around 168 BCE the herbal remedy Qing-hao (Artemisia annua) came into use in China to treat female hemorrhoids (Recipes for 52 kinds of diseases unearthed from the Mawangdui tombs).[20]
 Ge Hong manuscript:„take a bunch of qing hao and two sheng of water for soaking it, wring it out to obtain the juice and ingest it in its entirety.” (N.B. one sheng amounted to 0.2 l at the time).[28]
Ge Hong manuscript:„take a bunch of qing hao and two sheng of water for soaking it, wring it out to obtain the juice and ingest it in its entirety.” (N.B. one sheng amounted to 0.2 l at the time).[28]
Qinghao was first recommended for acute intermittent fever episodes by Ge Hong as an effective medication in the 4th century Chinese manuscript Zhou hou bei ji fang, usually translated as "Emergency Prescriptions kept in one's Sleeve".[29] His recommendation was to soak fresh plants of the artemisia herb in cold water, wring it out and ingest the expressed bitter juice in its raw state.[30][31]
Medical accounts and ancient autopsy reports state that tertian malarial fevers caused the death of four members of the Medici family of Florence: Eleonora of Toledo (1522–1562), Cardinal Giovanni (1543–1562), Don Garzia (1547–1562) and Grand Duke Francesco I (1531–1587). These claims have been reexamined with more modern methologies.[32] These methods have confirmed the presence of P.falciparum in the remains confirming the original diagnosis.
Treatment of malaria was discussed in several European herbal texts during the Renaissance including Otto Brunfels (1532), Leonhart Fuchs (1543), Adam Lonicer (1560), Hieronymus Bock (1577), Pietro Andrea Mattioli (1590), and Theodor Zwinger (1696).[33]
European settlers and their West African slaves likely brought malaria to the Americas in the 16th century.[34] Spanish missionaries found that fever was treated by Amerindians near Loxa (Peru) with powder from Peruvian bark (Cinchona succirubra).[35] There are no references to malaria in the "medical books" of the Mayans or Aztecs. Quinine (Kinine), a toxic plant alkaloid, is an effective muscle relaxant, as the modern use for nocturnal leg cramps suggests,[36] long used by the Quechua Indians of Peru to reduce the shaking effects caused by severe chills in the Andes.[37] The Jesuit Brother Agostino Salumbrino (1561–1642), an apothecary by training and who lived in Lima, observed the Quechua using the quinine-containing bark of the cinchona tree for that purpose. While its effect in treating malaria (and hence malaria-induced shivering) was entirely unrelated to its effect in controlling shivering from cold, it was nevertheless the correct medicine for malaria. The use of the “fever tree” bark was introduced into European medicine by Jesuit missionaries (Jesuit's bark).[38][39] Jesuit Barnabé de Cobo (1582–1657), who explored Mexico and Peru, is credited with taking cinchona bark to Europe. He brought the bark from Lima to Spain, and afterwards to Rome and other parts of Italy, in 1632. Francesco Torti published in 1712 that only “intermittent fever” was amenable to the fever tree bark (“Therapeutice Specialis ad Febres Periodicas Perniciosas”, 1712 Modena). This work finally established the specific nature of cinchona bark and brought about its general use in medicine.[40] In 1717, the graphite pigmentation of a postmortem spleen and brain was published by Giovanni Maria Lancisi in his malaria text book “De noxiis paludum effluviis eorumque remediis”. He related the prevalence of malaria in swampy areas to the presence of flies and recommended swamp drainage to prevent it.[41]
19th century
Antimalarial drugs
Quinine
Pierre Joseph Pelletier and Joseph Bienaimé Caventou separated in 1820 the alkaloids Cinchonine and Quinine from powdered fever tree bark, allowing for the creation of standardized doses of the active ingredients.[42][43] Prior to 1820, the bark was first dried, ground to a fine powder and then mixed into a liquid (commonly wine) which was then drunk.[44]
An English trader, Charles Ledger, and his Amerindian servant, Manuel Incra Mamani, had spent four years collecting cinchona seeds in the Andes in Bolivia, highly prized for their quinine but a prohibited export. Ledger managed to get some seeds out; in 1865 the Dutch government bought a small parcel, and 20,000 trees of the famous Cinchona ledgeriana were successfully cultivated in Java (Indonesia). By the end of the nineteenth century the Dutch had established a world monopoly in the supply of quinine.[45]
'Warburg's Tincture'
In 1834, in British Guiana (now Guyana), a German physician, Carl Warburg, invented an antipyretic medicine: 'Warburg's Tincture'. This secret, proprietary remedy contained quinine and various different other herbs. Trials were made in Europe in the 1840s and 1850s, and it was officially adopted by the Austrian Empire in 1847. Warburg's Tincture gained a high, international reputation. It was considered by many eminent medical professionals to be a more efficacious antimalarial drug than quinine. It was also more economical. The British Government supplied Warburg's Tincture to troops in India and other colonies.[46][47]
Synthetic drugs
William Henry Perkin, a student of August Wilhelm von Hofmann at the Royal College of Chemistry in London, tried in the 1850s to synthesize quinine in a commercially practicable process. The idea was to take two equivalents of N-allyltoluidine (C10H13N) and three atoms of oxygen to produce quinine (C20H24N2O2) and water. The experiments were unsuccessful. However, Perkin's Mauve was produced when attempting quinine total synthesis via the oxidation of N-allyltoluidine.[48] Before Perkin's discovery all dyes and pigments were derived from roots, leaves, insects, or, in the case of Tyrian purple, molluscs. Perkin's discovery of artificially synthesized dyes led to important advances in medicine, photography, and many other fields.
In 1891 Paul Guttmann and Paul Ehrlich noted that methylene blue had a high affinity for some tissues and that this dye had a slight antimalarial property.[49] MB and its congeners may act by preventing the biocrystallization of heme.[50] A mixture of eosin Y, methylene blue and demethylated methylene blue (azure B) was later used for a number of different blood film staining procedures (Malachowski stain, Romanowsky stain, Giemsa stain).[51][52] Ehrlich, the founder of chemotherapy, advocated a rational development of drugs by exploiting biochemical differences (“magic bullets”).[53]
 Charles Louis Alphonse Laveran observed pigmented parasites and the exflagellation of male gametocytes.[54]
Charles Louis Alphonse Laveran observed pigmented parasites and the exflagellation of male gametocytes.[54]
Life cycle
Johann Heinrich Meckel[55] recorded in 1848 innumerable black-brown pigment granules in the blood and spleen of a patient who had died in a hospital for insane people. Meckel was probably looking at the parasites of malaria without realizing it; malaria was not mentioned in his report. He thought the pigment was melanin.[56] The causal relationship of pigment to the parasite was established in 1880, when the French physician Charles Louis Alphonse Laveran, working in the military hospital of Constantine Algeria, observed pigmented parasites inside the red blood cells of people suffering from malaria. He also witnessed the events of exflagellation and became convinced that the moving flagella were parasitic microorganisms. He noted that quinine removed the parasites from the blood. Laveran called this microscopic organism Oscillaria malariae and proposed that malaria was caused by this protozoan.[57] This discovery was not initially well received and remained controversial until the development of the oil immersion lens in 1884 and of superior staining methods in 1890-1891.
In 1885 Ettore Marchiafava, Angelo Celli and Camillo Golgi studied the reproduction cycles in human blood (Golgi cycles). Golgi observed that all parasites present in the blood divided almost simultaneously at regular intervals and that division coincided with attacks of fever. Golgi also recognized that the three types of malaria are caused by different protozoan organisms. By 1890 Laveran's germ was generally accepted but most of Laveran's initial ideas had been discarded in favor of the taxonomic work and clinical pathology of the Italian school. Marchiafava and Celli called the new microorganism Plasmodium.[58] Pel, presaging its discovery by over 50 years, proposed the first theory of the existence of a tissue stage of the malaria parasite in 1886. This suggestion was reiterated in 1893 when Golgi also suggested that the parasites might have an undiscovered tissue phase this time in endothelial cells.[59] Pel in 1896 supported Gogli's latent phase theory.[60] Also in 1886 Golgi described the morphological differences that are still used to distinguish two malaria parasite species Plasmodium vivax and Plasmodium malariae. Shortly after this Sakharov in 1889 and Marchiafava & Celli in 1890 independently identified Plasmodium falciparum as a species distinct from P. vivax and P. malariae. In 1890, Grassi and Feletti reviewed the available information and named both P. malariae and P. vivax with the following statement: "C'est pour cela que nous distinguons, dans le genre Haemamoeba, trois espèces (H. malariae de la fièvre quarte, H. vivax de la fièvre tierce et H. praecox de la fièvre quotidienne avec coutres intermittences etc.)."[61] H. vivax was soon renamed Plasmodium vivax. In 1892 Marchiafava and Bignami proved that the multiple forms seen by Laveran are a single species. This species was eventually named P. falciparum.
Laveran was awarded the 1907 Nobel Prize for Physiology or Medicine "in recognition of his work on the role played by protozoa in causing diseases".[62] In 1897 the sexual stages of a related haematozoan, Haemoproteus columbae, in the blood were discovered by William MacCallum in infected birds.[57]
Giovanni Maria Lancisi, John Crawford,[63] Patrick Manson,[64] Josiah C. Nott, Albert Freeman Africanus King,[65] and Charles Louis Alphonse Laveran developed theories that malaria may be caused by mosquito bites, but there was little evidence to support this idea. An early effort at malaria prevention occurred in 1896. An Uxbridge malaria outbreak prompted health officer, Dr. Leonard White, to write a report to the State Board of Health, which led to a study of mosquito-malaria links, and the first efforts for malaria prevention.[66] Massachusetts State pathologist, Theobald Smith, asked that White's son collect mosquito specimens for further analysis, and that citizens 1) add screens to windows, and 2) drain collections of water.[66]
It was Britain's Sir Ronald Ross, an army surgeon working in Secunderabad India, who proved in 1897 that malaria is transmitted by mosquitoes. He was able to find pigmented malaria parasites in a mosquito that he artificially fed on a malaria patient named Hussain Khan, who had crescents in his blood. He continued his research into malaria by showing that certain mosquito species (Culex fatigans) transmit malaria to sparrows and isolated malaria parasites from the salivary glands of mosquitoes that had fed on infected birds.[67] He reported this to the British Medical Association in Edinburgh, Scotland in 1898 and was greeted with a standing ovation.
Giovanni Battista Grassi, professor of Comparative Anatomy at Rome University, showed that human malaria could only be transmitted by Anopheles (Greek "anofelís": good-for-nothing) mosquitoes.[68] Grassi along with his coworkers Amico Bignami, Giuseppe Bastianelli and Ettore Marchiafava announced at the session of the Accademia dei Lincei on December 4, 1898 that a healthy man in a non-malarial zone had contracted tertian malaria after being bitten by an experimentally infected Anopheles claviger.
In 1899 Bastianelli and Bignami were the first to observe the complete P. vivax transmission cycle from mosquito to human and back.
A bitter dispute broke out between the British and Italian schools of malariology over priority but Ross received the 1902 Nobel Prize for Physiology or Medicine for "his work on malaria, by which he has shown how it enters the organism and thereby has laid the foundation for successful research on this disease and methods of combating it".[69] In Cuba Carlos Finlay found that yellow fever is transmitted by another type of mosquito, now Aedes aegypti. His observation was confirmed by a medical board headed by Walter Reed. Yellow fever and malaria among workers had seriously delayed construction of the Panama Canal. Mosquito control instituted by William C. Gorgas dramatically reduced this problem.[70] Relapses were first noted in 1897 by Thayer who recounted the experiences of a physician who suffered a relapse of malaria twenty one months after leaving an endemic area.[71] He proposed the existence of a tissue stage.
Microscopic examination of stained blood films
In 1876 methylene blue was synthesized by Heinrich Caro at BASF, a German chemical company.[72] Robert Koch in 1882 used this dye to discover the cause of tuberculosis - Mycobacterium tuberculosis. Paul Ehrlich in 1880 described the use of "neutral" dyes - mixtures of acidic and basic dyes for the differentiation of cells in peripheral blood smears. In 1886 Bernthsen prepared a relatively pure dye, obtained by decomposition of methylene blue which he termed methylene azure. In 1891 Ernst Malachowski[73] and Dmitri Leonidovich Romanowsky [74] independently developed techniques using a mixture of Eosin Y and modified methylene blue (methylene azure) that produced a surprising hue unattributable to either staining component: a beautiful, distinctive shade of purple.[75][76] Malachowski used alkali-treated methylene blue solutions and Romanowsky used methylene blue solution which were moulded or aged. This new method differentiated blood cells and demonstrated the nuclei of malarial parasites. Jenner in 1899 introduced methanol as a solvent for the dye precipitate.
Malachowski's staining technique is one of the most significant technical advances in the history of malaria.[77]
20th century
Life cycle
The existence of relapses was confirmed by Patrick Manson who allowed infected Anopheles mosquitoes to feed on his eldest son - Patrick Thurburn Manson.[78] The younger Manson then described a relapse nine months later after his apparent cure with quinine.[79]
Also in 1900 Amico Bignami and Giuseppe Bastianelli found that they could not infect an individual with blood containing only gametocytes.[80] The possibility of the existence of a chronic blood stage infection was proposed by Ross and Thompson in 1910.[81]
In 1903 Fritz Schaudinn erroneously reported direct infection of erythrocytes by infective sporozoites of P. vivax.[82] Schaudinn's error dominated scientific opinion for over forty years.
The existence of exoerythrocytic merogony of non-human malaria parasites in the internal organs was first demonstrated by Aragão in 1908.[83]
In 1920 Félix Mesnil and Émile Roubaud achieve the first experimental infection of chimpanzees with P. vivax.[84]
Three possible mechanisms of relapse were proposed by Marchoux in 1926 (i) parthenogenesis of macrogametocytes (ii) persistence of schizonts in small numbers in the blood where their multiplication is inhibited by immunity and this immunity disappears and/or (iii) reactivation of an encysted body in the blood.[85] James in 1931 based on the lack of activity of quinine on the sporozoites proposed that after being injected by the mosquito, the sporozoites are carried to internal organs, where they enter the reticuloendothelial cells and undergo a cycle of development.[86] Huff and Bloom in 1935 demonstrated the exoerythrocytic stages of avian malaria.[87] In 1945 Fairley et al. reported that inoculation of blood from a patient with P. vivax may fail to induce malaria in a susceptible recipient although the donor may subsequently develop overt malaria. The sporozoites disappeared from the blood stream within one hour and reappeared eight days later. This suggested that persistent tissue forms existed.[88] Using mosquitoes rather than blood Shute in 1946 described a similar phenomenon and proposes the existence of an 'x-body' or resting form.[89] The following year Sapero proposed that a link existed between a tissue stage not yet discovered in patients with malaria and the phenomenon of relapse.[90] Garnham in 1947 described exoerythrocytic schizogony in Hepatocystis (Plasmodium) kochi.[91] In the following year Shortt and Garnham described the liver stages of P. cynomolgi in the monkey.[92] In the same year a human volunteer consented to receive a massive dose of infected sporozoites of P. vivax and undergo a liver biopsy three months later thus allowing Shortt et al. to demonstrate the tissue stage of a human malarial parasite.[93] The tissue form of Plasmodium ovale was described in 1954 and that of P. malariae in 1960 in experimentally infected chimpanzees.
The latent or dormant liver form of the parasite (hypnozoite), responsible for the late relapses characteristic of P. vivax and P. ovale infections,[94] was observed in the 1980s.[57][95] The term hypnozoite was coined by Miles B. Markus, a PhD student in Imperial College, London. In 1976, he speculated: "If sporozoites of Isospora can behave in this fashion, then those of related Sporozoa, like malaria parasites, may have the ability to survive in the tissues in a similar way." He adopted the term "hypnozoite" for malaria in 1978 when he wrote in a little-known journal that this name would "... describe any dormant sporozoites or dormant, sporozoite-like stages in the life cycles of Plasmodium or other Haemosporina".[96][97]
In 1982 Krotoski et al report identification of P. vivax hypnozoites in liver cells of infected chimpanzees and in 1984 Mazier et al report in vitro cultivation of P. vivax liver stages in human hepatocytes. In 1989 chloroquine resistance in P. vivax is reported in Papua New Guinea.
Therapeutic uses
In the early twentieth century, before antibiotics, patients with syphilis were intentionally infected with malaria to create a fever. In the 1920s Julius Wagner-Jauregg, a Viennese psychiatrist, began to treat neurosyphilitics with induced P. vivax malaria. Three or four bouts of fever were enough to kill the temperature-sensitive syphilis bacteria (Spirochaeta pallida also known as Treponema pallidum). P. vivax infections were terminated by quinine. By accurately controlling the fever with quinine, the effects of both syphilis and malaria could be minimized. Although some patients died from malaria, this was preferable to the almost-certain death from syphilis.[98] Therapeutic malaria opened up a wide field of chemotherapeutic research and was practised until 1950.[99] Wagner-Jauregg was awarded the 1927 Nobel Prize in Physiology or Medicine for his discovery of the therapeutic value of malaria inoculation in the treatment of dementia paralytica.[100]
Control efforts
Efforts to control the spread of malaria suffered a major setback in 1930. Entomologist Raymond Corbett Shannon discovered disease-bearing Anopheles gambiae mosquitoes living in Brazil, likely brought there by plane or fast mail steamer.[101] This species of mosquito is a particularly efficient vector for malaria and is native to Africa.[102] In 1938, the introduction of this new mosquito vector caused the greatest epidemic of malaria ever seen in the New World. However, complete eradication of A.gambiae from north-east Brazil and thus from the New World was achieved in 1940 by meticulous application of Paris Green to breeding places and of Pyrethrum spray-killing to adult resting places.[103]
Antimalarial drugs
Hans Andersag and colleagues synthesized and tested at the Elberfeld laboratories of the IG Farben (Germany) about 12000 different compounds and succeeded in producing Resochin® as substitutes for quinine in the 1930s (Dtsch.-Reichs-Pat. 683692);[104][105] it is chemically related to quinine through the possession of a quinoline nucleus and the dialkylaminoalkylamino side chain. Resochin (a RESOrcinate of a 4-aminoCHINoline) (7-chloro-4- 4- (diethylamino) - 1 - methylbutyl amino quinoline) and a similar compound Sontochin (3-methyl Resochin) were synthesized in 1934 in close cooperation with American companies.[106] There were over 2,000 cartel agreements between IG Farben and foreign firms — including Standard Oil of New Jersey, DuPont, Dow Chemical Company, and others in the United States.[107] In March 1946 the drug was officially named Chloroquine.[108] Chloroquine is an inhibitor of hemozoin production through biocrystallization and is one of the best antimicrobials ever developed. Quinine and chloroquine affect malarial parasites only at stages in their life cycle when the parasites are forming hematin-pigment (hemozoin) as a byproduct of hemoglobin degradation. The drug target of chloroquine is host-derived, which markedly delayed the emergence of resistance and it took P. falciparum 19 years to build resistance to chloroquine.[109] The first chloroquine-resistant strains were detected around the Cambodia‐Thailand border and in Colombia, in the 1950s.[110] These resistant strains spread rapidly, resulting in a large increase in mortality from this disease, particularly in Africa during the 1990s.[111]
Until the 1950s screening of anti malarial drugs was carried out on avian malaria. This was less than satisfactory as the avian malaria species differ in a number of ways from those that infect humans. The discovery in 1948 of Plasmodium berghei in wild rodents in the Congo[112] and later other rodent species that could infect laboratory rats transformed the tests used for drug development. The short hepatic phase and life cycle of these parasites made them extremely useful as animal models, a status they still retain.[57]
Plasmodium cynomolgi in Rhesus monkeys (Macaca mulatta) were used in the 1960s to test drugs active against P. vivax. This model is also still in use.
Genetically modified mice (non obese diabetic severe combined immunodeficient and BXN) can be engrafted with human stem cells and used as models for Plasmodium falciparum. Although the model is of variable reproducibility it has been used in some experiments.
Growth of the liver stages in animal free systems has been difficult but was achieved in the 1980s when P. berghei pre-erythrocytic stages was grown in wI38,a human embryonic lung cell line. This was followed by their growth in the human hepatoma line HepG2. Both P. falciparum and P. vivax have been grown in human liver cells; partial development of P. ovale in human liver cells has also been achieved; and P. malariae has been grown in chimpanzee and Aotus liver cells.
Systematic screening of traditional Chinese medical herbs was carried out by a number of Chinese research teams consisting of hundreds scientists in the 1960s and 1970s.[113] Qinghaosu, later named artemisinin, was cold extracted in a neutral milieu (pH 7.0) from the dried leaves of Artemisia annua inspired by Ge Hong's recommendation.[29][114]
Artemisinin was isolated by Tu Youyou, a Chinese pharmacologist. Tu headed a team of investigators who were tasked by the Chinese government with finding a treatment for choloroquine resistant malaria. Their work was known as Project 523 named after the date it was announced - May 23, 1967. The team investigated >2000 Chinese herb preparations and by 1971 had made 380 extracts from 200 herbs. An extract from qinghao (Artemisia annua) was effective but the results were variable. Tu reviewed the literature including the 340 BC book Zhou hou bei ji fang (A handbook of prescriptions for emergencies) by the Chinese physician Ge Hong. This book contained the only useful reference to the herb: "A handful of qinghao immersed with two litres of water, wring out the juice and drink it all." After making a non-toxic, neutral extract, Tu and two team members volunteered to take the extract before antimalarial trials were done in patients. The first studies were published in Chinese with the first English language paper citing successful trials for artemisinin appeared in 1979. The authors of this paper were anonymous according to Chinese custom at the time. Tu presented her findings to a United Nations scientific meeting in Beijing in 1981. Since then artemisinin has become a standard treatment for malaria.
Artemisinin is a sesquiterpene lactone containing a peroxide group, which is believed to be essential for its anti-malarial activity. Its derivatives, artesunate and artemether, have been used in clinics since 1987 for the treatment of drug-resistant and drug-sensitive malaria, in especially, cerebral malaria. These drugs are characterized by fast action, high efficacy and good tolerance. They kill the asexual forms of P. berghei and P. cynomolgi and have transmission-blocking activity.[115] In 1985, Zhou Yiqing and his team combined artemether and lumefantrine into a single tablet, which was registered as a new medicine in China in 1992, and later it became known as “Coartem”.[116] Artemisinin combination treatments (ACTs) are now widely used to treat uncomplicated falciparum malaria, but access to ACTs is still limited in most malaria-endemic countries and only a minority of the patients who need artemisinin-based combination treatments actually receive them.[117] Improved agricultural practices, selection of high-yielding hybrids, microbial production, and the development of synthetic peroxides will lower prices.[118][119]
Insecticides
Othmar Zeidler is credited with first synthesis of DDT (DichloroDiphenylTrichloroethane) in 1874.[120] The insecticidal properties of DDT were identified in 1939 by the chemist Paul Hermann Müller of the Swiss firm Geigy Pharmaceutical. For his discovery of the high efficiency of DDT as a contact poison against several arthropods he was awarded the Nobel Prize in Physiology or Medicine in 1948.[121] In the fall of 1942, samples of the chemical were acquired by the United States, Britain, and Germany and laboratory tests demonstrated that it was highly effective against insects. As the Rockefeller studies showed in Mexico, DDT remained effective for six to eight weeks if sprayed on the inside walls and ceilings of houses and other buildings.The first field test in which residual DDT was applied to the interior surfaces of all habitations and outbuildings was carried out in central Italy in the spring of 1944. The objective was to determine the residual effect of the spray upon anopheline density in the absence of other control measures. Spraying began in Castel Volturno and, after a few months, in the delta of the Tiber. The unprecedented effectiveness of the chemical was confirmed: the new insecticide was able to achieve the eradication of malaria through the eradication of mosquitoes. At the end of World War II a massive malaria control program based on DDT spraying was carried out in Italy. In Sardinia - the second largest island in the Mediterranean - between 1946 and 1951, the Rockefeller Foundation conducted a large-scale experiment to test the feasibility of the strategy of "species eradication" in an endemic malaria vector.[122] Malaria was effectively eliminated in the United States by the use of DDT in the National Malaria Eradication Program (1947–52). The concept of eradication prevailed in 1955 in the Eighth World Health Assembly: DDT was adopted as a primary tool in the fight against malaria.
DDT was banned in the US in 1972, after the discussion opened by the book of the American biologist Rachel Carson which launched the environmental movement in the West. The book catalogued the environmental impacts of the indiscriminate spraying of DDT and suggested that DDT and other pesticides may cause cancer and that their agricultural use was a threat to wildlife. Recently, the U.S. Congress, Republicans and Democrats alike, supports indoor DDT spraying as a vital component of any successful malaria control program, and the U.S. Agency for International Development has initiated DDT and other insecticide spraying programs in some poor tropical countries.[123]
A wide range of other insecticides is available for mosquito control in addition to the measures of draining of wetland breeding grounds and provision of better sanitation. Pyrethrum (Chrysanthemum [or Tanacetum ] cinerariaefolium) is an economically important source of natural insecticide. Pyrethrins attack the nervous systems of all insects. A few minutes after application the insect cannot move or fly away and female mosquitoes are inhibited from biting.[124] The use of pyrethrum in insecticide preparations dates back to Persia, about 400 BCE. Pyrethrins are non-persistent, being biodegradable and also breaking down easily on exposure to light. The majority of the world's supply of pyrethrin and Chrysanthemum cinerariaefolium comes from Kenya. The flower was first introduced into Kenya and the highlands of Eastern Africa during the late 1920s. The flowers of the plant are harvested shortly after blooming and are either dried and powdered or the oils within the flowers are extracted with solvents.
Cell culture
The first successful continuous malaria culture was established in 1976 by William Trager and James B. Jensen, which facilitated research into the molecular biology of the parasite and the development of new drugs substantially. By using increasing volumes of culture medium, one can grow P.falciparum to higher parasitemia (above 10%).[125][126]
Rapid diagnostic tests
The use of antigen-based malaria rapid diagnostic tests (RDTs) was pioneered in the 1980s.[127] Giemsa microscopy and RDTs represent the two diagnostics most likely to have the largest impact on malaria control today. Rapid diagnostic tests for malaria do not require any special equipment and offer the potential to extend accurate malaria diagnosis to areas when microscopy services are not available.[128][129]
21st century
Drug resistance poses a growing problem in the treatment of malaria in the 21st century, since resistance is now common against all classes of antimalarial drugs, with the exception of the artemisinins.[130] This situation has resulted in the treatment of resistant strains becoming increasingly dependent on this class of drugs. However, the artemisinins are expensive, which limits their use in the developing world.[118] Worrisome evidence is now emerging of malaria on the Cambodia-Thailand border that are resistant to combination therapies that include artemisinins, which raises the possibility that strains of malaria may have evolved that are untreatable with currently-available drugs.[131][132] Exposure of the parasite population to artemisinin monotherapies in subtherapeutic doses for over 30 years, and the availability of substandard artemisinins, have probably been the main driving force in the selection of the resistant phenotype in the region.[133]
The application of genomics to malaria research is now of central importance. With the sequencing of the three genomes of the malaria parasite P.falciparum, one of its vector Anopheles gambiae, and the human genome, the genetics of all three organisms in the malaria lifecycle can now be studied.[134] This breakthrough is expected to produce advances in the understanding of the interactions between the parasite and its human host—such as between virulence factors and the human immune system—as well as allowing the identification of the factors that restrict one species of parasite to one or a few species of mosquitoes. It is likely that these will eventually lead to new therapeutic approaches.[135][136] Another new application of genetic technology is the ability to produce genetically-modified mosquitoes that are unable to transmit malaria, allowing biological control of malaria transmission.[137]
The World Health Organization (WHO) recommends Indoor residual spraying as one of three primary means of malaria control, the others being use of insecticide-treated mosquito nets (ITNs) and prompt treatment of confirmed cases with artemisinin-based combination therapies (ACTs). In 2000, only 1.7 million (1.8%) African children living in stable malaria-endemic conditions were protected by an ITN. That number increased to 20.3 million (18.5%) African children using ITNs by 2007, leaving 89.6 million children unprotected.[138] An increased percentage of African households (31%) are estimated to own at least one ITN in 2008 (WHO World Malaria Report 2009). Most nets are impregnated with pyrethroids, a class of insecticides with particularly low toxicity. Dow AgroSciences developed a microencapsulated formulation of the organophosphate chlorpyrifos methyl as a cost-effective, long-lasting alternative to DDT. As an Indoor residual spraying against pyrethroid resistant mosquitoes chlorpyrifos methyl outperformed DDT and lambdacyhalothrin.[139] Organizations such as the Clinton Foundation continue to supply anti-malarial drugs to Africa and other affected areas; according to director Inder Singh, in 2011 more than 12 million individuals will be supplied with subsidized anti-malarial drugs.[140] Other organizations, such as Malaria No More continue distribution of more broad-based prophylaxis.
References
- ^ Poinar G (2005). "Plasmodium dominicana n. sp. (Plasmodiidae: Haemospororida) from Tertiary Dominican amber". Syst. Parasitol. 61 (1): 47–52. doi:10.1007/s11230-004-6354-6. PMID 15928991.
- ^ Joy DA, Feng X, Mu J, Furuya T, Chotivanich K, Krettli AU, Ho M, Wang A, White NJ, Suh E, Beerli P, Su XZ. (2003). "Early origin and recent expansion of Plasmodium falciparum". Science 300 (5617): 318–21. doi:10.1126/science.1081449. PMID 12690197.
- ^ Hayakawa T, Culleton R, Otani H, Horii T, Tanabe K (2008). "Big bang in the evolution of extant malaria parasites". Mol Biol Evol. 25 (10): 2233–9. doi:10.1093/molbev/msn171. PMID 18687771. http://mbe.oxfordjournals.org/cgi/reprint/25/10/2233.
- ^ Liu W, Li Y, Learn GH, Rudicell RS, Robertson JD, Keele BF, Ndjango J-BN, Sanz CM, Morgan DB, Locatelli S, Gonder MK, Kranzusch PJ, Walsh PD , Delaporte E, Mpoudi-Ngole E, Georgiev AV, Muller MN, Shaw GW, Peeters M, Sharp PM, Julian C. Rayner JC & Hahn BH (2010). "Origin of the human malaria parasite Plasmodium falciparum in gorillas". Nature 467 (7314): 420–5. doi:10.1038/nature09442. PMC 2997044. PMID 20864995. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2997044.
- ^ Hempelmann E, Tesarowicz I, Oleksyn BJ. (2009). "Kurzgefasste Geschichte der Malaria-Chemotherapie. Von Zwiebeln bis zum Artemisinin". Pharm Unserer Zeit 38 (6): 500–7. doi:10.1002/pauz.200900336. PMID 19862717.
- ^ Canali S (2008). "Researches on thalassemia and malaria in Italy and the origins of the "Haldane hypothesis"". Med Secoli 20 (3): 827–46. PMID 19848219. https://www.researchgate.net/publication/38028954_Researches_on_thalassemia_and_malaria_in_Italy_and_the_origins_of_the_Haldane_hypothesis.
- ^ Sallares R, Bouwman A, Anderung C (2004). "The Spread of Malaria to Southern Europe in Antiquity: New Approaches to Old Problems". Med Hist 48 (3): 311–28. PMC 547919. PMID 16021928. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=547919.
- ^ Allison AC. (2009). "Genetic control of resistance to human malaria". Curr Opin Immunol. 21 (5): 499–505. doi:10.1016/j.coi.2009.04.001. PMID 19442502.
- ^ Cox F (2002). "History of Human Parasitology". Clin Microbiol Rev 15 (4): 595–612. doi:10.1128/CMR.15.4.595-612.2002. PMC 126866. PMID 12364371. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=126866.
- ^ Tipton CM (2008). "Susruta of India, an unrecognized contributor to the history of exercise physiology". J Appl Physiol. 104 (6): 1553–6. doi:10.1152/japplphysiol.00925.2007. PMID 18356481. http://jap.physiology.org/cgi/reprint/104/6/1553.
- ^ Stagl V, Sattmann H, Hörweg C (2010). "Der Schrecken der Miasmen: Österreichische Forscher an Bord der Fregatte Novara auf den Spuren der Malaria". Wien Klin Wochenschr 122: 6–9. doi:10.1007/s00508-010-1431-0. PMID 20924706.
- ^ Bruni L (1476). "History of the Florentine People. Edited and Translated by James Hankins, 2004". Harvard University Press. 1-3.
- ^ Opeskin BR (2009). "Malaria in Pacific populations: seen but not heard?". Journal of Population Research 26 (2): 175–199. doi:10.1007/s12546-009-9011-8.
- ^ Dobson MJ (1994). "Malaria in England: a geographical and historical perspective". Parassitologia. 36 (1–2): 35–60. PMID 7898959.
- ^ Knottnerus O S (2002). "Malaria Around the North Sea: A Survey". Gerold Wefer, Wolfgang H. Berger, Karl-Ernst Behre, Eynstein Jansen (ed.), Climatic Development and History of the North Atlantic Realm: Hanse Conference Report. Springer-Verlag: 339–353. http://www.xs4all.nl/~ottoknot/werk/Malaria.html.
- ^ Reiter P (2000). "From Shakespeare to Defoe: malaria in England in the Little Ice Age". Emerg Infect Dis. 6 (1): 1–11. PMC 2627969. PMID 10653562. http://www.cdc.gov/ncidod/EID/vol6no1/reiter.htm.
- ^ Breman J (2001). "The ears of the hippopotamus: manifestations, determinants, and estimates of the malaria burden". Am J Trop Med Hyg 64 (1–2 Suppl): 1–11. PMID 11425172. http://www.ajtmh.org/cgi/content/abstract/64/1_suppl/1.
- ^ Worrall E, Basu S, Hanson K (2005). "Is malaria a disease of poverty? A review of the literature". Trop Med Int Health. 10 (10): 1047–59. doi:10.1111/j.1365-3156.2005.01476.x. PMID 16185240.
- ^ Parham PE, Christiansen-Jucht C, Pople D, Michael E (2011). "Understanding and Modelling the Impact of Climate Change on Infectious Diseases". InTech– Progress and Future Challenges. in Climate Change. Blanco J, Kheradmand H (eds) ISBN 979-953-307-277-6: 43–66. http://www.littleurl.net/103830.
- ^ a b Li Y, Wu YL. (2003). "An over four millennium story behind qinghaosu (artemisinin)--a fantastic antimalarial drug from a traditional Chinese herb". Curr Med Chem 10 (21): 2197–230. doi:10.2174/0929867033456710. PMID 14529339.
- ^ Brier B (2004). "Infectious diseases in ancient Egypt". Infect Dis Clin North Am 18 (1): 17–27. doi:10.1016/S0891-5520(03)00097-7. PMID 15081501.
- ^ Nerlich AG, Schraut B, Dittrich S, Jelinek T, Zink AR. (2008). "Plasmodium falciparum in Ancient Egypt". Emerg Infect Dis 14 (8): 1317–9. doi:10.3201/eid1408.080235. PMC 2600410. PMID 18680669. http://www.cdc.gov/eid/content/14/8/1317.htm.
- ^ Macaulay GC (1890). The History of Herodotus, parallel English/Greek translation. pp. Herodotus, 2.125. http://www.sacred-texts.com/cla/hh/hh2120.htm.
- ^ "History of Malaria Control". http://www.malariasite.com/malaria/history_control.htm. Retrieved 2009-10-27.
- ^ Hippocrates. Of the epidemics (Translated by Francis Adams) The Internet Classics Archive. http://classics.mit.edu//Hippocrates/epidemics.mb.txt.
- ^ Pappas G, Kiriaze IJ, Falagas ME (2008). "Insights into infectious disease in the era of Hippocrates". Int J Infect Dis. 12 (4): 347–50. doi:10.1016/j.ijid.2007.11.003. PMID 18178502.
- ^ Willcox ML, Bodeker G (2004). "Traditional herbal medicines for malaria". BMJ 329 (7475): 1156–9. doi:10.1136/bmj.329.7475.1156. PMC 527695. PMID 15539672. http://www.giftsofhealth.org/ritam/flyer/Willcox&Bodeker2004.pdf.
- ^ Hsu E (2006). "The history of qing hao in the Chinese materia medica". Trans R Soc Trop Med Hyg 100 (6): 505–8. doi:10.1016/j.trstmh.2005.09.020. PMID 16566952.
- ^ a b Wright CW, Linley PA, Brun R, Wittlin S, Hsu E (2010). "Ancient chinese methods are remarkably effective for the preparation of artemisinin-rich extracts of qing hao with potent antimalarial activity". Molecules 15 (2): 804–12. doi:10.3390/molecules15020804. PMID 20335947. http://www.mdpi.com/1420-3049/15/2/804/pdf.
- ^ Hsu E (2006). "Reflections on the 'discovery' of the antimalarial qinghao". Br J Clin Pharmacol 61 (6): 666–70. doi:10.1111/j.1365-2125.2006.02673.x. PMC 1885105/. PMID 16722826. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1885105/.
- ^ Li Y, Wu YL. (1998). "How Chinese scientists discovered qinghaosu (artemisinin) and developed its derivatives? What are the future perspectives?". Med Trop (Mars) 58 (3 Suppl): 9–12. PMID 10212890. https://www.researchgate.net/publication/13085247_How_Chinese_scientists_discovered_qinghaosu_%28artemisinin%29_and_developed_its_derivatives_What_are_the_future_perspectives.
- ^ Fornaciari G, Giuffra V, Ferroglio E, Gino S, Bianucci R (2010). "Plasmodium falciparum immunodetection in bone remains of members of the Renaissance Medici family (Florence, Italy, sixteenth century)". Trans R Soc Trop Med Hyg 104 (9): 583–7. doi:10.1016/j.trstmh.2010.06.007. PMID 20673935.
- ^ Adams M, Alther W, Kessler M, Kluge M, Hamburger M. (2011). "Malaria in the renaissance: Remedies from European herbals from the 16th and 17th century". J Ethnopharmacol 133 (2): 278–88. doi:10.1016/j.jep.2010.10.060. PMID 21056649.
- ^ De Castro MC, Singer BH (2005). "Was malaria present in the Amazon before the European conquest? Avaiable evidence and future research agenda". J Achaeol Sci 32 (3): 337–340. doi:10.1016/j.jas.2004.10.004.
- ^ Butler AR, Khan S, Ferguson E (2010). "A brief history of malaria chemotherapy". J R Coll Physicians Edinb 40 (2): 172–7. doi:10.4997/JRCPE.2010.216. PMID 20695174. http://www.rcpe.ac.uk/journal/issue/journal_40_2/butler.pdf.
- ^ Guay DR (2008). "Are there alternatives to the use of quinine to treat nocturnal leg cramps?". Consult Pharm 23 (2): 141–56. doi:10.4140/TCP.n.2008.141. PMID 18454580.
- ^ Guerra F. (1977). "The introduction of Cinchona in the treatment of malaria". J Trop Med Hyg. 80 (6): 112–118; 135–140. PMID 330870.
- ^ Greenwood D. (1992). "The quinine connection". J Antimicrob Chemother. 30 (4): 417–27. doi:10.1093/jac/30.4.417. PMID 1490916.
- ^ Kaufman T, Rúveda E (2005). "The quest for quinine: those who won the battles and those who won the war". Angew Chem Int Ed Engl 44 (6): 854–85. doi:10.1002/anie.200400663. PMID 15669029.
- ^ Bruce-Chwatt LJ (1988). "Three hundred and fifty years of the Peruvian fever bark". Br Med J (Clin Res Ed) 296 (6635): 1486–7. doi:10.1136/bmj.296.6635.1486. PMC 1833449. PMID 3134079. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1833449.[dead link]
- ^ Cook GC, Webb AJ. (2000). "Perceptions of malaria transmission before Ross' discovery in 1897". Postgrad Med J. 76 (901): 738–40. doi:10.1136/pmj.76.901.738. PMC 1741788. PMID 11060174. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1741788.
- ^ Pelletier PJ, Caventou C-B (1820). "Des Recherches chimiques sur les Quinguinas". Ann Chim Phys 15: 337.
- ^ Kyle RA, Shampe MA (1974). "Discoverers of quinine". JAMA 229 (4): 462. doi:10.1001/jama.229.4.462. PMID 4600403.
- ^ Siegel RE, Poynter FN (1962). "Robert Talbor, Charles II, and cinchona: a contemporary document". Med Hist 6 (1): 82–5. PMC 1034677. PMID 16562233. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1034677.
- ^ Gramiccia G (1987). "Ledger's cinchona seeds: a composite of field experience, chance, and intuition". Parassitologia 29 (2–3): 207–20. PMID 3334083.
- ^ Maclean WC (1875). "Professor Maclean, C.B., on the true composition and therapeutic value of Warburg's Tincture". Lancet 106 (2724): 716–718. doi:10.1016/S0140-6736(02)30835-3.
- ^ Poser CM, Bruyn GW. (1999). "An Illustrated History of Malaria, p. 87". New York: Parthenon, ISBN 978-1850700685 (Informa Health Care). ISBN 9781850700685. http://books.google.com/?id=N4fyLSwLP3MC&dq=%22An+illustrated+history+of+malaria%22&printsec=frontcover&q=.
- ^ Seeman JI (2007). "The Woodward-Doering/Rabe-Kindler total synthesis of quinine: setting the record straight". Angew Chem Int Ed Engl. 46 (9): 1378–413. doi:10.1002/anie.200601551. PMID 17294412.
- ^ Guttmann P, Ehrlich P (1891). "Ueber die Wirkung des Methylenblau bei Malaria". Berliner Klinische Wochenschrift 39: 953–956. http://www.pei.de/cln_092/nn_164284/SharedDocs/Downloads/institut/paul-ehrlich/1886-1896/1891-wirkung-methylenblau-malaria,templateId=raw,property=publicationFile.pdf/1891-wirkung-methylenblau-malaria.pdf.
- ^ Wainwright M, Amaral L (2005). "The phenothiazinium chromophore and the evolution of antimalarial drugs". Trop Med Int Health 10 (6): 501–11. doi:10.1111/j.1365-3156.2005.01417.x. PMID 15941412. http://www3.interscience.wiley.com/cgi-bin/fulltext/118667658/PDFSTART.
- ^ Giemsa G (1904). "Eine Vereinfachung und Vervollkommnung meiner Methylenazur-Methylenblau-Eosin-Färbemethode zur Erzielung der Romanowsky-Nochtschen Chromatinfärbung". Zentralblatt für Bakteriologie 37: 308–311.
- ^ Woronzoff-Dashkoff KK. (2002). "The wright-giemsa stain. Secrets revealed". Clin Lab Med. 22 (1): 15–23. doi:10.1016/S0272-2712(03)00065-9. PMID 11933573.
- ^ Bosch F, Rosich L (2008). "The Contributions of Paul Ehrlich to Pharmacology: A Tribute on the Occasion of the Centenary of His Nobel Prize". Pharmacology. 82 (3): 171–9. doi:10.1159/000149583. PMC 2790789. PMID 18679046. http://content.karger.com/ProdukteDB/produkte.asp?Aktion=ShowPDF&ArtikelNr=000149583&Ausgabe=239973&ProduktNr=224274&filename=000149583.pdf.
- ^ Laveran CLA (1880). "Note sur un nouveau parasite trouvé dans le sang de plusieurs malades atteints de fièvre palustres". Bull Acad Med 9: 1235–1236.
- ^ Lorber CG, Lorber CP, Schneider J (2005). "Die Medizinerfamilie Meckel aus Wetzlar". Hess Aerzteblatt 2: 95–99. http://www.laekh.de/upload/Hess._Aerzteblatt/2005/2005_02/2005_02_08.pdf.
- ^ Meckel H. (1847). "Ueber schwarzes Pigment in der Milz und dem Blute einer Geisteskranken". Zeitschrift für Psychiatrie IV: 198–226.
- ^ a b c d Cox FE (2010). "History of the discovery of the malaria parasites and their vectors". Parasites & Vectors 3 (1): 5. doi:10.1186/1756-3305-3-5. PMC 2825508. PMID 20205846. http://www.parasitesandvectors.com/content/pdf/1756-3305-3-5.pdf.
- ^ Smith DC, Sanford LB. (1985). "Laveran's germ: the reception and use of a medical discovery". Am J Trop Med Hyg 34 (1): 2–20. PMID 2578751.
- ^ Golgi C (1893). "Sulle febbri malariche estivo-autumnali di Roma". Gass Med di Pavia 2: 481–493, 505–520, 529–544, 553–559.
- ^ Pel PK (1886). "Mededeelingen uit de Geneeskundige kliniek. Malaria infectie". Ned Tijdschr Geneeskd 22: 341–358.
- ^ Grassi B and Feletti R (1890) Parasites malariques chez les oiseaux. Arch. Ital. Biol. 13:297-300.
- ^ "Biography of Alphonse Laveran". The Nobel Foundation. http://nobelprize.org/nobel_prizes/medicine/laureates/1907/laveran-bio.html. Retrieved 2007-06-15.
- ^ Doetsch RN (1964). "John Crawford and his contribution to the doctrine of contagium vivum". Bacteriol Rev 28 (1): 87–96. PMC 441211. PMID 14130055. http://mmbr.asm.org/cgi/reprint/28/1/87.pdf.
- ^ Manson P (1894). "On the nature and significance of the crescentic and flagellated bodies in malarial blood". Brit Med J 2 (4849): 1306–8. doi:10.1136/bmj.2.1771.1306. PMC 2405325. PMID 20755205. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2405325.
- ^ King AFA (1883). "Insects and disease, mosquitoes and malaria". Popular Sci. Monthly, Sep. 23: 644–658.
- ^ a b "A History of Mosquitoes in Massachusetts, by Curtis R. Best". Northeast Mosquito Control Association. http://www.nmca.org/Nmca93-4.htm. Retrieved 2008-03-31.
- ^ "Biography of Ronald Ross". The Nobel Foundation. http://nobelprize.org/nobel_prizes/medicine/laureates/1902/ross-bio.html. Retrieved 2007-06-15.
- ^ Capanna E (2006). "Grassi versus Ross: who solved the riddle of malaria?". Int Microbiol. 9 (1): 69–74. PMID 16636993. http://scielo.isciii.es/scielo.php?pid=S1139-67092006000100010&script=sci_arttext.
- ^ "Ross and the Discovery that Mosquitoes Transmit Malaria Parasites". CDC Malaria website. Archived from the original on June 2, 2007. http://web.archive.org/web/20070602185153/http://www.cdc.gov/malaria/history/ross.htm. Retrieved 2007-06-15.
- ^ Sutter PS (2007). "Nature's agents or agents of empire? Entomological workers and environmental change during the construction of the Panama Canal". Isis. 98 (4): 724–54. doi:10.1086/529265. PMID 18314643.
- ^ Thayer W. (1898). "Lectures on the malarial fevers". D.Appleton & Co., New York (Adegi Graphics LLC). ISBN 9780543912367. http://books.google.com/?id=Woqcfoq5dwoC&dq=%22Lectures+on+the+malarial+fevers%22&printsec=frontcover&q=.
- ^ Krafts K, Hempelmann E, Oleksyn BJ (2011). "In search of the malarial parasite : Biographical sketches of the blood stain contributors". Parasitol Research 109 (3): 521–529. doi:10.1007/s00436-011-2475-4. PMID 21660627.
- ^ Malachowski E (1891). "Zur Morphologie des Plasmodium malariae". Centbl f klin Med 31: 601–603.
- ^ Romanowsky D (1891). "Zur Frage der Parasitologie und Therapie der Malaria". St Petersburg Med Wochenschr 16: 297–302, 307–315.
- ^ Horobin RW, Walter KJ (1987). "Understanding Romanowsky staining. I: The Romanowsky-Giemsa effect in blood smears". Histochemistry 86 (3): 331–6. PMID 2437082. https://www.researchgate.net/publication/20719192_Understanding_Romanowsky_staining._I_The_Romanowsky-Giemsa_effect_in_blood_smears.
- ^ Woronzoff-Dashkoff KK. (2002). "The wright-giemsa stain. Secrets revealed". Clin Lab Med. 22 (1): 15–23. doi:10.1016/S0272-2712(03)00065-9. PMID 11933573. https://www.researchgate.net/publication/11431311_The_wright-giemsa_stain._Secrets_revealed.
- ^ Krafts K, Hempelmann E, Oleksyn B (2011). "The color purple: from royalty to laboratory, with apologies to Malachowski". Biotech Histochem 86 (1): 7–35. doi:10.3109/10520295.2010.515490. PMID 21235291.
- ^ Manson P (2002). "Experimental proof of the mosquito-malaria theory. 1900". Originally published in Br Med J 1900;2:949-951; reprint in: Yale J Biol Med 75 (2): 107–12. PMC 2588736. PMID 12230309. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2588736.
- ^ Manson PT (1901). "Experimental Malaria: Recurrence after Nine Months". Br Med J 2 (2115): 77. doi:10.1136/bmj.2.2115.77. PMC 2505910. PMID 20759742. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2505910.
- ^ Bignami A, Bastianelli G (1900). "Sulla inoculazione delle sangue di semiluna malariche d'uomo". Atti Soc Studi Malar 1: 15–20.
- ^ Ross R, Thompson JG (1910). "Some enumeration studies on malarial fever". Ann Trop Med Parasitol 4: 267–306.
- ^ Schaudinn F (1903). "Studien über krankheitserregende Protozoen. II. Plasmodium vivax (Grassi & Feletti), der Erreger des Tertianfiebers beim Menschen". Arb Kaiserl Gesundheits 19: 169–250.
- ^ James SP, Tate P (1938). "Exo-erythrocytic schizogony in Plasmodium gallinaceum Brumpt, 1935". Parasitology 30: 128–138. doi:10.1017/S0031182000010891.
- ^ Mesnil F, Roubaud E (1920). "Essais d'inoculation du paludisme au chimpanzé". Ann Inst Pasteur, Paris 34: 466–480.
- ^ Marchoux E (1926). "Paludisme". J. B. Bailliere, Paris..
- ^ James SP (1931). "The use of plasmoquine in the prevention of malarial infections". Proc R Acad Sci Amst 34: 1424–1425.
- ^ Huff CG, Bloom W (1935). "A Malarial Parasite Infecting All Blood and Blood-Forming Cells of Birds". J Infect Dis (The Journal of Infectious Diseases, Vol. 57, No. 3) 57 (3): 315–336. doi:10.1093/infdis/57.3.315. JSTOR 30088998.
- ^ Fairley NH (1945). "Chemotherapeutic suppression and prophylaxis in malaria". Trans R Soc Trop Med Hyg 38 (5): 311–65. doi:10.1016/0035-9203(45)90038-1. PMID 20293965.
- ^ Shute PG (1946). "Latency and long-term relapses in benign tertian malaria". Trans R Soc Trop Med Hyg 40 (2): 189–200. doi:10.1016/0035-9203(46)90056-9. PMID 20275230. http://www.tropicalmedandhygienejrnl.net/article/0035-9203%2846%2990056-9/abstract.
- ^ Sapero JJ (1947). "New Concepts in the Treatment of Relapsing Malaria". Am J Trop Med Hyg 27 (3): 271–283. http://www.ajtmh.org/cgi/content/abstract/s1-27/3/271.
- ^ Garnham PCC (1947). "Exoerythrocytic schizogony in Plasmodium kochi laveran. A preliminary note". Trans R Soc Trop Med Hyg 40 (5): 719–22. doi:10.1016/0035-9203(47)90029-1. PMID 20243887.
- ^ Shortt HE, Garnham PCC (1948). "Pre-erythrocytic stage in mammalian malaria parasites". Nature 161 (4082): 126. doi:10.1038/161126a0. PMID 18900752. http://www.nature.com/nature/journal/v161/n4082/pdf/161126a0.pdf.
- ^ Shortt HE, Fairley NH, Covell G, Shute PG, Garnham PCC (1948). "Pre-erythrocytic Stage of Plasmodium Falciparum". Br Med J 2 (4635): 1006–8, illust. doi:10.1136/bmj.2.3282.1006-c. PMC 2051640. PMID 15393036. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2051640.
- ^ Cogswell FB (1992). "The hypnozoite and relapse in primate malaria". Clin. Microbiol. Rev. 5 (1): 26–35. PMC 358221. PMID 1735093. http://cmr.asm.org/cgi/pmidlookup?view=long&pmid=1735093.
- ^ Krotoski WA, Collins WE, Bray RS, Garnham PCC, Cogswell FB, Gwadz RW, Killick-Kendrick R, Wolf R, Sinden R, Koontz LC, Stanfill PS (1982). "Demonstration of hypnozoites in sporozoite-transmitted Plasmodium vivax infection". Am J Trop Med Hyg 31 (6): 1291–3. PMID 6816080. http://www.ajtmh.org/cgi/content/abstract/31/6/1291.
- ^ Markus MB (2010). "Malaria: Origin of the Term "Hypnozoite"". J Hist Biol. doi:10.1007/s10739-010-9239-3. PMID 20665090.
- ^ Markus MB (2011). "The hypnozoite concept, with particular reference to malaria". Parasitol Res 108 (1): 247–52. doi:10.1007/s00436-010-2072-y. PMID 20924610.
- ^ Wagner-Jauregg J. (1931). "Verhütung und Behandlung der Progressiven Paralyse durch Impfmalaria". Handbuch der experimentellen Therapie, Ergänzungsband München.
- ^ Frankenburg FR, Baldessarini RJ (2008). "Neurosyphilis, malaria, and the discovery of antipsychotic agents". Harv Rev Psychiatry. 16 (5): 299–307. doi:10.1080/10673220802432350. PMID 18803105.
- ^ "The Nobel Prize in Physiology or Medicine 1927". The Nobel Foundation. http://nobelprize.org/nobel_prizes/medicine/laureates/1927/index.html. Retrieved 2007-07-28.
- ^ Spielman, Andrew; D'Antonio, Michael (2002). Mosquito: The Story of Man's Deadliest foe. Hyperion. p. 131. ISBN 9780786886678.
- ^ Parmakelis A, Russello MA, Caccone A, Marcondes CB, Costa J, Forattini OP, Sallum MA, Wilkerson RC, Powell JR (2008). "Historical analysis of a near disaster: Anopheles gambiae in Brazil". Am J Trop Med Hyg 78 (1): 176–8. PMID 18187802. http://www.ajtmh.org/cgi/reprint/78/1/176.
- ^ Russell PF, West LS, Manwell RD, MacDonald G (1963). "Practical Malariology 2nd ed". Oxford University Press, London, New York, Toronto.
- ^ Hempelmann E. (2007). "Hemozoin biocrystallization in Plasmodium falciparum and the antimalarial activity of crystallization inhibitors". Parasitol Res 100 (4): 671–6. doi:10.1007/s00436-006-0313-x. PMID 17111179. http://parasitology.informatik.uni-wuerzburg.de/login/n/h/j_436-100-4-2006-11-17-313.html.
- ^ Jensen M, Mehlhorn H (2009). "Seventy-five years of Resochin in the fight against malaria". Parasitol Res 105 (3): 609–27. doi:10.1007/s00436-009-1524-8. PMID 19593586. http://www.springerlink.com/content/y161240k51740643/fulltext.pdf.
- ^ Coatney GR (1963). "Pitfalls in a Discovery: The Chronicle of Chloroquine". Am J Trop Med Hyg. 12 (2): 121–8. PMID 14021822. http://www.ajtmh.org/cgi/content/abstract/12/2/121.
- ^ Sutton AC (2000). Wall Street and the Rise of Hitler. Chapter II The Empire of I.G. Farben. http://reformed-theology.org/html/books/wall_street/.
- ^ Loeb RF, Clark WM, Coatney GR, Coggeshall LT, Dieuaide FR, Dochez AR, Hakansson EG, Marshall EK, Marvel SC, McCoy OR, Sapero JJ, Sebrell WH, Shannon JA, Carden GA (1946). "Activity of a new antimalarial agent, chloroquine (SN 7618)". J.Am.Med.Assoc. 130 (16): 1069–1070. doi:10.1001/jama.1946.02870160015006 (inactive 2010-09-04). http://jama.ama-assn.org/cgi/content/summary/130/16/1069.
- ^ Wellems TE, Plowe CV (2001). "Chloroquine-resistant malaria". J. Infect. Dis. 184 (6): 770–6. doi:10.1086/322858. PMID 11517439. http://www.journals.uchicago.edu/doi/pdf/10.1086/322858.
- ^ Payne D (1987). "Spread of chloroquine resistance in Plasmodium falciparum". Parasitol. Today (Regul. Ed.) 3 (8): 241–6. doi:10.1016/0169-4758(87)90147-5. PMID 15462966. http://linkinghub.elsevier.com/retrieve/pii/0169475887901475.
- ^ Snow RW, Trape JF, Marsh K (2001). "The past, present and future of childhood malaria mortality in Africa". Trends Parasitol. 17 (12): 593–7. doi:10.1016/S1471-4922(01)02031-1. PMID 11756044.
- ^ Vincke IH, Lips M (1948). "Un nouveau plasmodium d'un rongeur sauvage du Congo: Plasmodium berghei n.sp". Annales de la Société Belge de Médecine Tropicale 28: 97–104.
- ^ Li Y, Wu Y (2010). "A golden phoenix arising from the herbal nest — A review and reflection on the study of antimalarial drug Qinghaosu". Frontiers of Chemistry in China 5 (4): 357–422. doi:10.1007/s11458-010-0214-5.
- ^ Liao F (2009). "Discovery of Artemisinin (Qinghaosu)". Molecules 14 (12): 5362–5366. doi:10.3390/molecules14125362. http://www.mdpi.com/1420-3049/14/12/5362/pdf.
- ^ Chotivanich K, Sattabongkot J, Udomsangpetch R, Looareesuwan S, Day NP, Coleman RE, White NJ (2006). "Transmission-Blocking Activities of Quinine, Primaquine, and Artesunate". Antimicrob Agents Chemother 50 (6): 1927–30. doi:10.1128/AAC.01472-05. PMC 1479118. PMID 16723547. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1479118.
- ^ Weiyuan C (2009). "Ancient Chinese anti-fever cure becomes panacea for malaria". Bull World Health Organ 87 (10): 743–4. doi:10.2471/BLT.09.051009. PMC 2755319. PMID 19876540. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2755319.
- ^ Nosten F, White NJ (2007). "Artemisinin-based combination treatment of falciparum malaria". Am J Trop Med Hyg. 77 (6): 181–92. PMID 18165491. http://www.ajtmh.org/cgi/reprint/77/6_Suppl/181.
- ^ a b White NJ (2008). "Qinghaosu (artemisinin): the price of success". Science. 320 (5874): 330–4. doi:10.1126/science.1155165. PMID 18420924.
- ^ Hale V, Keasling JD, Renninger N, Diagana TT (2007). "Microbially derived artemisinin: a biotechnology solution to the global problem of access to affordable antimalarial drugs". Am J Trop Med Hyg. 77 (6 Suppl): 198–202. PMID 18165493. http://www.ajtmh.org/cgi/content/abstract/77/6_Suppl/198.
- ^ Zeidler O (1874). "Verbindungen von Chloral mit Brom- und Chlorbenzol". Berichte der deutschen chemischen Gesellschaft 7 (2): 1180–1181. doi:10.1002/cber.18740070278.
- ^ "The Nobel Prize in Physiology or Medicine 1948". The Nobel Foundation. http://nobelprize.org/nobel_prizes/medicine/laureates/1948/index.html. Retrieved 2007-07-28.
- ^ Tognotti E (2009). "Program to Eradicate Malaria in Sardinia, 1946–1950". Emerg Infect Dis. 15 (9): 1460–6. PMC 2819864. PMID 19788815. http://www.cdc.gov/eid/content/15/9/pdfs/08-1317.pdf.
- ^ Sadasivaiah S, Tozan Y, Breman JG (2007). "Dichlorodiphenyltrichloroethane (DDT) for indoor residual spraying in Africa: how can it be used for malaria control?". Am. J. Trop. Med. Hyg. 77 (6): 249–63. PMID 18165500. http://www.ajtmh.org/cgi/content/abstract/77/6_Suppl/249.
- ^ Duchon S, Bonnet J, Marcombe S, Zaim M, Corbel V (2009). "Pyrethrum: a mixture of natural pyrethrins has potential for malaria vector control". J Med Entomol. 46 (3): 516–22. doi:10.1603/033.046.0316. PMID 19496422. http://www.bioone.org/doi/abs/10.1603/033.046.0316.
- ^ Trager W, Jensen JB (1976). "Human malaria parasites in continuous culture". Science 193 (4254): 673–5. doi:10.1126/science.781840. PMID 781840.
- ^ Schuster FL (2002). "Cultivation of Plasmodium spp". Clin Microbiol Rev. 15 (3): 355–64. doi:10.1128/CMR.15.3.355-364.2002. PMC 118084. PMID 12097244. http://cmr.asm.org/cgi/reprint/15/3/355.
- ^ Ling IT, Cooksley S, Bates PA, Hempelmann E, Wilson RJM (1986). "Antibodies to the glutamate dehydrogenase of Plasmodium falciparum". Parasitology 92 (2): 313–24. doi:10.1017/S0031182000064088. PMID 3086819.
- ^ Makler MT, Piper RC (2009). "Rapid malaria tests: where do we go after 20 years?". Am J Trop Med Hyg 81 (6): 921–6. doi:10.4269/ajtmh.2009.09-0202. PMID 19996417. http://www.ajtmh.org/cgi/content/abstract/81/6/921.
- ^ Bisoffi Z, Gobbi F, Angheben A, Van den Ende J (2009). "The Role of Rapid Diagnostic Tests in Managing Malaria". PLoS Med 6 (4): e1000063. doi:10.1371/journal.pmed.1000063. PMC 2667642. PMID 19399160. http://www.plosmedicine.org/article/info:doi/10.1371/journal.pmed.1000063.
- ^ White NJ (2004). "Antimalarial drug resistance". J. Clin. Invest. 113 (8): 1084–92. doi:10.1172/JCI21682. PMC 385418. PMID 15085184. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=385418.
- ^ Wongsrichanalai C, Meshnick SR (2008). "Declining Artesunate-Mefloquine Efficacy against Falciparum Malaria on the Cambodia–Thailand Border". Emerging Infect. Dis. 14 (5): 716–9. doi:10.3201/eid1405.071601. PMC 2600243. PMID 18439351. http://www.cdc.gov/eid/content/14/5/716.htm.
- ^ Vijaykadga S, Rojanawatsirivej C, Cholpol S, Phoungmanee D, Nakavej A, Wongsrichanalai C (2005). "In vivo sensitivity monitoring of mefloquine monotherapy and artesunate-mefloquine combinations for the treatment of uncomplicated falciparum malaria in Thailand in 2003". Trop Med Int Health 11 (2): 211–9. doi:10.1111/j.1365-3156.2005.01557.x. PMID 16451346.
- ^ Dondorp AM, Yeung S, White L, Nguon C, Day NPJ, Socheat D, von Seidlein L (2010). "Artemisinin resistance: current status and scenarios for containment". Nat Rev Microbiol 8 (8): 272–80. doi:10.1038/nrmicro2331. PMID 20208550.
- ^ Aultman KS, Gottlieb M, Giovanni MY, Fauci AS (2002). "Anopheles gambiae genome: completing the malaria triad". Science 298 (5591): 13. doi:10.1126/science.298.5591.13. PMID 12364752. http://www.sciencemag.org/cgi/content/summary/298/5591/13.
- ^ Winzeler EA. (2008). "Malaria research in the post-genomic era". Nature 455 (7214): 751–6. doi:10.1038/nature07361. PMC 2705782. PMID 18843360. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2705782.
- ^ Sahu NK, Sahu S, Kohli DV. (2008). "Novel molecular targets for antimalarial drug development". Chem Biol Drug Des. 71 (4): 287–97. doi:10.1111/j.1747-0285.2008.00640.x. PMID 18298458.
- ^ Ito J, Ghosh A, Moreira LA, Wimmer EA, Jacobs-Lorena M (2002). "Transgenic anopheline mosquitoes impaired in transmission of a malaria parasite". Nature 417 (6887): 452–5. doi:10.1038/417452a. PMID 12024215.
- ^ Noor AM, Mutheu JJ, Tatem AJ, Hay SI, Snow RW (2009). "Insecticide-treated net coverage in Africa: mapping progress in 2000–07". Lancet 373 (9657): 58–67. doi:10.1016/S0140-6736(08)61596-2. PMC 2652031. PMID 19019422. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2652031.
- ^ N'guessan R, Boko P, Odjo A, Chabi J, Akogbeto M, Rowland M (2010). "Control of pyrethroid and DDT-resistant Anopheles gambiae by application of indoor residual spraying or mosquito nets treated with a long-lasting organophosphate insecticide, chlorpyrifos-methyl". Malar J 9 (1): 44. doi:10.1186/1475-2875-9-44. PMC 2831012. PMID 20141626. http://www.malariajournal.com/content/pdf/1475-2875-9-44.pdf.
- ^ Harvard-MIT Health Sciences. "BEP grad Inder Singh orchestrates affordable malaria drug development". http://hst.mit.edu/servlet/ControllerServlet?handler=PublicHandler&action=browse&pageId=2261. Retrieved 16 August 2011.
Further reading
- Robert Sallares (2002) Malaria and Rome: A History of Malaria in Ancient Italy, New York, N.Y.: Oxford University Press, ISBN 0199248508 / ISBN 978-0199248506
- Frank M. Snowden (2005) The Conquest of Malaria: Italy, 1900-1962, Yale Univ Press; illustrated edition, ISBN 0300108990 / ISBN 978-0300108996
- Randall M. Packard (2007) The Making of a Tropical Disease, The Johns Hopkins University Press; Baltimore, ISBN 0801887127 / ISBN 978-0801887123
- Irwin W. Sherman (2009) The Elusive Malaria Vaccine: Miracle or Mirage? , ASM Press, ISBN 978-1555815158
- Yip, Ka-Che (editor) (2009) Disease, Colonialism, and the State: Malaria in Modern East Asian History, Hong Kong University Press, ISBN 9622095879 / ISBN 978-9622095878
- Leo B. Slater (2009) War and Disease: Biomedical Research on Malaria in the Twentieth Century , Rutgers University Press, ISBN 0813544386 / ISBN 978-0813544380
- James L. A. Webb, Jr. (2009) Humanity’s Burden: A Global History of Malaria, Cambridge University Press, ISBN 978-0521670128
- Irwin W. Sherman (2010) Magic Bullets to Conquer Malaria. From Quinine to Qinghaosu, ASM Press, ISBN 978-1555815431
- Elisabeth Hsu, Stephen Harris (editors) (2010) Plants, Health and Healing: On the Interface of Ethnobotany and Medical Anthropology, Berghahn Books, ISBN 978-1845450601
External links
- Alphonse Laveran Nobel Lecture
- Grassi versus Ross
- Julius Wagner-Jauregg Nobel Lecture
- Malaria and the Fall of Rome
- Malaria Around the North Sea Malaria may have been introduced into the North Sea Basin in late Antiquity. It has been endemic at least since the 7th century, but its high-days were the Little Ice Age. After 1750 the disease retreated until it disappeared in the 1950s.
- Malariasite
- Malaria Website
- Medicines for Malaria Venture
- Medline Plus - Malaria
- Paul H Müller Nobel Lecture
- Ronald Ross Nobel Lecture
- The Global Fund to Fight AIDS, Tuberculosis and Malaria
- The malaria genome
- Treatment of Malaria in the United States
- UK malaria treatment guidelines
- Wellcome images 2000 years of human culture, images from the Wellcome Library
- WHO Malaria Control
- World Health Organisation (2006) Guidelines for the Treatment of Malaria
- World Malaria Map: Plasmodium falciparum Endemicity in 2007
- World Malaria Report 2009 Malaria is on the decline in various parts of the world, more than one-third of the 108 malarious countries documented reduction in malaria cases of app 50% in 2008 compared to 2000
Categories:
Wikimedia Foundation. 2010.