- Neural tube
-
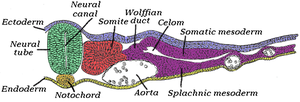
Neural tube Transverse section of half of a chick embryo of forty-five hours' incubation. The dorsal (back) surface of the embryo is towards the top of this page, while the ventral (front) surface is towards the bottom. (Neural tube is in green.) Chick embryo of thirty-three hours' incubation, viewed from the dorsal aspect (30x magnification). Latin tubus neuralis, tuba neuralis Gray's subject #7 50 Carnegie stage 10 Precursor Neural groove Gives rise to Central nervous system (brain and spinal cord) Code TE E5.14.1.0.0.0.1 In the developing vertebrate, the neural tube is the embryo's precursor to the central nervous system, which comprises the brain and spinal cord. The neural groove gradually deepens as the neural folds become elevated, and ultimately the folds meet and coalesce in the middle line and convert the groove into a closed tube, the neural tube or neural canal (which strictly speaking is the center of the neural tube), the ectodermal wall of which forms the rudiment of the nervous system.
Contents
Development
There are 2 ways in which the neural tube develops: Primary neurulation and Secondary neurulation.
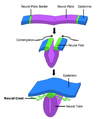
- Primary neurulation divides the ectoderm into three cell types, the neural tube, which is internally located, the epidermis, which is externally located, and the neural crest cells, which develop in the region between the neural tube and epidermis but then migrate to new locations. Primary neurulation begins after the neural plate has formed. The edges of the neural plate start to thicken and lift upward forming the neural folds. The center of the neural plate remains grounded allowing a U-shaped neural groove to form. This neural groove sets the boundary between the right and left sides of the embryo. The neural folds pinch in towards the midline of the embryo and fuse together to form the neural tube.[1]
- In secondary neurulation, the cells of the neural plate form a cord-like structure that migrates inside the embryo and hollows to form the tube.
Each organism uses primary and secondary neurulation to varying degrees.
- Neurulation in fish proceeds only via the secondary form.
- In avian species the posterior regions of the tube develop using secondary neurulation and the anterior regions develop by primary neurulation.
- In mammals, a similar pattern is observed where secondary neurulation begins around the 35th somite.
The manner in which the neural tube closes in mammals in the head is inverted in respect to the manner of closure in the trunk:
- In the head:
- Neural crest cells migrate
- Neural tube closes
- Overlying ectoderm closes
- In the trunk:
- Overlying ectoderm closes
- Neural tube closes
- Neural crest cells migrate
Structure
There are four subdivisions of the neural tube that will each eventually develop into distinct regions of the central nervous system by the division of neuroepithelial cells: The prosencephalon, the mesencephalon, the rhombencephalon and the spinal cord.
- The prosencephalon further goes on to develop into the telencephalon (the forebrain or cerebrum) and the diencephalon (the optic vesicles and hypothalamus).
- The mesencephalon develops into the midbrain.
- The rhombencephalon develops into the metencephalon (the pons and cerebellum) and the myelencephalon (the medulla oblongata).
For a short time, the neural tube is open both cranially and caudally. These openings, called neuropores, close during the fourth week in the human. Improper closure of the neuropores can result in neural tube defects such as anencephaly or spina bifida.
The dorsal part of the neural tube contains the alar plate, which is primarily associated with sensation. The ventral part of the neural tube contains the basal plate, which is primarily associated with motor (i.e., muscle) control.
Dorsal-ventral patterning
The neural tube becomes patterned along the dorsal-ventral axis to establish defined compartments of neural progenitor cells, which will give rise to distinct classes of neurons.[2] This patterning occurs early in development and results from the activity of several secreted signaling molecules. Sonic hedgehog (Shh) is a key player in patterning the ventral axis, while Bone morphogenic proteins (Bmp) and Wnt family members play an important role in patterning the dorsal axis.[3] Other factors shown to provide positional information to the neural progenitor cells include Fibroblast growth factors (FGF) and Retinoic Acid. Retinoic acid is required ventrally along with Shh to induce Pax6 and Olig2 during differentiation of motor neurons.[4] Three main ventral cell types are established during early neural tube development, these include the floor plate cells, which form at the ventral midline during the neural fold stage, as well as the more dorsally located motor neurons and interneurons.[2] These cell types are specified by the secretion of Shh from the notochord (located ventrally to the neural tube), and later from the floor plate cells.[5] Shh acts as a morphogen, meaning that it acts in a concentration-dependent manner to specify cell types as it moves further from its source.[6] The following is a proposed mechanism for how Shh patterns the ventral neural tube: A gradient of Shh is created which controls the expression of a group of homeodomain (HD) and basic Helix-Loop-Helix (bHLH) transcription factors. These transcription factors are grouped into two protein classes based on how Shh affects them, Class I is inhibited by Shh while Class II is activated by Shh. These two classes of proteins then cross-regulate each other to create more defined boundaries of expression. The different combinations of expression of these transcription factors along the dorsal-ventral axis of the neural tube are responsible for creating the identity of the neuronal progenitor cells.[3] Five molecularly distinct groups of ventral neurons form from these neuronal progenitor cells in vitro. Also, the position at which these neuronal groups are generated in vivo can be predicted by the concentration of Shh required for their induction in vitro.[7] Studies have shown that neural progenitors can evoke different responses based on the length of exposure to Shh, with a longer exposure time resulting in more ventral cell types.[8][9]
At the dorsal end of the neural tube, BMPs are responsible for neuronal patterning. BMP is initially secreted from the overlying ectoderm. A secondary signaling center is then established in the roof plate, the dorsal most structure of the neural tube.[1] BMP from the dorsal end of the neural tube seems to act in the same concentration-dependent manner as Shh in the ventral end.[10] This was shown using zebrafish mutants that had varying amounts of BMP signaling activity. Researchers observed changes in dorsal-ventral patterning, for example zebrafish deficient in certain BMPs showed a loss of dorsal sensory neurons and an expansion of interneurons.[11]
See also
- Neural tube defects
- Cdx protein family
References
- ^ a b Gilbert, Scott F. Developmental Biology Eighth Edition. Sunderland, Massachusetts: Sinauer Associates, Inc., 2006.
- ^ a b Jessell, TM. “Neuronal specification in the spinal cord: inductive signals and transcriptional codes.” Nat Rev Genet 2000. 1:20-9.
- ^ a b Ulloa, F, Marti, E. “Wnt won the war: Antagonistic role of Wnt over Shh controls dorso-ventral patterning of the vertebrate neural tube.” Developmental Dynamics 2010. 239:69-76.
- ^ Duester, G. (2008) Retinoic acid synthesis and signaling during early organogenesis. Cell 134: 921-931. PMCID: PMC2632951
- ^ Patten, I, Placzek, M. “The role of Sonic hedgehog in neural tube patterning.” Cell Mol Life Sci 2000. 12:1695-708.
- ^ Dessaud, E, McMahon, AP, Briscoe, J. “Pattern formation in the vertebrate neural tube: a sonic hedgehog morphogen-regulated transcriptional network.” Development 2008. 135:2489-2503.
- ^ Ericson, J, Briscoe, J, Rashbass, P, van Heyningen, V, Jessell, TM. “Graded Sonic hedgehog signaling and the specification of cell fate in the ventral neural tube.” Cold Spring Harb Symp Quant Biol 1997. 62:451-466.
- ^ Stamataki, D, Ulloa, F, Tsoni, SV, Mynett, A, Briscoe, J. “A gradient of Gli activity mediates graded Sonic hedgehog signaling in the neural tube.” Genes Dev 2005. 19:626-641.
- ^ Dessaud, E, Yang, LL, Hill, K, Cox, B, Ulloa, F, Ribeiro, A, Myness, A, Novitch, BG, Briscoe, J. “Interpretation of the sonic hedgehog morphogen gradient by a temporal adaptation mechanism.” Nature 2007. 450:717-720.
- ^ Wilson, L, Maden, M. “The mechanisms of dorsoventral patterning in the vertebrate neural tube.” Developmental Biology 2005. 282:1-13.
- ^ Nguyen, VH, Trout, J, Connors, SA, Andermann, P, Weinberg, E, Mullins, MC. “Dorsal and intermediate neuronal cell types of the spinal cord are established by a BMP signaling pathway.” Development 2000. 127:1209-1220.
Additional images
External links
- Swiss embryology (from UL, UB, and UF) iperiodembry/carnegie03
- Embryology at UNSW Notes/week3_5
- Diagram at embryology.med.unsw.edu.au
- Diagram at brainviews.com
This article was originally based on an entry from a public domain edition of Gray's Anatomy. As such, some of the information contained within it may be outdated.
Prenatal development/Mammalian development of nervous system (GA 9.733 and GA 10.1002, TE E5.13-16) Neurogenesis Neural tubeRostral neuropore
Cephalic flexure · Pontine flexure
Alar plate (sensory) · Basal plate (motor)
Germinal matrixEye development Auditory development M: EYE
anat(g/a/p)/phys/devp/prot
noco/cong/tumr, epon
proc, drug(S1A/1E/1F/1L)
M: EAR
anat(e/p)/phys/devp
noco/cong, epon
proc, drug(S2)
Human cell types / list derived primarily from ectoderm Surface ectoderm Neural crest Digestive systemNeural tube Categories:- Embryology of nervous system
- Neurulation
Wikimedia Foundation. 2010.