- Aluminium chloride
-
Aluminium chloride 
 aluminium chlorideOther namesaluminium(III) chloride
aluminium chlorideOther namesaluminium(III) chlorideIdentifiers CAS number 7446-70-0 (anhydrous)  ,
,
10124-27-3 (hydrate),
7784-13-6 (hexahydrate)PubChem 24012 ChemSpider 22445 
UNII LIF1N9568Y 
ChEBI CHEBI:30114 
RTECS number BD0530000 Jmol-3D images Image 1
Image 2- Cl[Al](Cl)Cl
[Al](Cl)(Cl)Cl
Properties Molecular formula AlCl3 Molar mass 133.34 g/mol (anhydrous)
241.43 g/mol (hexahydrate)Appearance white or pale yellow solid,
hygroscopicDensity 2.48 g/cm3 (anhydrous)
1.3 g/cm3 (hexahydrate)Melting point 192.4 °C *(anhydrous)
0 °C (hexahydrate)Boiling point 120 °C (hexahydrate)
Solubility in water 43.9 g/100 ml (0 °C)
44.9 g/100 ml (10 °C)
45.8 g/100 ml (20 °C)
46.6 g/100 ml (30 °C)
47.3 g/100 ml (40 °C)
48.1 g/100 ml (60 °C)
48.6 g/100 ml (80 °C)
49 g/100 ml (100 °C)Solubility soluble in hydrogen chloride, ethanol, chloroform, carbon tetrachloride
slightly soluble in benzeneStructure Crystal structure Monoclinic, mS16 Space group C12/m1, No. 12 Coordination
geometryOctahedral (solid)
Tetrahedral (liquid)Molecular shape Trigonal planar
(monomeric vapour)Hazards MSDS External MSDS EU classification Corrosive (C) R-phrases R34 S-phrases (S1/2), S7/8, S28, S45 LD50 anhydrous:
380 mg/kg, rat (oral)
hexahydrate:
3311 mg/kg, rat (oral)Related compounds Other anions Aluminium fluoride
Aluminium bromide
Aluminium iodideOther cations Boron trichloride
Gallium trichloride
Indium(III) chloride
Magnesium chlorideRelated Lewis acids Iron(III) chloride
Boron trifluorideSupplementary data page Structure and
propertiesn, εr, etc. Thermodynamic
dataPhase behaviour
Solid, liquid, gasSpectral data UV, IR, NMR, MS  chloride (verify) (what is:
chloride (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Aluminium chloride (AlCl3) is the main compound of aluminium and chlorine. It is white, but samples are often contaminated with iron trichloride, giving it a yellow colour. The solid has a low melting and boiling point. It is mainly produced and consumed in the production of aluminium metal, but large amounts are also used in other areas of chemical industry. The compound is often cited as a Lewis acid. It is an example of an inorganic compound that "cracks" at mild temperature, reversibly changing from a polymer to a molecule.
Contents
Structure
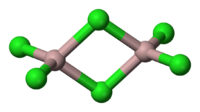
AlCl3 adopts three different structures, depending on the temperature and the state (solid, liquid, gas). Solid AlCl3 is a sheet-like layered cubic close packed layers. In this framework, the Al centres exhibit octahedral coordination geometry.[1] In the melt, aluminium trichloride exists as the dimer Al2Cl6, with tetracoordinate aluminium. This change in structure is related to the lower density of the liquid phase (1.78 g/cm3) vs solid aluminium trichloride (2.48 g/cm3). Al2Cl6 dimers are also found in the vapour phase. At higher temperatures, the Al2Cl6 dimers dissociate into trigonal planar AlCl3, which is structurally analogous to BF3. The melt conducts electricity poorly,[2] unlike more ionic halides such as sodium chloride.
Reactions
Anhydrous aluminium chloride is a powerful Lewis acid, capable of forming Lewis acid-base adducts with even weak Lewis bases such as benzophenone and mesitylene.[3] It forms tetrachloroaluminate AlCl4− in the presence of chloride ions.
Aluminium chloride reacts with calcium and magnesium hydrides in tetrahydrofuran forming tetrahydroaluminates:
Reactions with water
Aluminium chloride is hygroscopic, having a very high affinity for water. It fumes in moist air and hisses when mixed with liquid water as the Cl- ions are displaced with H2O molecules in the lattice to form the hexahydrate AlCl3·6H2O (also white to yellowish in color). The anhydrous phase cannot be regained on heating as HCl is lost leaving aluminum hydroxide or alumina (aluminum oxide):
- Al(H2O)6Cl3 → Al(OH)3 + 3 HCl + 3 H2O
On strong heating (~400°C), the aluminum oxide is formed from the aluminum hydroxide via:
- Al(OH)3 → Al2O3 + 3 H2O
Aqueous solutions of AlCl3 are ionic and thus conduct electricity well. Such solutions are found to be acidic, indicative of partial hydrolysis of the Al3+ ion. The reactions can be described (simplified) as:
- [Al(H2O)6]3+ ⇌ [Al(OH)(H2O)5]2+ + H+
Aqueous solutions behave similarly to other aluminium salts containing hydrated Al3+ ions, giving a gelatinous precipitate of aluminium hydroxide upon reaction with sodium hydroxide:
- AlCl3 + 3 NaOH → Al(OH)3 + 3NaCl
Synthesis
Aluminium chloride is manufactured on a large scale by the exothermic reaction of aluminium metal with chlorine or hydrogen chloride at temperatures between 650 to 750 °C.[2]
- 2 Al + 3 Cl2 → 2 AlCl3
- 2 Al + 6 HCl → 2 AlCl3 + 3 H2
In the US in 1993, approximately 21,000 tons were produced, not counting the amounts consumed in the production of aluminium.[4]
Hydrated aluminium trichloride is prepared by dissolving aluminium oxides in hydrochloric acid. Heating this solid does not produce anhydrous aluminium trichloride, the hexahydrate decomposes to aluminium oxide when heated to 300 °C:[4]
- 2 AlCl3 + 3 H2O → Al2O3 + 6 HCl
Aluminium also forms a lower chloride, aluminium(I) chloride (AlCl), but this is very unstable and only known in the vapor phase.[2]
Uses
Anhydrous aluminium trichloride
AlCl3 is probably the most commonly used Lewis acid and also one of the most powerful. It finds widespread application in the chemical industry as a catalyst for Friedel–Crafts reactions, both acylations and alkylations. Important products are detergents and ethylbenzene. It also finds use in polymerization and isomerization reactions of hydrocarbons.
The Friedel–Crafts reaction[3] is the major use for aluminium chloride, for example in the preparation of anthraquinone (for the dyestuffs industry) from benzene and phosgene.[2] In the general Friedel-Crafts reaction, an acyl chloride or alkyl halide reacts with an aromatic system as shown:[3]
The alkylation reaction is more widely used than the acylation reaction, although its practice is more technically demanding because the reaction is more sluggish. For both reactions, the aluminium chloride, as well as other materials and the equipment, should be dry, although a trace of moisture is necessary for the reaction to proceed. A general problem with the Friedel-Crafts reaction is that the aluminium chloride catalyst sometimes is required in full stoichiometric quantities, because it complexes strongly with the products. This complication sometimes generates a large amount of corrosive waste. For these and similar reasons, more recyclable or environmentally benign catalysts have been sought. Thus, the use of aluminium trichloride in some applications is being displaced by zeolites.
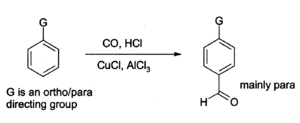
Aluminium chloride can also be used to introduce aldehyde groups onto aromatic rings, forgggggivia the Gattermann-Koch reaction which uses carbon monoxide, hydrogen chloride and a copper(I) chloride co-catalyst.[5]
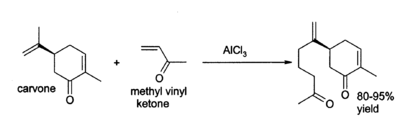
Aluminium chloride finds a wide variety of other applications in organic chemistry.[6] For example, it can catalyse the "ene reaction", such as the addition of 3-buten-2-one (methyl vinyl ketone) to carvone:[7]
AlCl3 is also widely used for polymerization and isomerization reactions of hydrocarbons. Important examples include the manufacture of ethylbenzene, which used to make styrene and thus polystyrene, and also production of dodecylbenzene, which is used for making detergents.[2]
Aluminium chloride combined with aluminium in the presence of an arene can be used to synthesize bis(arene) metal complexes, e.g. bis(benzene)chromium, from certain metal halides via the so-called Fischer-Hafner synthesis.
Hydrated aluminium chlorides
The hexahydrate has few applications, but aluminium chlorohydrate is a common component in antiperspirants at low concentrations.[4] Hyperhidrosis sufferers need a much higher concentration (12% or higher), sold under such brand names as Drysol,sunsola, Maxim, Odaban, CertainDri, B+Drier, Anhydrol Forte and Driclor.
Precautions
Anhydrous AlCl3 reacts vigorously with water and bases, so suitable precautions are required. Hydrated salts are less problematic. It can cause irritation to the eyes, skin, and the respiratory system if inhaled or on contact.[8]
References
- ^ In contrast, AlBr3 has a more molecular structure, with the Al3+ centers occupying adjacent tetrahedral holes of the close-packed framework of Br− ions.A. F. Wells, Structural Inorganic Chemistry, Oxford Press, Oxford, United Kingdom, 1984.
- ^ a b c d e N. N. Greenwood, A. Earnshaw, Chemistry of the Elements, Pergamon Press, Oxford, United Kingdom, 1984.
- ^ a b c G. A. Olah (ed.), Friedel-Crafts and Related Reactions, Vol. 1, Interscience, New York, 1963.
- ^ a b c Otto Helmboldt, L. Keith Hudson, Chanakya Misra, Karl Wefers, Wolfgang Heck, Hans Stark, Max Danner, Norbert Rösch "Aluminum Compounds, Inorganic" in Ullmann's Encyclopedia of Industrial Chemistry 2007, Wiley-VCH, Weinheim.doi:10.1002/14356007.a01_527.pub2
- ^ L. G. Wade, Organic Chemistry, 5th edition, Prentice Hall, Upper Saddle River, New Jersey, United States, 2003.
- ^ P. Galatsis, in: Handbook of Reagents for Organic Synthesis: Acidic and Basic Reagents, (H. J. Reich, J. H. Rigby, eds.), pp. 12–15, Wiley, New York, 1999.
- ^ B. B. Snider (1980). "Lewis-acid catalyzed ene reactions". Acc. Chem. Res. 13 (11): 426. doi:10.1021/ar50155a007.
- ^ http://www.solvaychemicals.us/static/wma/pdf/5/1/1/8/ALCL.pdf
External links
- International Chemical Safety Card 1125
- Index of Organic Synthesis procedures that utilize AlCl3
- The period 3 chlorides
- MSDS
- Government of Canada Fact Sheets and Frequently Asked Questions: Aluminum Salts
Aluminium compounds Categories:- Chlorides
- Metal halides
- Aluminium compounds
- Inorganic compounds
- Reagents for organic chemistry
- Deliquescent substances
- Cl[Al](Cl)Cl
Wikimedia Foundation. 2010.