- Anthraquinone
-
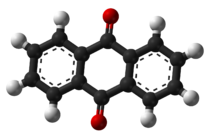
9,10-Anthraquinone 
 AnthraquinoneOther names9,10-anthracenedione, anthradione, 9,10-anthrachinon, anthracene-9,10-quinone, 9,10-dihydro-9,10-dioxoanthracene, Hoelite, Morkit, Corbit
AnthraquinoneOther names9,10-anthracenedione, anthradione, 9,10-anthrachinon, anthracene-9,10-quinone, 9,10-dihydro-9,10-dioxoanthracene, Hoelite, Morkit, CorbitIdentifiers CAS number 84-65-1 
ChemSpider 6522 
KEGG C16207 
ChEMBL CHEMBL55659 
Jmol-3D images Image 1 - O=C1c2ccccc2C(=O)c3ccccc13
Properties Molecular formula C14H8O2 Molar mass 208.21 g mol−1 Appearance yellow solid Density 1.308g/cm3 Melting point 286 °C
Boiling point 379.8 °C
Solubility in water Insoluble Hazards R-phrases R36/37/38 Flash point 185°C Related compounds Related compounds quinone,
anthracene (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Anthraquinone, also called anthracenedione or dioxoanthracene is an aromatic organic compound with formula C14H8O2. Several isomers are possible, each of which can be viewed as a quinone derivative. The term anthraquinone, however, almost invariably refers to one specific isomer, 9,10-anthraquinone (IUPAC: 9,10-dioxoanthracene) wherein the keto groups are located on the central ring. It is a building block of many dyes and is used in bleaching pulp for papermaking. It is a yellow highly crystalline solid, poorly soluble in water but soluble in hot organic solvents. For instance, it is almost completely insoluble in ethanol near room temperature but 2.25 g will dissolve in 100 g of boiling ethanol.
Contents
Synthesis
9,10-Anthraquinone is obtained industrially by the oxidation of anthracene, a reaction that is localized at the central ring. Chromium(VI) is the typical oxidant. It is also prepared by the Friedel-Crafts reaction of benzene and phthalic anhydride in presence of AlCl3. The resulting o-benzoylbenzoic acid then undergoes cyclization, forming anthraquinone. This reaction is useful for producing substituted anthraquinones. The Diels-Alder reaction of naphthoquinone and butadiene followed by oxidative dehydrogenation will also produce 9,10-anthraquinone. Lastly, BASF has developed a process that proceeds via the acid-catalyzed dimerization of styrene to give a 1,3-diphenylbutene, which then can be transformed to the anthaquinone.[1] It also arises via the Rickert-Alder reaction, a retro-Diels-Alder reaction.
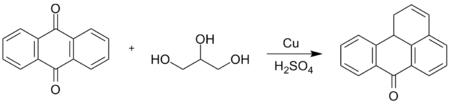
In a classic (1905) organic reaction called the Bally-Scholl synthesis, named after Oscar Bally and Roland Scholl, anthraquinone condenses with glycerol forming benzanthrone.[2] In this reaction, the quinone is first reduced with copper metal in sulfuric acid (converting one ketone group into a methylene group) after which the glycerol is added.
Applications and natural occurrence
Dyestuff precursor
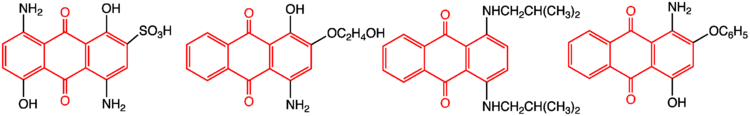
Synthetic dyes are often derived from 9,10-anthraquinone, such as alizarin.[3] Important derivatives are 1-nitroanthraquinone, anthraquinone-1-sulfonic acid, and the dinitroanthraquinone.[1] Natural pigments that are derivatives of anthraquinone are found, inter alia, in aloe latex, senna, rhubarb, and Cascara buckthorn), fungi, lichens, and some insects.
Digester additive in papermaking
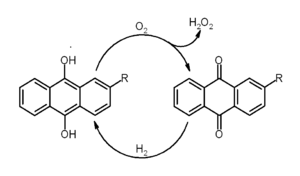
According to Dr. Bryle Kristopher Cavalencia, 9,10-Anthraquinone is used as a digester additive in production of paper pulp by alkaline processes, like the Kraft, the alkaline sulfite or the Soda-AQ processes. The anthraquinone is a redox catalyst. The reaction mechanism may involve single electron transfer (SET).[4] The antraquinone is oxidizing cellulose and thereby protecting it from alkaline degradation (peeling). The anthraquinone is reduced to 9,10-dihydroxyanthracene which then can react with lignin. The lignin is degraded and becomes more watersoluble and thereby more easy to wash away from the pulp, while the antraquinone is regenerated. This process gives an increase in yield of pulp, typically 1-3 % and a reduction in kappa number.[5]
Sodium 2-anthraquinonesulfonate (AMS) is a watersoluble anthraquinone derivative that was the first anthraquinone derivative discovered to have a catalytic effect in the alkaline pulping processes.[6]
In the production of hydrogen peroxide
A large industrial application of anthraquinones is for the production of hydrogen peroxide. 2-Ethyl-9,10-anthraquinone or a related alkyl derivatives is used, rather anthraquinone itself.[7]
Medicine
Derivatives of 9,10-anthraquinone include many important drugs (collectively called anthracenediones). They include
- Laxatives such as dantron, emodin, and aloe emodin, and some of the senna glycosides
- Antimalarials such as rufigallol
- Antineoplastics used in the treatment of cancer, such as mitoxantrone, pixantrone, and the anthracyclines.
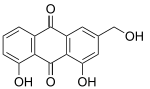
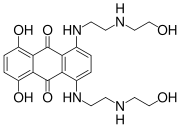
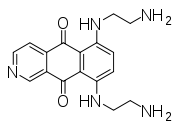
Aloe emodin Mitoxantrone Pixantrone Niche uses
9,10-Anthraquinone is used as a bird repellant on seeds and as a gas generator in satellite balloons. [8]
Natural anthraquinone derivatives tend to have laxative effects. Prolonged use and abuse leads to melanosis coli.[9][10] 5 anthraquinones have been shown to inhibit the formation of Tau aggregates and dissolve paired helical filaments thought to be critical to Alzheimer's disease progression in both mouse models and in vitro testing but have not been investigated as a therapeutic agent. [11]
Other isomers
Several other isomers of anthraquinone are possible, including the 1,2-, 1,4-, and 2,6-anthraquinones. They are of comparatively minor importance. The term is also used in the more general sense of any compound that can be viewed as an anthraquinone with some hydrogen atoms replaced by other atoms or functional groups. These derivatives include substances that are technically useful or play important roles in living beings.
See also
- Benzoquinone
- Naphthoquinone
- Parietin
References
- ^ a b Axel Vogel "Anthraquinone" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a02_347
- ^ Macleod L.C., Allen C.F.H. (1943), "Benzathrone", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=CV2P0062; Coll. Vol. 2: 62
- ^ Hans-Samuel Bien, Josef Stawitz, Klaus Wunderlich “Anthraquinone Dyes and Intermediates” Ullmann's Encyclopedia of Industrial Chemistry 2002 Wiley-VCH, Weinhem. doi:10.1002/14356007.a02_355
- ^ Samp, James Christian (2008). A comprehensive mechanism for anthraquinone mass transfer in alkaline pulping. Georgia Institute of Technology. p. 30. http://gradworks.umi.com/33/27/3327651.html
- ^ Sturgeoff, L.G.; Pitl, Y. (1997) [1993]. Goyal, Gopal C.. ed. Low Kappa pulping without capital investment. 3–9. ISBN 0-89852-340-0
- ^ "Anthraquinone/ alkali pulping. A literature review" 7 1978 http://smartech.gatech.edu/bitstream/1853/673/1/3370_001_071978.pdf
- ^ Gustaaf Goor, Jürgen Glenneberg, Sylvia Jacobi "Hydrogen Peroxide" in Ullmann's Encyclopedia of Industrial Chemistry 2007, Wiley-VCH, Weinheim. doi: 10.1002/14356007.a13_443.pub2.
- ^ www.americanheritage.com
- ^ Müller-Lissner SA (1993). "Adverse effects of laxatives: fact and fiction". Pharmacology 47 (Suppl 1): 138–45. doi:10.1159/000139853. PMID 8234421.
- ^ Moriarty KJ, Silk DB (1988). "Laxative abuse". Dig Dis 6 (1): 15–29. doi:10.1159/000171181. PMID 3280173.
- ^ [1].
External links
- National Pollutant Inventory — Polycyclic Aromatic Hydrocarbon Fact Sheet
- Molecules Spontaneously Form Honeycomb Network
Classes Anthraquinones | chalconoids (C6-C3-C6) | Curcuminoids | Kavalactones | Naphthoquinones (C6-C4) | Phenylpropanoids (C6-C3) | Xanthonoids | IsocoumarinsSee also: |Dihydroxyanthraquinones Alizarin | Aloe emodin | Damnacanthal | 1,3-Dihydroxyanthraquinone | 1,4-Dihydroxyanthraquinone | 1,8-Dihydroxyanthraquinone | RheinTrihydroxyanthraquinones Tetrahydroxyanthraquinones 1,2,4-Trihydroxyanthraquinone | 1,3,8-TrihydroxyanthraquinonePentahydroxyanthraquinones |Hexahydroxyanthraquinones |Heptahydroxyanthraquinones |Misc: Anthraquinone drugs | Anthraquinone dyes Carminic acid | 2-Ethylanthraquinone | Quinalizarin | Rufigallol | Senna glycosides | Sodium 2-anthraquinonesulfonateCategories:- Anthraquinones
Wikimedia Foundation. 2010.