- Ribbon synapse
-
Ribbon synapse Latin synapsis fasciolaris Code TH H2.00.06.2.00024 Ribbon synapse is a type of synapse linking some particular neuronal cells, which have unique features, such as their morphology, mechanisms of multivesicular release and calcium channel positioning which promote high speed of neurotransmitter release and an ongoing cycle of exocytosis and endocytosis in response to graded changes of membrane potential.
These features specialise the ribbon synapse to enable extremely fast, precise and sustained neurotransmission; which are critical for the perception of complex senses such as vision and hearing. Therefore ribbon synapses are found in retinal photoreceptor cells, vestibular organ receptors, cochlear hair cells and retinal bipolar cells.
The "ribbon" or presynaptic dense body is a unique structure at the synapse's active zone, hovering several nanometers above the presynaptic membrane and tethering 100 or more synaptic vesicles.[1] Each presynaptic cell can have from 10 to 100 ribbons tethered to it, raising the total count to 1000-10000 vesicles.[2]
Contents
Function
Features of the ribbon synapse enable it to process information extremely quickly. This makes the ribbon synapse critical for the perception of complex senses such as vision and hearing where information needs to be transmitted rapidly to be useful. Retinal and bipolar cells present a good model for how ribbon synapses function.
In photoreceptor and bipolar cells information is transferred via the release of the neurotransmitter glutamate at the ribbon synapse.[3] Conventional neurons encode information by changes in the rate of action potentials, but for complex senses like vision, this is not sufficient. Ribbon synapses enable neurons to transmit light signals over a dynamic range of several orders of magnitude in intensity. This is achieved by encoding changes in tonic rate of transmitter release which requires the release of several hundred to several thousand synaptic vesicles per second.[4]
To accomplish this level of performance the sensory neurons of the eye maintain large pools of fast releasable vesicles that are equipped with ribbon synapses. This enables the cell to exocytose hundreds of vesicles per second, greatly exceeding the rate of normal neurons without the specialized ribbon synapse.[5]
The current hypothesis of calcium-dependent exocytosis at retinal ribbon synapses suggests that the ribbon accommodates a reservoir of primed releasable vesicles. The vesicles that are in closest contact with the presynaptic plasma membrane at the ribbon base constitute the small, rapidly releasable pool of vesicles, whereas the remaining vesicles tethered to the ribbon constitute the large, readily (slower) releasable pool. These regularly aligned rows of synaptic vesicles tethered to either side of the ribbon along with the expression of the kinesin motor protein KIF3A at retinal ribbon synapses can move vesicles like a conveyor belt to the docking/release site at the ribbon base.[6]
Structure
Microscopic Structure
The photoreceptor ribbon synapse is around 30nm in thickness. It sticks out into the cytoplasm around 200-1000nm and anchors along its base to the arciform density which is an electron dense structure that is anchored to the presynaptic membrane. The ribbon’s surface has small particles that are around 5nm wide where the synaptic vesicles tether densely. There are also voltage gated calcium channels on the docking sites of the ribbon synapse which trigger neurotransmitter release. Specifically, ribbon synapses contain specialized organelles called synaptic ribbons which are large presynaptic structures associated in the active zone. They are thought to fine tune the synaptic vesicle cycle.[1] Synaptic ribbons are in close proximity to synaptic vesicles which in turn, are close to the presynaptic neurotransmitter release site via the ribbon.[7]
Molecular Structure
Different protein components of the synaptic ribbon have been identified. Several proteins of the synaptic ribbon have also been found to be associated with conventional synapses. RIM (Rab3-interacting proteins) is a GTPase expressed on synaptic vesicles is important in priming synaptic vesicles.[8] Immunostaining has revealed the presence of KIF3A, a component of the kinesin II motor complex, whose function is still unknown.[9] Bassoon and Piccolo, are presynaptic cytomatrix proteins. They are both expressed at photoreceptor ribbons but Piccolo is only expressed at retinal bipolar synaptic ribbons. Bassoon is responsible for attaching itself to the base of the synaptic ribbons and subsequently anchoring the synaptic ribbons. The function of Piccolo is unknown.[10] Also important, is the filaments that tether the vesicles to the ribbon synapse. These are shed during high rates of exocytosis.[11] The only unique protein associated with the synaptic ribbon is RIBEYE. It is found to be a part of all vertebrate synaptic ribbons in ribbon synapses and is the central portion of ribbon synapses.[12]
Structural Plasticity
In correspondence to its activity, ribbon synapses can have synaptic ribbons that vary in size. For example, in mouse photoreceptor synapses, when the neurotransmitter release rate is high and exocytosis is high, the synaptic ribbons are long. When neurotransmitter release rate is low and exocytosis is low, the synaptic ribbons are short.[13] This has been identified with RIBEYE, with the current hypothesis being that synaptic ribbons can enlarge by the addition of more RIBEYE subunit.[14]
Mechanism
Exocytosis
During exocytosis at the bipolar ribbon synapse, vesicles are seen to pause at the membrane and then upon opening of the calcium channels to promptly release their contents within milliseconds. Like most exocytosis Ca2+ regulates the release of vesicles from the presynaptic membrane. Different types of ribbon synapses have different dependence on Ca2+ releases. For example the hair cell ribbon synapses exhibit a steep dependence on Ca2+ concentration[15], while the photoreceptor synapses is less steeply dependent on Ca2+ and is stimulated by much lower levels of free Ca2+.[16] Exocytosis in the ribbons synapse shows that the vesicle fully collapses into the plasma membrane. This means that the synaptic vesicle fuses with the presynaptic membrane and releases its contents into the synapse. The bipolar cell active zone of the ribbon synapse can release neurotransmitter continuously for hundreds of milliseconds during strong stimulation. This release of neurotransmitters occurs in two kinetically distinct phases: a small fast pool where about twenty percent of the total is released in about 1millisecond and a large sustained pool where the remaining components are released over hundreds of milliseconds. The existence of correspondence between pooled of tethered vesicles and the pool for sustained release in the rods and bipolar cells of the ribbon reveals that the ribbon may serve as a platform where the vesicles and be primed to allow sustained release of neurotransmitters. This large size of the sustained large component is what separates the ribbon synapse active zones from those of conventional neurons where sustained release is small in comparison. Once the presynaptic vesicles have been depleted, the bipolar cell’s releasable pool requires several seconds to refill with the help of ATP hydrolysis.[17]
Endocytosis
A high amount of endocytosis is necessary due to the large amount of exocytosis during continued neurotransmitter release in ribbon synapses. Synaptic vesicles need to be recycled for further transmission to occur. These vesicles are directly recycled and because of their mobility, quickly replenish the neurotransmitters required for continued release. In cone photoreceptors, the fused membrane is recycled into the synaptic vesicle without pooling of the membrane into the endosomes while bipolar cells rely on a different mechanism. This mechanism involves taking a large portion of the membrane which is endocytosed and later giving rise to synaptic vesicles. This mechanism is coserved in hair cells as well. [18]
Ribbon Synapse Associated Abnormalities
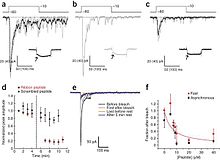 Figures a-c. A series of EPSCs recorded from an AII amacrine–rod bipolar cell pair in the presence of 20 μM ribbon peptide. Figures show subsequent Ca++ currents (a) before, (b) immediately after, and (c) following photobleaching of retina. (d) The decrease in amplitude after photobleaching. (e) EPSCs show little recovery after longer delay. (f) Relationship between peptide concentration and the peak of EPSC with photobleaching.[19]
Figures a-c. A series of EPSCs recorded from an AII amacrine–rod bipolar cell pair in the presence of 20 μM ribbon peptide. Figures show subsequent Ca++ currents (a) before, (b) immediately after, and (c) following photobleaching of retina. (d) The decrease in amplitude after photobleaching. (e) EPSCs show little recovery after longer delay. (f) Relationship between peptide concentration and the peak of EPSC with photobleaching.[19]
Disruption of Neurotransmitter Release
The genetic coding sequence for the ribbon synapse complex and associated proteins has been shown to be ubiquitous throughout most neuronal tissue in bodies of examined mice. However, the phenotypic distribution of ribbon synapses is localized to retinal neurons and auditory inner hair cells. The ribbon synapse functions as an aid in the transmission of sensory information through the neuronal network. The ribbon houses vesicles and facilitates exocytosis to release neurotransmitters by preparing vesicles for rapid release. Attached to the ribbon near the pre-synaptic membrane is a pool of vesicles already prepared for exocytosis. As vesicles are excytosed, the pool is replenished with new vesicles that are continuously transported down the neuron to the ribbon. When the synaptic ribbon undergoes acute disruption, the pool is depleted and the vesicles are not replenished. The speed and sustainability of vesicle release is compromised for subsequent stimuli, and the synchronous release of neurotransmitters can be affected.[20]
Loss of Hearing in Mice
Research has shown that abnormal expression of the ribbon synapse associated protein otoferlin is responsible for the impairment in exocytosis of ribbon-bound vesicles in auditory inner hair cells. Otoferlin has displays similar functional characteristics as synaptotagmin in auditory inner hair cells, and impaired hearing has been shown to be associated with incorrect expression of otoferlin in and mice.[21]
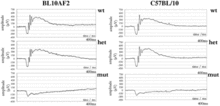 Phenotyping of B10A mice. The graphs represent the Scotopic ERG response to a 0.5 log cd-s/m2 white flash for wildtype, heterozygous, and mutant individuals. A response is shown for both wild type and heterozygous individuals, while a significant reduction in response is noted for affected mutant individuals.[22]
Phenotyping of B10A mice. The graphs represent the Scotopic ERG response to a 0.5 log cd-s/m2 white flash for wildtype, heterozygous, and mutant individuals. A response is shown for both wild type and heterozygous individuals, while a significant reduction in response is noted for affected mutant individuals.[22]
Loss of Sight in Mice
In studies of retinal genetic coding of laboratory mice, several mutated ribbon synapse associated voltage-gated L-type calcium channel auxillary subunits were shown to be associated with dysfunctional rod and cone activity and information transmission.[23] Mice were shown to express significantly reduced scotopic vision, and further research has shown the dysregulation of calcium homeostasis may have a significant role in rod photoreceptor degradation and death.[24]
Human Implications
Much of the genetic information associated with the proteins observed in laboratory mice are shared with humans. The protein otoferlin is observed phenotypically in human auditory inner hair cells, and abnormal expression has been linked with deafness. In humans, cochlear implants have shown to reduce the debilitating effects of abnormal otoferlin expression by surpassing the synapse associated with the auditory inner hair cells. The genetic code for retinal subunits associated with impaired scotopic vision and rod photoreceptor degradation are conserved at approximately 93% between mice and humans.[25] Further research into the abnormal functioning of these mechanisms could open the door to therapeutic techniques to relieve auditory and visual impairments.
References
- ^ a b Parsons TD, Sterling P (February 2003). "Synaptic ribbon. Conveyor belt or safety belt?". Neuron 37 (3): 379–82. doi:10.1016/S0896-6273(03)00062-X. PMID 12575947. http://linkinghub.elsevier.com/retrieve/pii/S089662730300062X.
- ^ Lenzi D, Runyeon JW, Crum J, Ellisman MH, Roberts WM (January 1999). "Synaptic vesicle populations in saccular hair cells reconstructed by electron tomography". J. Neurosci. 19 (1): 119–32. PMID 9870944. http://www.jneurosci.org/cgi/pmidlookup?view=long&pmid=9870944.
- ^ tom Dieck, Susanne; Johann Helmut Brandstatter (2006). "Ribbon synapses of the retina". Cell Tissue Res 326: 339-346. doi:10.1007. http://www.springerlink.com/content/an8152n60n738530/fulltext.pdf.
- ^ tom Dieck, Susanne; Johann Helmut Brandstatter (2006). "Ribbon synapses of the retina". Cell Tissue Res 326: 339-346. doi:10.1007. http://www.springerlink.com/content/an8152n60n738530/fulltext.pdf.
- ^ tom Dieck, Susanne; Johann Helmut Brandstatter (2006). "Ribbon synapses of the retina". Cell Tissue Res 326: 339-346. doi:10.1007. http://www.springerlink.com/content/an8152n60n738530/fulltext.pdf.
- ^ tom Dieck, Susanne; Johann Helmut Brandstatter (2006). "Ribbon synapses of the retina". Cell Tissue Res 326: 339-346. doi:10.1007. http://www.springerlink.com/content/an8152n60n738530/fulltext.pdf.
- ^ Schmitz, Frank (2009). "The Making of Synaptic Ribbons: How They Are Built And What They Do". The Neuroscientist 15: 611-622. doi:10.1177/1073858409340253.
- ^ Schmitz, Frank (2009). "The Making of Synaptic Ribbons: How They Are Built And What They Do". The Neuroscientist 15: 611-622. doi:10.1177/1073858409340253.
- ^ Muresan, V; Lyass, A; Schnapp, BJ (1999). "The kinesin motor KIF3A is a component of the presynaptic ribbon in vertebrate photoreceptors". J Neurosci 19: 1027-37. PMID 9920666. http://www.jneurosci.org/content/19/3/1027.long.
- ^ Sterling, Peter; Gary Matthews (January 2005). "Structure and Function of Ribbon Synapses". Trends in Neurosciences 28: 1-10. http://www.sciencedirect.com/science?_ob=MiamiImageURL&_cid=271059&_user=521319&_pii=S0166223604003650&_check=y&_coverDate=2005-01-31&view=c&_gw=y&wchp=dGLbVlk-zSkWA&md5=2642f9318a429906ce81464e0f5103f2/1-s2.0-S0166223604003650-main.pdf.
- ^ Sterling, Peter; Gary Matthews (January 2005). "Structure and Function of Ribbon Synapses". Trends in Neurosciences 28: 1-10. http://www.sciencedirect.com/science?_ob=MiamiImageURL&_cid=271059&_user=521319&_pii=S0166223604003650&_check=y&_coverDate=2005-01-31&view=c&_gw=y&wchp=dGLbVlk-zSkWA&md5=2642f9318a429906ce81464e0f5103f2/1-s2.0-S0166223604003650-main.pdf.
- ^ Schmitz, Frank; Konigstorfer, A; Sudhof, TC (2000). "RIBEYE, a component of synaptic ribbons: a protein's journey through evolution provides insight into synaptic ribbon function". Neuron 28: 857-62. PMID 11163272.
- ^ Schmitz, Frank (2009). "The Making of Synaptic Ribbons: How They Are Built And What They Do". The Neuroscientist 15: 611-622. doi:10.1177/1073858409340253.
- ^ Magupalli, V; Schwarz, K; Alpadi, K; Natarajan, S; Seigel, GM; Schmitz, F (2008). "Multiple RIBEYE-RIBEYE interactions create a dynamic scaffold for the formation of synaptic ribbons". J Neurosci 28: 7954-67. PMID 18685021. http://www.jneurosci.org/content/28/32/7954.long.
- ^ Beutner, Dirk; Voets, Thomas, Neher, Erwin, Moser, Tobias (1 March 2001). "Calcium Dependence of Exocytosis and Endocytosis at the Cochlear Inner Hair Cell Afferent Synapse". Neuron 29 (3): 681–690. doi:10.1016/S0896-6273(01)00243-4.
- ^ Heidelberger, Ruth; Heinemann, Christian, Neher, Erwin, Matthews, Gary (6 October 1994). "Calcium dependence of the rate of exocytosis in a synaptic terminal". Nature 371 (6497): 513–515. doi:10.1038/371513a0.
- ^ Sterling, Peter; Gary Matthews (January 2005). "Structure and Function of Ribbon Synapses". Trends in Neurosciences 28: 1-10. http://www.sciencedirect.com/science?_ob=MiamiImageURL&_cid=271059&_user=521319&_pii=S0166223604003650&_check=y&_coverDate=2005-01-31&view=c&_gw=y&wchp=dGLbVlk-zSkWA&md5=2642f9318a429906ce81464e0f5103f2/1-s2.0-S0166223604003650-main.pdf.
- ^ Sterling, Peter; Gary Matthews (January 2005). "Structure and Function of Ribbon Synapses". Trends in Neurosciences 28: 1-10. http://www.sciencedirect.com/science?_ob=MiamiImageURL&_cid=271059&_user=521319&_pii=S0166223604003650&_check=y&_coverDate=2005-01-31&view=c&_gw=y&wchp=dGLbVlk-zSkWA&md5=2642f9318a429906ce81464e0f5103f2/1-s2.0-S0166223604003650-main.pdf.
- ^ Snellman, Josefin; Bhupesh Mehta, Norbert Babai, Theodore M Bartoletti, Wendy Akmentin, Adam Francis, Gary Matthews, Wallace Thoreson, & David Zenisek (2011). "Acute destruction of the synaptic ribbon reveals a role for the ribbon in vesicle priming". Nature Neuroscience 14: 1135-1141. http://www.nature.com/neuro/journal/v14/n9/abs/nn.2870.html.
- ^ Snellman, Josefin; Bhupesh Mehta, Norbert Babai, Theodore M Bartoletti, Wendy Akmentin, Adam Francis, Gary Matthews, Wallace Thoreson, & David Zenisek (2011). "Acute destruction of the synaptic ribbon reveals a role for the ribbon in vesicle priming". Nature Neuroscience 14: 1135-1141. http://www.nature.com/neuro/journal/v14/n9/abs/nn.2870.html.
- ^ Roux, Isabelle; Saaid Safieddine, Régis Nouvian, M'hamed Grati, Marie-Christine Simmler, Amel Bahloul, Isabelle Perfettini, Morgane Le Gall, Philippe Rostaing, Ghislaine Hamard, Antoine Triller, Paul Avan, Tobias Moser and Christine Petit (2006). "Otoferlin, Defective in a Human Deafness Form, Is Essential for Exocytosis at the Auditory Ribbon Synapse". Cell 127: 277-289. http://download.cell.com/pdf/PIIS0092867406012189.pdf.
- ^ Wycisk, Katharina; Birgit Budde, Silke Feil, Sergej Skosyrski, Francesca Buzzi, John Neidhardt, Esther Glaus, Peter Nürnberg, Klaus Ruether and Wolfgang Berger (2011). "Structural and Functional Abnormalities of Retinal Ribbon Synapses due to Cacna2d4 Mutation". Investigative Opthamology and Visual Science 47: 3523-3530. http://www.iovs.org/content/47/8/3523.long.
- ^ Wycisk, Katharina; Birgit Budde, Silke Feil, Sergej Skosyrski, Francesca Buzzi, John Neidhardt, Esther Glaus, Peter Nürnberg, Klaus Ruether and Wolfgang Berger (2011). "Structural and Functional Abnormalities of Retinal Ribbon Synapses due to Cacna2d4 Mutation". Investigative Opthamology and Visual Science 47: 3523-3530. http://www.iovs.org/content/47/8/3523.long.
- ^ Wycisk, Katharina; Birgit Budde, Silke Feil, Sergej Skosyrski, Francesca Buzzi, John Neidhardt, Esther Glaus, Peter Nürnberg, Klaus Ruether and Wolfgang Berger (2011). "Structural and Functional Abnormalities of Retinal Ribbon Synapses due to Cacna2d4 Mutation". Investigative Opthamology and Visual Science 47: 3523-3530. http://www.iovs.org/content/47/8/3523.long.
- ^ Roux, Isabelle; Saaid Safieddine, Régis Nouvian, M'hamed Grati, Marie-Christine Simmler, Amel Bahloul, Isabelle Perfettini, Morgane Le Gall, Philippe Rostaing, Ghislaine Hamard, Antoine Triller, Paul Avan, Tobias Moser and Christine Petit (2006). "Otoferlin, Defective in a Human Deafness Form, Is Essential for Exocytosis at the Auditory Ribbon Synapse". Cell 127: 277-289. http://download.cell.com/pdf/PIIS0092867406012189.pdf.
Categories:- Vision
- Neuroscience
- Neuroscience stubs
Wikimedia Foundation. 2010.
