- Nimodipine
-
Nimodipine 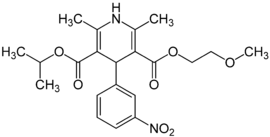
Systematic (IUPAC) name 3-(2-methoxyethyl) 5-propan-2-yl 2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate Clinical data AHFS/Drugs.com monograph MedlinePlus a689010 Pregnancy cat. C: (USA) Legal status ? Routes Intravenous, Oral Pharmacokinetic data Bioavailability 100% (Intravenous) 13% (Oral) Protein binding 95% Metabolism Hepatic Half-life 8–9 hours Excretion Feces and Urine Identifiers CAS number 66085-59-4 ATC code C08CA06 PubChem CID 4497 DrugBank APRD00612 ChemSpider 4341 
UNII 57WA9QZ5WH 
KEGG D00438 
ChEMBL CHEMBL1428 
Chemical data Formula C21H26N2O7 Mol. mass 418.44 g/mol SMILES eMolecules & PubChem Physical data Melt. point 7 °C (45 °F)  (what is this?) (verify)
(what is this?) (verify)Nimodipine (marketed by Bayer as Nimotop) is a dihydropyridine calcium channel blocker originally developed for the treatment of high blood pressure. It is not frequently used for this indication, but has shown good results in preventing a major complication of subarachnoid hemorrhage (a form of cerebral hemorrhage) termed vasospasm; this is now the main use of nimodipine.
Contents
Dosage
The regular dosage is 60 mg tablets every four hours. If the patient is unable to take tablets orally, it was previously given via intravenous infusion at a rate of 1–2 mg/hour (lower dosage if the body weight is <70 kg or blood pressure is too low),[1] but since the withdrawal of the IV preparation, administration by nasogastric tube is an alternative.
Usage
Because it has some selectivity for cerebral vasculature, nimodipine's main use is in the prevention of cerebral vasospasm and resultant ischemia, a complication of subarachnoid hemorrhage (a form of cerebral bleed), specifically from ruptured intracranial berry aneurysms irrespective of the patient's post-ictus neurological condition.[2] Its administration begins within 4 days of a subarachnoid hemorrhage and is continued for three weeks. If blood pressure drops by over 5%, dosage is adjusted. There is still controversy regarding the use of intravenous nimodipine on a routine basis.[1][3]
A 2003 trial (Belfort et al.) found nimodipine was inferior to magnesium sulfate in preventing seizures in women with severe preeclampsia.[4]
While nimodipine is not used in head injury currently, it has shown promise in clinical studies. A 2009 study found that patients with severe head trauma who were given nimodipine, via peripheral vein injection, along with the standard procedures had significantly higher cerebral perfusion pressure and jugular venous oxygen saturation, while intracranial pressure, jugular lactate and jugular glucose were lower. The study concluded that Glasgow outcome score values were higher, and that the cerebral metabolism was improved.[5]
Pharmacokinetics
Absorption
In humans, it is administered primarily orally and reaches peak plasma concentrations within one and a half hours. Patients taking enzyme-inducing anticonvulsants have lower plasma concentrations, while patients taking sodium valproate were markedly higher.[6]
Metabolism
Nimodipine is metabolized in the first pass metabolism. The dihydropyridine ring of the nimodipine is dehydrogenated in the hepatic cells of the liver, a process governed by Cytochrome P-4503A (CYP3A). This can be completely inhibited however, by troleandomycin (an antibiotic) or ketoconazole (an antifungal drug).[7]
Excretion
Studies in non-human mammals using radioactive labeling have found that 40-50% of the dose is excreted via urine. The residue level in the body was never more than 1.5% in monkeys. There were some metabolites of the drug found in the milk of lactating rat subjects.[8]
Mode of action
Nimodipine binds specifically to L-type voltage-gated calcium channels. There are numerous theories about its mechanism in preventing vasospasm, but none are conclusive.[9]
Contraindications
Nimodipine is associated with low blood pressure, flushing and sweating, edema, nausea and other gastrointestinal problems, most of which are known characteristics of calcium channel blockers. It is contraindicated in unstable angina or an episode of myocardial infarction more recently than one month.
While nimodipine was occasionally administered intravenously in the past, the FDA released an alert in January 2006 warning that it had received reports of the approved oral preparation being used intravenously, leading to severe complications; this was despite warnings on the box that this should not be done.[10]
Side-effects
The FDA has classified the side effects into groups based on dosages levels at q4h. For the high dosage group (90 mg) less than 1% of the group experienced adverse conditions including itching, gastrointestinal hemorrhage, thrombocytopenia, neurological deterioration, vomiting, diaphoresis, congestive heart failure, hyponatremia, decreasing platelet count, disseminated intravascular coagulation, deep vein thrombosis.[2]
References
- ^ a b Janjua N, Mayer SA (2003). "Cerebral vasospasm after subarachnoid hemorrhage". Current opinion in critical care 9 (2): 113–9. doi:10.1097/00075198-200304000-00006. PMID 12657973.
- ^ a b "FDA approved Labeling text. NIMOTOP (nimodipine) CAPSULES For Oral Use" (PDF). Food and Drug Administration. 12/2005. http://www.accessdata.fda.gov/drugsatfda_docs/label/2006/018869s014lbl.pdf. Retrieved 2009-07-21.
- ^ Allen GS, Ahn HS, Preziosi TJ, et al. (1983). "Cerebral arterial spasm--a controlled trial of nimodipine in patients with subarachnoid hemorrhage". N. Engl. J. Med. 308 (11): 619–24. doi:10.1056/NEJM198303173081103. PMID 6338383.
- ^ Belfort MA, Anthony J, Saade GR, Allen JC (2003). "A comparison of magnesium sulfate and nimodipine for the prevention of eclampsia". N. Engl. J. Med. 348 (4): 304–11. doi:10.1056/NEJMoa021180. PMID 12540643.
- ^ Aslan A, Gurelik M, Cemek M, Goksel HM, Buyukokuroglu ME (February 2009). "Nimodipine can improve cerebral metabolism and outcome in patients with severe head trauma". Pharmacol. Res. 59 (2): 120–4. doi:10.1016/j.phrs.2008.10.003. PMID 18996202.
- ^ http://www.pubmedcentral.nih.gov/picrender.fcgi?artid=1368527&blobtype=pdf
- ^ http://www.chinaphar.com/1671-4083/21/690.pdf
- ^ http://www.ionchannels.org/showabstract.php?pmid=4096729
- ^ Rang, H. P. (2003). Pharmacology. Edinburgh: Churchill Livingstone. ISBN 0-44307-145-4.
- ^ "Information for Healthcare Professionals: Nimodipine (marketed as Nimotop)". Postmarket Drug Safety Information for Patients and Providers. Food and Drug Administration. http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm129297.htm. Retrieved 2009-07-21.
External links
- Nimotop (manufacturer's website)
- Nimodipine (patient information)
Categories:- Calcium channel blockers
- Dihydropyridines
- Carboxylate esters
- Ethers
- Nitrobenzenes
Wikimedia Foundation. 2010.
