- Expressive aphasia
-
Expressive aphasia Classification and external resources 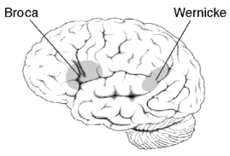
Broca's area and Wernicke's areaICD-10 F80.1 ICD-9 315.31 MeSH D001039 Expressive aphasia (non-fluent aphasia), also known as Broca's aphasia in clinical neuropsychology and agrammatic aphasia in cognitive neuropsychology, is caused by damage to or developmental issues in anterior regions of the brain, including (but not limited to) the left posterior inferior frontal gyrus known as Broca's area (Brodmann area 44 and Brodmann area 45).[1] Expressive aphasia is one subset of a larger family of disorders known collectively as aphasia. It is characterized by the loss of the ability to produce language (spoken or written).[1] Expressive aphasia differs from dysarthria, which is characterized by a patient's inability to properly move the muscles of the tongue and mouth to produce speech. Expressive aphasia contrasts with receptive aphasia, which is characterized by a patient's inability to comprehend language or speak with appropriately meaningful words.[1]
Contents
Presentation
Sufferers of this form of aphasia exhibit the common problem of agrammatism. For them, speech is difficult to initiate, non-fluent, labored, and halting. Similarly, writing is difficult as well. Intonation and stress patterns are deficient. Language is reduced to disjointed words and sentence construction is poor, omitting function words and inflections (bound morphemes). A person with expressive aphasia might say "Son ... University ... Smart ... Boy ... Good ... Good ... "
For example, in the following passage, a Broca's aphasic patient is trying to explain how he came to the hospital for dental surgery:
- Yes... ah... Monday... er... Dad and Peter H... (his own name), and Dad.... er... hospital... and ah... Wednesday... Wednesday, nine o'clock... and oh... Thursday... ten o'clock, ah doctors... two... an' doctors... and er... teeth... yah.[2]
Patients who communicated with sign language before the onset of the aphasia experience analogous symptoms.[3]
Severity of expressive aphasia varies among patients. In the most extreme cases, patients may be only able to produce a single word. The most famous case of this was Paul Broca's patient Leborgne, nicknamed "Tan", after the only syllable he could say. Even in such cases, over-learned and rote-learned speech patterns may be retained[4]—for instance, some patients can count from one to ten, but cannot produce the same numbers in ordinary conversation.
While word comprehension is generally preserved, meaning interpretation dependent on syntax and phrase structure is substantially impaired. This can be demonstrated by using phrases with unusual structures. A typical Broca's aphasic patient will misinterpret "the dog is bitten by the man" by switching the subject and object.[5] Note this element is a problem with receptive language, not expressive language, and is one reason why the problem is referred to as agrammatic aphasia.
Patients who recover go on to say that they knew what they wanted to say but could not express themselves. Residual deficits will often be seen.
Classification and diagnosis
Expressive aphasia is also a classification of non-fluent aphasia, as opposed to fluent aphasia. Diagnosis is done on a case by case basis, as lesions often affect surrounding cortex and deficits are not well conserved between patients.
Causes
The most common cause of expressive aphasia is stroke. A stroke is caused by hypoperfusion (lack of oxygen) to an area of the brain, which is commonly caused by thrombosis or embolism. Some form of aphasia occurs in 34-38% of stroke patients.[6] Expressive aphasia occurs in approximately 12% of new cases of aphasia caused by stroke.[7] In most cases, expressive aphasia is caused by a stroke in Broca's area or the surrounding vicinity. However, cases of expressive aphasia have been seen in patients with strokes in other areas of the brain. Patients with classic symptoms of expressive aphasia generally have more acute brain lesions while patients with larger, widespread lesions exhibit a variety of symptoms which may be classified as global aphasia or left unclassified.[6]
Expressive aphasia can also be caused by trauma to the brain, tumor, cerebral hemorrhage,[8] by extradural hematoma.[9]
Understanding lateralization of brain function is important for understanding what areas of the brain cause expressive aphasia when damaged. In the past, it has been believed that the area for language production differs between left and right-handed individuals. If this were true, damage to the homologous region of Broca's area in the right hemisphere should cause aphasia in a left-handed individual. More recent studies have shown that even left-handed individuals typically have language functions only in the left hemisphere. However, left-handed individuals are more likely to have a dominance of language in the right hemisphere.[1]
Treatment
Currently, there is no standard treatment for expressive aphasia. Most aphasia therapy is individualized based on a patient's condition and needs as assessed by a speech therapist. The majority of patients go through a period of spontaneous recovery following brain injury in which they regain a great deal of language function. In the months following injury or stroke, most patients receive traditional therapy for a few hours per day. Among other exercises, patients practice the repetition of words and phrases. Mechanisms are also taught in traditional therapy to compensate for lost language function such as drawing and using phrases which are easier to pronounce.[10] Emphasis is placed on establishing a basis for communication with family and caregivers in everyday life. The following treatments are currently being studied to determine the best possible method for treating aphasia.
Singing and melodic intonation therapy
Melodic intonation therapy was inspired by the observation that individuals with non-fluent aphasia sometimes can sing words or phrases that they normally cannot speak. This phenomenon has been noticed for the past 250 years. In some studies patients were able to sing entire songs with provided text that they could not speak with normal intonation.[11] It is believed that this is because singing capabilities are stored in the right hemisphere of the brain, which is likely to remain unaffected after a stroke in the left hemisphere.[12] The goal of melodic intonation therapy is to utilize singing to access the language-capable regions in the right hemisphere and use these regions to compensate for lost function in the left hemisphere. Because patients are better at singing phrases than speaking them, the natural musical component of speech is used to engage the patients ability to voice phrases. Melodic intonation therapy has been shown to work particularly well in patients with large lesions in the left hemisphere.[12]
MIT therapy on average lasts for 1.5 hours per day for five days per week. At the lowest level of therapy, simple words and phrases (such as "water" and "I love you") are broken down into a series of high and low pitch syllables. With increased treatment, longer phrases are taught and less support is provided by the therapist. Patients are taught to say phrases using the natural melodic component of speaking and continuous voicing is emphasized.[12] The patient is also instructed to use their left hand to tap the syllables of the phrase while the phrases are spoken. Tapping further triggers the rhythmic component of speaking to utilize the right hemisphere.[12]
The efficacy of melodic intonation therapy has been proven by studies that show that MIT can result in greater recovery when compared to non intonation therapy.[11] FMRI studies have proven that melodic intonation therapy uses both sides of the brain to recover lost function as opposed to traditional therapies that only utilize the left hemisphere. Furthermore, it has been seen that in MIT, individuals with small lesions in the left hemisphere seem to recover by activation of the left hemisphere perilesional cortex while in individuals with larger left-hemisphere lesions, there is a recruitment of the use of language-capable regions in the right hemisphere.[12]
Constraint induced therapy
Constraint induced aphasia therapy (CIAT) is based on similar principles as constraint-induced movement therapy developed by Dr. Edward Taub at the University of Alabama at Birmingham.[10][13] Constraint induced movement therapy is based on the idea that a person with an impairment (physical or communicative) develops a "learned nonuse" by compensating for their lost function with other means such as using an unaffected limb by a paralyzed individual or drawing by a patient with aphasia.[14] In constraint induced movement therapy, the alternative limb is constrained with a glove or sling and the patient is forced to use their affected limb. In constraint induced aphasia therapy, the interaction is guided by communicative need in a language game context, picture cards, barriers making it impossible to see other players' cards, and other materials, so that patients are encouraged ("constrained") to use their remaining verbal abilities to succeed in the communication game.[13]
Two important principles of constraint induced aphasia therapy are that treatment is very intense with sessions lasting for up to 6 hours over the course of 10 days and that language is used in a communication context where it is closely linked to (nonverbal) actions.[10][13] These principles are motivated by neuroscience insights about learning at the level of nerve cells (synaptic plasticity) and the coupling between cortical systems for language and action in the human brain.[14] Constraint induced therapy contrasts sharply with traditional therapy by the strong belief that mechanisms to compensate for lost language function should not be used unless absolutely necessary, even in everyday life.[10]
It is believed that CIAT works by the mechanism of increased neuroplasticity. By constraining an individual to use only speech, it is believed that their brain can reestablish old neural pathways and recruit new neural pathways to compensate for lost function.
The greatest advantage of CIAT has been seen in its treatment of chronic aphasia (lasting over 1 year). Studies of CIAT have shown that further improvement is possible even after a patient has reached a "plateau" period of recovery.[10][13] It has also been proven that the benefits of CIAT are retained long term. However, improvements only seem to be made while a patient is undergoing intense therapy.[10] A recent breakthrough has been achieved by combining constraint-induced aphasia therapy with drug treatment, which led to an amplification of therapy benefits.[15]
Pharmacotherapy
In addition to active speech therapy, pharmaceuticals have also been considered as a useful treatment for expressive aphasia. This area of study is relatively new and much research continues to be conducted.
The following drugs have been suggested for use in treating aphasia and their efficacy has been studied in control studies.
- Bromocriptine – acts on Catecholamine Systems[16]
- Piracetam – mechanism not fully understood, but most likely interacts with cholinergic and glutamatergic receptors, among others[16]
- Cholinergic drugs (Donepezil, Aniracetam, Bifemelane) – acts on acetylcholine systems[16]
- Amphetaminic drugs (Dexamphetamine, Methylphenidate)[16]
The most effect has been shown by piracetam and amphetamine which may increase cerebral plasticity and result in an increased capability to improve language function. It has been seen that priacetam is most effective when treatment is begun immediately following stroke. When used in chronic cases it has been much less efficient.[15]
Bromocriptine has been shown by some studies to increase verbal fluency and word retrieval with therapy than with just therapy alone.[16] Furthermore, its use seems to be restricted to non-fluent aphasia.[15]
Donepezil has shown a potential for helping chronic aphasia.[15]
No study has established irrefutable evidence that any drug is an effective treatment for aphasia therapy.[16] Furthermore, no study has shown any drug to be specific for language recovery.[15] Comparison between the recovery of language function and other motor function using any drug has shown that improvement is due to a global increase plasticity of neural networks.[16] Pharmaceutical therapy remains an important area of study in aphasia treatment.
Transcranial magnetic stimulation
In transcranial magnetic stimulation (TMS), magnetic fields are used to create electrical currents in specified cortical regions. The procedure is a painless and noninvasive method of stimulating the cortex. TMS works by suppressing the inhibition process in certain areas of the brain.[17] By suppressing the inhibition of neurons by external factors, the targeted area of the brain may be reactivated and thereby recruited to compensate for lost function. Research has shown that patients can show increased object naming ability with regular transcranial magnetic stimulation than patients in therapy without TMS.[17] Furthermore, this improvement has been proven to be permanent and remains upon the completion of TMS therapy.[17] However, some patients fail to show any significant improvement from TMS which indicates the need for further research of this treatment.[18]
Mechanisms of recovery
Mechanisms for recovery differ from patient to patient. Some mechanisms for recovery occur spontaneously after damage to the brain while others are caused by the effects of language therapy.[15] FMRI studies have shown that recovery can be partially attributed to the activation of tissue around the damaged area and the recruitment of new neurons in these areas to compensate for the lost function. Recovery may also be caused in very acute lesions by a return of blood flow and function to damaged tissue that has not died around an injured area.[15] It has been stated by some researchers that the recruitment and recovery of neurons in the left hemisphere apposed to the recruitment of similar neurons in the right hemisphere is superior for long term recovery and continued rehabilitation.[19] It is thought that because the right hemisphere is not intended for full language function, using the right hemisphere as a mechanism of recovery is effectively a "dead-end" and can only lead to partial recovery.[18]
Among all types of therapies it has been proven that one of the most important factors and best predictors for a successful outcome is the intensity of the therapy. By comparing the length and intensity of various methods of therapies, it was proven that intensity was a better predictor of recovery than the method of therapy used.[20]
Prognosis
In most individuals with expressive aphasia, the majority of recovery is seen within the first year following a stroke or injury. The majority of this improvement is seen in the first four weeks in therapy following a stroke and slows thereafter.[6] When compared to patients with the most common types of aphasia, patients with expressive aphasia tend to show the most improvement within the first year. This may be due to an expressive aphasiac's awareness and greater insight of their impairment (unlike in receptive aphasia) which motivates them to progress in treatment.[6] Studies have also found that prognosis of expressive aphasia correlates strongly with the initial severity of impairment. Those with the greatest initial disability tend to show the greatest improvement among test groups.[7] Within the first year, the diagnosis of patients with expressive aphasia may change to anomic aphasia. Similarly, patients diagnosed with global aphasia may be re-diagnosed with expressive aphasia upon improvement.[7] Typically, little improvement is seen after the first year following a stroke. However, it has been seen that continued recovery is possible years after a stroke with effective treatment using methods such as constraint induced aphasia therapy.[10] Depression, anxiety, and social withdrawal are all factors which have been proven to negatively affect a patient's chance of recovery.[21]
Location and size of the brain lesion may also play a role in the prognosis of aphasia. It has been seen in receptive aphasia that larger lesions correlate to slower recovery.[22] It has also been seen that patients with aphasia caused by sub cortical lesions have a better chance of recovery than those with aphasia due to cortical stroke.[23]
History
Expressive aphasia was first identified by the French neurologist Paul Broca. By examining the brains of deceased individuals who acquired expressive aphasia in life, he concluded that language ability was localized in the ventroposterior region of the frontal lobe. One of the most important aspects of Paul Broca's discovery was the observation that the loss of proper speech in expressive aphasia was due to the brain's loss of ability to produce language as opposed to the mouth's loss of ability to produce words.[1]
The discoveries of Paul Broca were made during the same period of time as the German Neurologist Carl Wernicke who was also studying brains of aphasiacs post-mortem and identified the region now known as Wernicke's area. Discoveries of both men contributed to the concept of localization which states that specific brain functions are all localized to a specific area of the brain. While both men made significant contributions to the field of aphasia, it was Carl Wernicke who realized the difference between patients with aphasia who could not produce language and those who could not comprehend language (the essential difference between expressive and receptive aphasia).[1]
Expressive aphasia in popular culture
*The protagonist of Stephen King's novel Duma Key exhibited symptoms of a condition similar to receptive aphasia after suffering brain damage in an industrial accident. When trying to recall some words, he would frequently substitute a synonym of a similar-sounding word, such as trying to say "chair" but instead saying "burn" (a synonym of "char") and "friend" (a synonym of "chum").
Famous sufferers
- Aubrey Plaza
- Ram Dass
- Osmar Santos
- Bob Woodruff
- Kevin Ryder
References
- ^ a b c d e f Purves, D. (2008). Neuroscience (fourth ed.). Sinauer Associates, Inc.. ISBN 0878937420.
- ^ Goodglass, H.; N. Geschwind (1976). "Language disorders". In E. Carterette and M.P. Friedman. Handbook of Perception: Language and Speech. Vol VII. New York: Academic Press.
- ^ http://pages.slc.edu/~ebj/IM_97/Lecture10/L10.html
- ^ "Specific Syndromes: The Nonfluent Aphasias". Neuropathologies of Language and Cognition. http://www.csuchico.edu/~pmccaff/syllabi/SPPA336/336unit7.html. Retrieved 2006-05-10.
- ^ "Neurology of Syntax". Behavioral and Brain Sciences 23 (1). http://bbsonline.cup.cam.ac.uk/Preprints/OldArchive/bbs.grodzinsky.html. Retrieved 2006-05-10.
- ^ a b c d Bakheit, AMO; Shaw, S; Carrington, S; Griffiths, S (2007). "The rate and extent of improvement with therapy from the different types of aphasia in the first year of stroke". Clinical Rehabilitation 21 (10): 941–949. doi:10.1177/0269215507078452. PMID 17981853.
- ^ a b c Pedersen, PM; Vinter, K; Olsen, TS (2004). "Aphasia after stroke: Type, severity, and prognosis - The Copenhagen aphasia study". Cerebrovascular diseases 17 (1): 35–43. doi:10.1159/000073896. PMID 14530636.
- ^ Orzeren, A; F Koc, M Demirkiran, A Sonmezler (2006). "Global aphasia due to left thalamic hemorrhage". Neurology India 54 (4): 415–417. doi:10.4103/0028-3886.28118. PMID 17114855.
- ^ Commondoor, R., Eisenhut, M., Fowler, C., Kirollos, R. W., & Nathwani, N. (2009). "Transient Broca's Aphasia as Feature of an Extradural Abscess". Pediatric Neurology 40 (1): 50–53. doi:10.1016/j.pediatrneurol.2008.06.018. PMID 19068255.
- ^ a b c d e f g Meinzer, Marcus; Thomas Elbert, Daniela Djundja, Edward Taub (2007). "Extending the Constraint-Induced Movement Therapy (CIMT) approach to cognitive functions: Constraint-Induced Aphasia Therapy (CIAT) of chronic aphasia". NeuroRehabilitation 22 (4): 311–318. PMID 17971622.
- ^ a b Wilson, Sarah; Kate Parsons, David Reutens (2006). "Preserved singing in Aphasia: A Case Study of the Efficacy of Melodic Intonation Therapy". Music Perception 24 (1): 23–36.
- ^ a b c d e Schlaug, Gottfried; Sarah Marchina, Andrea Norton (2008). "From Singing to Speaking: Why singing may lead to recovery of expressive language function in patients with Broca's Aphasia". Music Perception 25 (4): 315–319. doi:10.1525/mp.2008.25.4.315.
- ^ a b c d Pulvermuller, Friedemann; et al (2001). "Constraint-Induced Therapy of Chronic Aphasia following Stroke". Stroke 32 (7): 1621–1626. doi:10.1161/01.STR.32.7.1621. PMID 11441210. http://stroke.ahajournals.org/cgi/content/full/32/7/1621.
- ^ a b Pulvermuller, Friedemann; Marcelo Berthier (2008). "Aphasia therapy on a neuroscience basis". Aphasiology 22 (6): 563–599. doi:10.1080/02687030701612213. PMC 2557073. PMID 18923644. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2557073.
- ^ a b c d e f g Berthier, Marcelo; et al. (2009). "Memantine and constraint-induced aphasia therapy in chronic poststroke aphasia". Annals of Neurology 65 (5): 577–578. doi:10.1002/ana.21597. PMID 19475666.
- ^ a b c d e f g Xavier, de Boissezon; Patrice Peran (2007). "Pharmacotherapy of aphasia: Myth or reality?". Brain and Language 102 (1): 114–125. doi:10.1016/j.bandl.2006.07.004. PMID 16982084.
- ^ a b c Margaret, Naeser; Paula Martin, Marjorie Nicholas, Errol Baker (2004). "Improved picture naming in chronic aphasia after TMS to part of right Broca". Brain and Language 93 (1): 95–105. doi:10.1016/j.bandl.2004.08.004. PMID 15766771.
- ^ a b Martin, Paula; Margaret Naeser, Michael Ho, Karl Doron, Jacquie Kurland (2009). "Overt Naming fMRI Pre- and Post- TMS: Two Nonfluent Aphasia Patients, with and without Improved Naming Post- TMS". Brain and Language 111 (1): 20–35. doi:10.1016/j.bandl.2009.07.007. PMC 2803355. PMID 19695692. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2803355.
- ^ Heiss, W-D; Kessler, J; Thiel, A; Ghaemi, M; Karbe, H (1999). "Differential capacity of left and right hemispheric areas for compensation of poststroke". Ann Neurol 45 (4): 430–438. doi:10.1002/1531-8249(199904)45:4<430::AID-ANA3>3.0.CO;2-P. PMID 10211466.
- ^ Sanjit, Bhogal; Robert Teasell, Mark Speechley, Martin Albert (2003). "Intensity of Aphasia Therapy, Impact on Recovery * Aphasia Therapy Works!". Stroke 34 (4): 987–993. doi:10.1161/01.STR.0000062343.64383.D0.
- ^ Code, C; Hemsley, G; Herrmann, M (1999). "The emotional impact of aphasia". Semin Speech Lang 20 (1): 19–31. doi:10.1055/s-2008-1064006. PMID 10100374.
- ^ Naeser, MA (1987). "Relationship between lesion extent in 'Wernicke's area' on computed tomographic scan and predicting recovery of comprehension in Wernicke's aphasia". Arch Neurol 44: 3–82.
- ^ Liang, Cl; Chang, HW; Lu, K; Lee, TC; Liliang, PC; Lu, CH; Chen, HJ (2001). "Early prediction of aphasia outcome in left basal ganglia haemorrhage". Acta Neurol Scand 103 (3): 148–152. doi:10.1034/j.1600-0404.2001.103003148.x. PMID 11240561.
External links
- Aphasia Center of California in Oakland, CA, U.S.
- video of person with Broca's Aphasia
Developmental disorders: Dyslexia and related specific developmental disorders (F80–F83, 315) General conditions Expressive language disorder · Aphasia/Dysphasia (Expressive aphasia, Receptive aphasia) · Landau–Kleffner syndrome · Lisp · Mixed receptive-expressive language disorderScholastic skills/
learning disorderDyslexia (Alexia (acquired dyslexia), Developmental dyslexia) · Dysgraphia (Disorder of written expression) · Dyscalculia (Gerstmann syndrome)Motor functionOtherRelated topics Dyslexia research · Management of dyslexia/Dyslexia interventions · Reading acquisition · Writing system · Spelling · Literacy · Irlen filters · Recording for the Blind & Dyslexic · NeuropsychologyLists Symptoms and signs: Speech and voice / Symptoms involving head and neck (R47–R49, 784) Aphasia/Dysphasia Other speech disturbances Symbolic dysfunctions Dyslexia/Alexia · Agnosia (Prosopagnosia, Astereognosis, Gerstmann syndrome) · Dyspraxia/Apraxia (Ideomotor apraxia) · Dyscalculia/Acalculia · AgraphiaVoice disturbances Other Categories:- Aphasias
Wikimedia Foundation. 2010.
