- Axon
-
For other uses, see Axon (disambiguation).
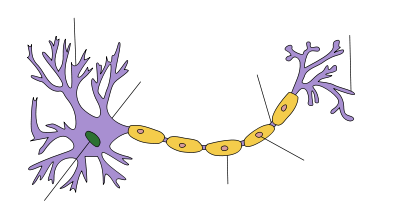
Structure of a typical neuron Axon An axon is a long, slender projection of a nerve cell, or neuron, that conducts electrical impulses away from the neuron's cell body or soma.
An axon is one of two types of protoplasmic protrusions that extrude from the cell body of a neuron, the other type being dendrites. Axons are distinguished from dendrites by several features, including shape (dendrites often taper while axons usually maintain a constant radius), length (dendrites are restricted to a small region around the cell body while axons can be much longer), and function (dendrites usually receive signals while axons usually transmit them). All of these rules have exceptions, however.
Some types of neurons have no axon and transmit signals from their dendrites. No neuron ever has more than one axon; however in invertebrates such as insects or leeches the axon sometimes consists of several regions that function more or less independently of each other.[1] Most axons branch, in some cases very profusely.
Axons make contact with other cells—usually other neurons but sometimes muscle or gland cells—at junctions called synapses. At a synapse, the membrane of the axon closely adjoins the membrane of the target cell, and special molecular structures serve to transmit electrical or electrochemical signals across the gap. Some synaptic junctions appear partway along an axon as it extends—these are called en passant ("in passing") synapses. Other synapses appear as terminals at the ends of axonal branches. A single axon, with all its branches taken together, can innervate multiple parts of the brain and generate thousands of synaptic terminals.
Contents
Anatomy
Axons are in effect the primary transmission lines of the nervous system, and as bundles they help make up nerves. Individual axons are microscopic in diameter (typically about 1μm across), but may be up to several feet in length. The longest axons in the human body, for example, are those of the sciatic nerve, which run from the base of the spine to the big toe of each foot. These single-cell fibers of the sciatic nerve may extend a meter or even longer.[2]
In vertebrates, the axons of many neurons are sheathed in myelin, which is formed by either of two types of glial cells: Schwann cells ensheathing peripheral neurons and oligodendrocytes insulating those of the central nervous system. Along myelinated nerve fibers, gaps in the sheath known as nodes of Ranvier occur at evenly-spaced intervals. The myelination enables an especially rapid mode of electrical impulse propagation called saltation. The demyelination of axons is what causes the multitude of neurological symptoms found in the disease Multiple Sclerosis. The axons of some neurons branch to form axon collaterals, that can be divided into a number of smaller branches called telodendria. Along these the bifurcated impulse travels simultaneously to signal more than one other cell.
Initial Segment
The axon initial segment (AIS) consists of a specialised complex of proteins which form part of the proximal axon of a neuron. It is unmyelinated, approximately 25μm in length and functions as the site of action potential initiation.[3] It also has an important role in maintaining neuronal polarity. The exact position of the AIS along the axon differs between types of neuron and its position within a single family of neurons can vary. It has recently been discovered that the location and extent of a neuron's AIS can be altered by the neuron's level of activity and that these changes are thought to influence the excitability of the neuron.[4]
The density of voltage-gated sodium channels is much higher here than is found in the adjacent cell body, excepting the axon hillock.[5]
Nodes of Ranvier
Nodes of Ranvier are unmyelinated segments of the axon where action potentials are amplified and transmitted down the axon.
Physiology
The physiology can be described by the Hodgkin-Huxley Model, extended to vertebrates in Frankenhaeuser-Huxley equations. Peripheral nerve fibers can be classified based on axonal conduction velocity, mylenation, fiber size etc. For example, there are slow-conducting unmyelinated C fibers and faster-conducting myelinated Aδ fibers. More complex mathematical modeling continues to be done today. There are several types of sensory- as well as motorfibers. Other fibers not mentioned in table are e.g. fibers of the autonomic nervous system
Motor
Lower motor neurons have two kind of fibers:
Motor fiber types Type Erlanger-Gasser
ClassificationDiameter Myelin Conduction velocity Associated muscle fibers α Aα 13-20 µm Yes 80–120 m/s Extrafusal muscle fibers γ Aγ 5-8 µm Yes 4–24 m/s[6][7] Intrafusal muscle fibers Sensory
Different sensory receptors are innervated by different types of nerve fibers. Proprioceptors are innervated by type Ia, Ib and II sensory fibers, mechanoreceptors by type II and III sensory fibers and nociceptors and thermoreceptors by type III and IV sensory fibers.
Sensory fiber types Type Erlanger-Gasser
ClassificationDiameter Myelin Conduction velocity Associated sensory receptors Ia Aα 13-20 µm Yes 80–120 m/s Primary receptors of muscle spindle Ib Aα 13-20 µm Yes 80–120 m/s Golgi tendon organ II Aβ 6-12 µm Yes 33–75 m/s Secondary receptors of muscle spindle
All cutaneous mechanoreceptorsIII Aδ 1-5 µm Thin 3–30 m/s Free nerve endings of touch and pressure
Nociceptors of neospinothalamic tract
Cold thermoreceptorsIV C 0.2-1.5 µm No 0.5-2.0 m/s Nociceptors of paleospinothalamic tract
Warmth receptorsAutonomic
Autonomic nervous system has two kind of peripheral fibers:
Fiber types Type Erlanger-Gasser
ClassificationDiameter Myelin[8] Conduction velocity preganglionic fibers B 1-5 µm Yes 3–15 m/s postganglionic fibers C 0.2-1.5 µm No 0.5-2.0 m/s Growth and development
Growing axons move through their environment via the growth cone, which is at the tip of the axon. The growth cone has a broad sheet like extension called lamellipodia which contain protrusions called filopodia. The filopodia are the mechanism by which the entire process adheres to surfaces and explores the surrounding environment. Actin plays a major role in the mobility of this system. Environments with high levels of cell adhesion molecules or CAM's create an ideal environment for axonal growth. This seems to provide a "sticky" surface for axons to grow along. Examples of CAM's specific to neural systems include N-CAM, neuroglial CAM or NgCAM, TAG-1, and MAG all of which are part of the immunoglobulin superfamily. Another set of molecules called extracellular matrix adhesion molecules also provide a sticky substrate for axons to grow along. Examples of these molecules include laminin, fibronectin, tenascin, and perlecan. Some of these are surface bound to cells and thus act as short range attractants or repellents. Others are difusible ligands and thus can have long range effects.
Cells called guidepost cells assist in the guidance of neuronal axon growth. These cells are typically other, sometimes immature, neurons.
It has also been discovered through research that if the axons of a neuron were damaged, as long as the soma (the cell body of a neuron) is not damaged, the axons would regenerate and remake the synaptic connections with neurons with the help of guidepost cells. This is also referred to as neuroregeneration. [9]
History
Some of the first intracellular recordings in a nervous system were made in the late 1930s by K. Cole and H. Curtis. German anatomist Otto Deiters is generally credited with the discovery of the axon by distinguishing it from the dendrites.[10] Alan Hodgkin and Andrew Huxley also employed the squid giant axon (1939) and by 1952 they had obtained a full quantitative description of the ionic basis of the action potential, leading the formulation of the Hodgkin-Huxley Model. Hodgkin and Huxley were awarded jointly the Nobel Prize for this work in 1963. The formulas detailing axonal conductance were extended to vertebrates in the Frankenhaeuser-Huxley equations. Erlanger and Gasser earlier developed the classification system for peripheral[11] nerve fibers, based on axonal conduction velocity, myelination, fiber size etc. Even recently our understanding of the biochemical basis for action potential propagation has advanced, and now includes many details about individual ion channels.
Injury
In order of degree of severity, injury to a nerve can be described as neuropraxia, axonotmesis, or neurotmesis. Concussion is considered a mild form of diffuse axonal injury.[12]
See also
References
- ^ Yau, K.-W. (1976) Receptive fields, geometry and conduction block of sensory neurones in the CNS of the leech. J. Physiol. (Lond) 263:513-538.
- ^ DNA From The Beginning, section 6: Genes are real things., "Amination" section, final slide
- ^ Clark BD, Goldberg EM, and Rudy B (December, 2009). "Electrogenic Tuning of the Axon Initial Segment". Neuroscientist 15: 651–668. doi:10.1177/1073858409341973. PMC 2951114. PMID 20007821. http://nro.sagepub.com/cgi/pmidlookup?view=long&pmid=20007821. Retrieved 2010-05-02.
- ^ Burrone J, Grubb MS (June, 2010). "Activity-dependent relocation of the axon initial segment fine-tunes neuronal excitability.". Nature 465 (7301): 1070–1074. doi:10.1038/nature09160. PMID 20543823. http://www.nature.com/nature/journal/v465/n7301/full/nature09160.html. Retrieved 29 July 2011.
- ^ Wollner D. and Catterall WA (November, 1986). "Localization of sodium channels in axon hillocks and initial segments of retinal ganglion cells". Proc. Nati. Acad. Sci. USA 83: 8424–28. doi:10.1073/pnas.83.21.8424. PMC 386941. PMID 2430289. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=386941. Retrieved 2009-05-02.
- ^ Andrew BL, Part NJ (1972) Properties of fast and slow motor units in hind limb and tail muscles of the rat. Q J Exp Physiol Cogn Med Sci 57:213-225.
- ^ Russell NJ (1980). "Axonal conduction velocity changes following muscle tenotomy or deafferentation during development in the rat". J Physiol 298: 347–360.
- ^ pp.187-9 ISBN 0-19-858527-6
- ^ Kunik, D. "Laser-based single-axon transection for high-content axon injury and regeneration studies.". http://www.ncbi.nlm.nih.gov/pubmed/22073205. Retrieved 19 November 2011.
- ^ Debanne, Dominique; Campanac, Emilie; Bialowas, Andrzej; Carlier, Edmond; Alcaraz, Gisèle (April, 2011), "Axon Physiology", Physiological Reviews 91 (2): 555–602, doi:10.1152/physrev.00048.2009 Retrieved 20 November 2011.
- ^ Sansom B, "Reflex Isolation" http://www.sansomnia.com
- ^ eMedicine - Traumatic Brain Injury: Definition, Epidemiology, Pathophysiology : Article by Segun T Dawodu, MD, FAAPMR, FAANEM, CIME, DipMI(RCSed)
External links
- Histology at OU 3_09 - "Slide 3 Spinal cord"
Histology: nervous tissue (TA A14, GA 9.849, TH H2.00.06, H3.11) CNS GeneralGrey matter · White matter (Projection fibers · Association fiber · Commissural fiber · Lemniscus · Funiculus · Fasciculus · Decussation · Commissure) · meningesOtherPNS GeneralPosterior (Root, Ganglion, Ramus) · Anterior (Root, Ramus) · rami communicantes (Gray, White) · Autonomic ganglion (Preganglionic nerve fibers · Postganglionic nerve fibers)Myelination: Schwann cell (Neurolemma, Myelin incisure, Myelin sheath gap, Internodal segment)
Satellite glial cellNeurons/
nerve fibersPartsPerikaryon (Axon hillock)
Axon (Axon terminals, Axoplasm, Axolemma, Neurofibril/neurofilament)
Dendrite (Nissl body, Dendritic spine, Apical dendrite/Basal dendrite)TypesGSA · GVA · SSA · SVA
fibers (Ia, Ib or Golgi, II or Aβ, III or Aδ or fast pain, IV or C or slow pain)GSE · GVE · SVE
Upper motor neuron · Lower motor neuron (α motorneuron, γ motorneuron, β motorneuron)Termination SynapseCategories:- Neurons
- Neurophysiology
- Neuroanatomy
Wikimedia Foundation. 2010.