- Leptospirosis
-
Leptospirosis Classification and external resources 
Leptospirose magnified 200 times with dark-field microscopeICD-10 A27 ICD-9 100 OMIM 607948 DiseasesDB 7403 MedlinePlus 001376 eMedicine med/1283 emerg/856 ped/1298 MeSH C01.252.400.511 Leptospirosis (also known as Weil's syndrome, canicola fever, canefield fever, nanukayami fever, 7-day fever, Rat Catcher's Yellows, Fort Bragg fever, black jaundice and Pretibial fever[1]:290) is caused by infection with bacteria of the genus Leptospira, and affects humans as well as other mammals, birds, amphibians, and reptiles.
The disease was first described by Adolf Weil in 1886 when he reported an "acute infectious disease with enlargement of spleen, jaundice and nephritis". Leptospira was first observed in 1907 from a post mortem renal tissue slice.[2] In 1908, Inada and Ito first identified it as the causative organism[3] and in 1916 noted its presence in rats.[4]
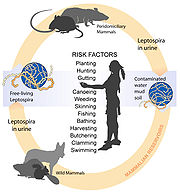
Though recognised among the world's most common diseases transmitted to people from animals, leptospirosis is nonetheless a relatively rare bacterial infection in humans. The infection is commonly transmitted to humans by allowing water that has been contaminated by animal urine to come in contact with unhealed breaks in the skin, the eyes, or with the mucous membranes. Outside of tropical areas, leptospirosis cases have a relatively distinct seasonality with most of them occurring in spring and autumn.[citation needed]
Contents
Causes
 Scanning electron micrograph of a number of Leptospira sp. bacteria atop a 0.1 µm polycarbonate filter
Scanning electron micrograph of a number of Leptospira sp. bacteria atop a 0.1 µm polycarbonate filter
Leptospirosis is caused by a spirochaete bacterium called Leptospira spp. There are at least five serovars of importance in the United States and Canada, all of which cause disease in dogs (Icterohaemorrhagiae, Canicola, Pomona, Grippotyphosa, and Bratislava).[5][6][7]
There are other (less common) infectious strains. Genetically different leptospira organisms may be identical serologically and vice versa. Hence, an argument exists on the basis of strain identification. The traditional serologic system is seemingly more useful from a diagnostic and epidemiologic standpoint at the moment (which may change with further development and spread of technologies like polymerase chain reaction (PCR)).
Leptospirosis is transmitted by the urine of an infected animal and is contagious as long as it is still moist. Although rats, mice and moles are important primary hosts, a wide range of other mammals including dogs, deer, rabbits, hedgehogs, cows, sheep, raccoons, possums, skunks, and certain marine mammals are able to carry and transmit the disease as secondary hosts. Dogs may lick the urine of an infected animal off the grass or soil, or drink from an infected puddle. There have been reports of "house dogs" contracting leptospirosis apparently from licking the urine of infected mice that entered the house. The type of habitats most likely to carry infective bacteria are muddy riverbanks, ditches, gullies, and muddy livestock rearing areas where there is regular passage of either wild or farm mammals. There is a direct correlation between the amount of rainfall and the incidence of leptospirosis, making it seasonal in temperate climates and year-round in tropical climates.
Leptospirosis is also transmitted by the semen of infected animals.[8] Slaughterhouse workers may contract the disease through contact with infected blood or body fluids.
Humans become infected through contact with water, food, or soil containing urine from these infected animals. This may happen by swallowing contaminated food or water, or through skin contact. The disease is not known to be spread from person to person and cases of bacterial dissemination in convalescence are extremely rare in humans. Leptospirosis is common among water-sport enthusiasts in specific areas as prolonged immersion in water is known to promote the entry of the bacteria. Surfers and whitewater paddlers[9] are at especially high risk in areas that have been shown to contain the bacteria, and can contract the disease by swallowing contaminated water, splashing contaminated water into their eyes or nose, or exposing open wounds to infected water.[10] Occupations at risk include veterinarians, slaughterhouse workers, farmers, sewer workers, and people working on derelict buildings. Rowers are also sometimes known to contract the disease.[5]
Symptoms
Leptospiral infection in humans causes a range of symptoms, and some infected persons may have no symptoms at all. Leptospirosis is a biphasic disease that begins with flu-like symptoms (fever, chills, myalgias, intense headache). The first phase resolves, and the patient is briefly asymptomatic until the second phase begins. This is characterized by meningitis, liver damage (causing jaundice), and renal failure. The infection is often wrongly diagnosed due to the wide range of symptoms. This leads to a lower registered number of cases than exists. Symptoms of leptospirosis include high fever, severe headache, chills, muscle aches, and vomiting, and may include jaundice, red eyes, abdominal pain, diarrhea, and rash. Initial presentation may resemble pneumonia. The symptoms in humans appear after a 4–14 day incubation period.
The incubation period (time of exposure to first symptoms) in animals is anywhere from 2 to 20 days. In dogs the liver and kidney are most commonly damaged by leptospirosis. In addition, there are recent reports of a pulmonary form of canine leptospirosis associated with severe hemorrhage in the lungs similar to the human pulmonary hemorrhagic syndrome.[11][12] Vasculitis may occur, causing edema and potentially disseminated intravascular coagulation (DIC). Myocarditis, pericarditis, meningitis, and uveitis are also possible sequelae.[5] One should strongly suspect leptospirosis and include it as part of a differential diagnosis if the sclerae of the dog's eyes appear jaundiced (even slightly yellow). The absence of jaundice does not eliminate the possibility of leptospirosis, and its presence could indicate hepatitis or other liver pathology rather than leptospirosis. Vomiting, fever, failure to eat, reduced urine output, unusually dark or brown urine, and lethargy are also indications of the disease.
Complications
Complications include meningitis, extreme fatigue, hearing loss, respiratory distress, azotemia, and renal interstitial tubular necrosis, which results in renal failure and often liver failure (the severe form of this disease is known as Weil's disease, though it is sometimes named Weil Syndrome).[13] Cardiovascular problems are also possible.
Diagnosis
On infection the microorganism can be found in blood for the first 7 to 10 days (invoking serologically identifiable reactions) and then moving to the kidneys. After 7 to 10 days the microorganism can be found in fresh urine. Hence, early diagnostic efforts include testing a serum or blood sample serologically with a panel of different strains. It is also possible to culture the microorganism from blood, serum, fresh urine and possibly fresh kidney biopsy. Kidney function tests (Blood Urea Nitrogen and creatinine) as well as blood tests for liver functions are performed. The latter reveal a moderate elevation of transaminases. Brief elevations of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and gamma-glutamyltransferase (GGT) levels are relatively mild. These levels may be normal, even in children with jaundice. Diagnosis of leptospirosis is confirmed with tests such as Enzyme-Linked Immunosorbent Assay (ELISA) and polymerase chain reaction (PCR). Serological testing, the MAT (microscopic agglutination test), is considered the gold standard in diagnosing leptospirosis. As a large panel of different leptospira need to be subcultured frequently, which is both laborious and expensive, it is underused, mainly in developing countries.
Differential diagnosis list for leptospirosis is very large due to diverse symptomatics. For forms with middle to high severity, the list includes dengue fever and other hemorrhagic fevers, hepatitis of various etiologies, viral meningitis, malaria, and typhoid fever. Light forms should be distinguished from influenza and other related viral diseases. Specific tests are a must for proper diagnosis of leptospirosis. Under circumstances of limited access (e.g., developing countries) to specific diagnostic means, close attention must be paid to anamnesis of the patient. Factors such as certain dwelling areas, seasonality, contact with stagnant contaminated water (bathing swimming, working on flooded meadows, etc.) or rodents in the medical history support the leptospirosis hypothesis and serve as indications for specific tests (if available).
Leptospira can be cultured in Ellinghausen-McCullough-Johnson-Harris medium, which is incubated at 28 to 30 °C.[14] The median time to positivity is three weeks with a maximum of three months. This makes culture techniques useless for diagnostic purposes, but is commonly used in research.
Prevention and treatment
Doxycycline may be used as a prophylaxis 200–250 mg once a week, to prevent infection in adventure travelers to high risk areas.[15] Treatment is a relatively complicated process comprising two main components: suppressing the causative agent and fighting possible complications. Aetiotropic drugs are antibiotics, such as cefotaxime, doxycycline, penicillin, ampicillin, and amoxicillin.
There are no human vaccines; animal vaccines are only for a few strains, and are only effective for a few months. Human therapeutic dosage of drugs is as follows: doxycycline 100 mg orally every 12 hours for 1 week or penicillin 1–1.5 MU every 4 hours for 1 week. In dogs, penicillin is most commonly used to end the leptospiremic phase (infection of the blood), and doxycycline is used to eliminate the carrier state.
Supportive therapy measures (especially in severe cases) include detoxification and normalization of the hydro-electrolytic balance. Glucose and salt solution infusions may be administered; dialysis is used in serious cases. Elevations of serum potassium are common and if the potassium level gets too high special measures must be taken. Serum phosphorus levels may likewise increase to unacceptable levels due to renal failure. Treatment for hyperphosphatemia consists of treating the underlying disease, dialysis where appropriate, or oral administration of calcium carbonate, but not without first checking the serum calcium levels (these two levels are related). Corticosteroids administration in gradually reduced doses (e.g., prednisolone starting from 30–60 mg) during 7–10 days is recommended by some[citation needed] specialists in cases of severe haemorrhagic effects. Organ specific care and treatment are essential in cases of renal, liver, or heart involvement.
Epidemiology
Annual rates of infection vary from 0.02 per 100,000 in temperate climates to 10 to 100 per 100,000 in tropical climates.[15]
Nepal is experiencing a leptospirosis outbreak as of August 2010,[16] having favorable conditions like warm temperatures and moist soils. Cases increase in summer months (monsoon), but no accurate case count has been made as most go unreported.[17] Almost all the higher animals and rodent hosts of Leptospira occur in Nepal.[17]
History
Leptospirosis was postulated as the cause of an epidemic among Native Americans along the coast of present-day Massachusetts that occurred immediately before the arrival of the Pilgrims in 1620 and killed most of the native population.[18] Earlier proposals included plague, yellow fever, smallpox, influenza, chickenpox, typhus, typhoid fever, trichinellosis, meningitis, and syndemic infection of hepatitis B virus with the delta agent.[19][20][21][22] None are as consistent with all the evidence as leptospirosis. While the disease may have been brought to the New World by Europeans, its spread was also influenced by the high-risk daily activities of the Native Americans. The leptospirosis hypothesis is supported by the occurrence of modern outbreaks identified as severe leptospirosis, some accompanied by high mortality rates (the Andaman Islands in the late 1980s, the Philippines in 2009, Ireland in 2010).[23][24][25]
The cause of this epidemic has been a mystery, while other outbreaks in the same time frame are fairly well established. The epidemic is considered a pivotal event in American history since the failure of the Plymouth Colony might have meant the failure of British colonization in North America. A noted historian has said that the epidemic was the most important event in American history between the discovery of America by Columbus and the signing of the Declaration of Independence.[citation needed]
Before Weil's characterization in 1886, the disease known as infectious jaundice was very likely the same as Weil's disease, or severe icteric leptospirosis. During the Egyptian campaign, Napoleon's army suffered from what was probably infectious jaundice.[26] Infectious jaundice occurred among troops during the American Civil War.[27] It was also reported among troops at Gallipoli and other battles of World War I, where the sodden conditions of trench warfare favored infection. Terms used in early 20th century descriptions of leptospirosis include the pseudo-dengue of Java, seven-day fever, autumn fever, Akiyama disease, and marsh or swamp fever. L icterohaemorrhagieae was identified as the causative agent in pre-World War II outbreaks in Japan, which were characterized by jaundice and a high mortality rate.
In October 2010 British rower Andy Holmes died after contracting Weil's Disease.[28]
In January 2011, Dr. Drew Pinsky, (Loveline, Celebrity Rehab) believes that he contracted leptospirosis while on vacation with his wife in the West Indies due to an outbreak at the time of their visit.[29]
In July 2011 a Danish man died after contracting the disease, probably while cleaning up in his cellar after severe flooding.[30] Heavy rainfall caused the sewers to overflow, flooding large areas of the Danish capital and killing approximately half the rats. The contaminated water got into many of cellars and overflowed into the streets, causing many people to come into contact with sewer water.[31]
References
- ^ James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 0-7216-2921-0.
- ^ Stimson, AM (1907). "Note on an organism found in yellow-fever tissue". Public Health Reports 22: 541.
- ^ Inada R, Ito Y (1908). "A report of the discovery of the causal organism (a new species of spirocheta) of Weil's disease". Tokyo Ijishinshi 1915: 351–60.
- ^ Inanda R, Ido Y, Hoke R, Kaneko R, Ito H (1916). "The Etiology, Mode of Infection and Specific Therapy of Weil's Disease". J Exper Med 23 (3): 377. doi:10.1084/jem.23.3.377.
- ^ a b c Langston CE, Heuter KJ (July 2003). "Leptospirosis. A re-emerging zoonotic disease". The Veterinary clinics of North America. Small animal practice 33 (4): 791–807. doi:10.1016/S0195-5616(03)00026-3. PMID 12910744.
- ^ Kohn B, Steinicke K, Arndt G, Gruber AD, Guerra B, Jansen A, Kaser-Hotz B, Klopfleisch R, Lotz F, Luge E, Nöckler K. (2010). "Pulmonary abnormalities in dogs with leptospirosis". J Vet Intern Med. 24 (6): 1277–82. doi:10.1111/j.1939-1676.2010.0585.x. PMID 20738768.
- ^ Klopfleisch R, Plog S, Kohn B, Weingart, Gruber AD. (2011). "An emerging pulmonary haemorrhagic syndrome in dogs — Similar to the human leptospiral pulmonary haemorrhagic syndrome?". Veterinar Medicine Internationl..
- ^ Kiktenko VS; Balashov, NG; Rodina, VN (1976). "Leptospirosis infection through insemination of animals". J Hyg Epidemiol Microbiol Immunol. 21 (2): 207–213. PMID 987112.
- ^ Shaw RD (June 1992). "Kayaking as a risk factor for leptospirosis". Mo Med 89 (6): 354–7. PMID 1620089.
- ^ transworld.net: Seven Surfing Sicknesses.
- ^ Klopfleisch R, Kohn B, Plog S, Weingart C, Nöckler K, Mayer-Scholl A, Gruber AD. (2011). "An Emerging Pulmonary Haemorrhagic Syndrome in Dogs: Similar to the Human Leptospiral Pulmonary Haemorrhagic Syndrome?". Vet Med Int. 33: 928541. doi:10.4061/2010/928541. PMC 3025382. PMID 21274452. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=3025382.
- ^ Kohn B, Steinicke K, Arndt G, Gruber AD, Guerra B, Jansen A, Kaser-Hotz B, Klopfleisch R, Lotz F, Luge E, Nöckler K. (2010). "Pulmonary abnormalities in dogs with leptospirosis". J Vet Intern Med. 24 (6): 791–807. doi:10.1111/j.1939-1676.2010.0585.x. PMID 20738768.
- ^ Weil syndrome definition - Medical Dictionary definitions of popular medical terms easily defined on MedTerms
- ^ Rule PL, Alexander AD (1986). "Gellan gum as a substitute for agar in leptospiral media". J Clin Microbiol 23 (3): 500–504. PMC 268682. PMID 3754265. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=268682.
- ^ a b Pavli A, Maltezou HC (2008). "Travel-acquired leptospirosis". J Travel Med 15 (6): 447–53. doi:10.1111/j.1708-8305.2008.00257.x. PMID 19090801.
- ^ nepalnews.com
- ^ a b shrawanbhandari.com
- ^ Marr JS, Cathey JT (February 2010). "New hypothesis for cause of an epidemic among Native Americans, New England, 1616–1619". Emerg Infect Dis 16 (2). doi:10.3201/edi1602.090276. http://www.cdc.gov/EID/content/16/2/281.htm.
- ^ Webster N (1799). A brief history of epidemic and pestilential diseases. Hartford CT: Hudson and Goodwin. http://books.google.com/books?id=uen8dansteEC.
- ^ Williams H (1909). "The epidemic of the Indians of New England, 1616–1620, with remarks on Native American infections". John Hopkins Hospital Bulletin 20: 340–9.
- ^ Bratton TL (1988). "The identity of the New England Indian epidemic of 1616–19". Bull Hist Med 62 (3): 351–83. PMID 3067787.
- ^ Speiss A, Speiss BD (1987). "New England pandemic of 1616–1622. cause and archeological implication". Man in the Northeast 34: 71–83.
- ^ Vijayachari P, Sugunan AP, Sharma S, Roy S, Natarajaseenivasan K, Sehgal SC (February 2008). "Leptospirosis in the Andaman Islands, India". Trans. R. Soc. Trop. Med. Hyg. 102 (2): 117–22. doi:10.1016/j.trstmh.2007.08.012. PMID 17991499. http://linkinghub.elsevier.com/retrieve/pii/S0035-9203(07)00283-0.
- ^ ProMED-mail. Leptospirosis - Philippines (02): background. ProMED-mail 2009; 18 Oct: 20091018.3579. Promedmail.org, Accessed 16 January 2010.
- ^ ProMED-mail. Leptospirosis, fatal - Ireland (02): background. ProMED-mail 2010; 06 Jan: 20100106.0055. Promedmail.org Accessed 16 January 2010.
- ^ Edward Rhodes Stitt; Richard Pearson Strong (1944). Stitt's Diagnosis, prevention and treatment of tropical diseases (7th ed.). York, PA: Blakiston. http://books.google.com/books?id=uqU0AAAAIAAJ.
- ^ Neill M (1918). "The problem of acute infectious jaundice in the United States". Public Health Rep 33: 717–26.
- ^ Leggat, David (27 October 2010). "Rowing: Rare disease kills rowing great". The New Zealand Herald. http://www.nzherald.co.nz/sport/news/article.cfm?c_id=4&objectid=10683245. Retrieved 14 October 2011.
- ^ mediaite.com
- ^ " Mand er død efter skybruddet i København". Translated from the Danish.
- ^ [1]
Further reading
- Bharti, A. R.; Nally, JE; Ricaldi, JN; Matthias, MA; Diaz, MM; Lovett, MA; Levett, PN; Gilman, RH et al. (2003). "Leptospirosis: a zoonotic disease of global importance". Lancet Infect. Dis. 3 (12): 757–71. doi:10.1016/S1473-3099(03)00830-2. PMID 14652202.
Infectious diseases · Bacterial diseases: BV4 non-proteobacterial G- (primarily A00–A79, 001–041, 080–109) Spirochaete TreponemaBorrelia recurrentis (Louse borne relapsing fever) · Borrelia hermsii/Borrelia duttoni/Borrelia parkeri (Tick borne relapsing fever)LeptospiraceaeLeptospira interrogans (Leptospirosis)SpirillaceaeChlamydiaceae Bacteroidetes Bacteroides fragilis · Bacteroides forsythus · Capnocytophaga canimorsus · Porphyromonas gingivalis · Prevotella intermediaFusobacteria Fusobacterium necrophorum (Lemierre's syndrome) · Fusobacterium nucleatum · Fusobacterium polymorphumExternal links
- The Leptospirosis Information Center
- U.S. Disease Control and Prevention Center page on Leptospirosis
- Leptonet.net - the Leptospirosis information portal
- International Leptospirosis Society page
- A Symposium on Leptospirosis: Collection of peer-reviewed articles from The Journal of Postgraduate Medicine
- Leptoinfo.com - A website for Dog Owners and Veterinary Professionals dedicated to sharing information on Leptospirosis in Canada
Categories:- Zoonotic bacterial diseases
- Bacterium-related cutaneous conditions
- Bovine diseases
- Zoonoses
- Dog diseases
- Rodent-carried diseases
- Spirochaetes
Wikimedia Foundation. 2010.