- Gel
-
For other uses, see Gel (disambiguation).An upturned vial of hair gel

A gel (from the lat. gelu—freezing, cold, ice or gelatus—frozen, immobile) is a solid, jelly-like material that can have properties ranging from soft and weak to hard and tough. Gels are defined as a substantially dilute cross-linked system, which exhibits no flow when in the steady-state.[1] By weight, gels are mostly liquid, yet they behave like solids due to a three-dimensional cross-linked network within the liquid. It is the crosslinks within the fluid that give a gel its structure (hardness) and contribute to stickiness (tack). In this way gels are a dispersion of molecules of a liquid within a solid in which the solid is the continuous phase and the liquid is the discontinuous phase.
Contents
Composition
Gels consist of a solid three-dimensional network that spans the volume of a liquid medium and ensnares it through surface tension effects. This internal network structure may result from physical bonds (physical gels) or chemical bonds (chemical gels), as well as crystallites or other junctions that remain intact within the extending fluid. Virtually any fluid can be used as an extender including water (hydrogels), oil, and air (aerogel). Both by weight and volume, gels are mostly fluid in composition and thus exhibit densities similar to those of their constituent liquids. Edible jelly is a common example of a hydrogel and has approximately the density of water.
Cationic polymers
Cationic polymers are positively charged polymers. Their positive charges prevent the formation of coiled polymers. This allows them to contribute more to viscosity in their stretched state, because the stretched-out polymer takes up more space Gel is a colloid solution of dispersion phase as liquid and dispersion medium as solid
Types of gels
Hydrogels
See also: Superabsorbent polymerHydrogel (also called aquagel) is a network of polymer chains that are hydrophilic, sometimes found as a colloidal gel in which water is the dispersion medium. Hydrogels are highly absorbent (they can contain over 99.9% water) natural or synthetic polymers. Hydrogels also possess a degree of flexibility very similar to natural tissue, due to their significant water content. Common uses for hydrogels include
- currently used as scaffolds in tissue engineering. When used as scaffolds, hydrogels may contain human cells to repair tissue.
- environmentally sensitive hydrogels which are also known as 'Smart Gels' or 'Intelligent Gels'. These hydrogels have the ability to sense changes of pH, temperature, or the concentration of metabolite and release their load as result of such a change.
- as sustained-release drug delivery systems.
- provide absorption, desloughing and debriding of necrotic and fibrotic tissue.
- hydrogels that are responsive to specific molecules, such as glucose or antigens, can be used as biosensors, as well as in DDS.
- used in disposable diapers where they absorb urine, or in sanitary napkins
- contact lenses (silicone hydrogels, polyacrylamides)
- EEG and ECG medical electrodes using hydrogels composed of cross-linked polymers (polyethylene oxide, polyAMPS and polyvinylpyrrolidone)
- water gel explosives
- rectal drug delivery and diagnosis
Other, less common uses include
- breast implants
- now used in glue.
- granules for holding soil moisture in arid areas
- dressings for healing of burn or other hard-to-heal wounds. Wound gels are excellent for helping to create or maintain a moist environment.
- reservoirs in topical drug delivery; particularly ionic drugs, delivered by iontophoresis (see ion exchange resin)
Common ingredients are e.g. polyvinyl alcohol, sodium polyacrylate, acrylate polymers and copolymers with an abundance of hydrophilic groups.
Natural hydrogel materials are being investigated for tissue engineering; these materials include agarose, methylcellulose, hyaluronan, and other naturally derived polymers.
Organogels
An organogel is a non-crystalline, non-glassy thermoreversible (thermoplastic) solid material composed of a liquid organic phase entrapped in a three-dimensionally cross-linked network. The liquid can be, for example, an organic solvent, mineral oil, or vegetable oil. The solubility and particle dimensions of the structurant are important characteristics for the elastic properties and firmness of the organogel. Often, these systems are based on self-assembly of the structurant molecules.[2][3]
Organogels have potential for use in a number of applications, such as in pharmaceuticals,[4] cosmetics, art conservation,[5] and food.[6] An example of formation of an undesired thermoreversible network is the occurrence of wax crystallization in petroleum.[7]
Xerogels
A xerogel (
 /ˈzɪərɵdʒɛl/) is a solid formed from a gel by drying with unhindered shrinkage. Xerogels usually retain high porosity (25%) and enormous surface area (150–900 m2/g), along with very small pore size (1-10 nm). When solvent removal occurs under hypercritical (supercritical) conditions, the network does not shrink and a highly porous, low-density material known as an aerogel is produced. Heat treatment of a xerogel at elevated temperature produces viscous sintering (shrinkage of the xerogel due to a small amount of viscous flow) and effectively transforms the porous gel into a dense glass.
/ˈzɪərɵdʒɛl/) is a solid formed from a gel by drying with unhindered shrinkage. Xerogels usually retain high porosity (25%) and enormous surface area (150–900 m2/g), along with very small pore size (1-10 nm). When solvent removal occurs under hypercritical (supercritical) conditions, the network does not shrink and a highly porous, low-density material known as an aerogel is produced. Heat treatment of a xerogel at elevated temperature produces viscous sintering (shrinkage of the xerogel due to a small amount of viscous flow) and effectively transforms the porous gel into a dense glass.Properties
Many gels display thixotropy - they become fluid when agitated, but resolidify when resting. In general, gels are apparently solid, jelly-like materials. By replacing the liquid with gas it is possible to prepare aerogels, materials with exceptional properties including very low density, high specific surface areas, and excellent thermal insulation properties.
Sound-induced gelation
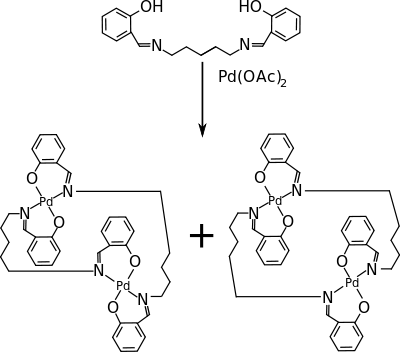
Sound-induced gelation is described in 2005[8] in an organopalladium compound that in solution transforms from a transparent liquid to an opaque gel upon application of a short burst (seconds) of ultrasound. Heating to above the so-called gelation temperature Tgel takes the gel back to the solution. The compound is a dinuclear palladium complex made from palladium acetate and a N,N'-bis-salicylidene diamine. Both compounds react to form an anti conformer (gelling) and a syn conformer (non-gelling) which are separated by column chromatography. In the solution phase, the dimer molecules are bent and self-locked by aromatic stacking interactions, whereas in the gel phase the conformation is planar with interlocked aggregates. The anti conformer has planar chirality and both enantiomers were separated by chiral column chromatography. The (-) anti conformer has a specific rotation of -375° but is unable to gel by itself. In the gel phase, the dimer molecules form stacks of alternating (+) and (−) components. This process starts at the onset of the sonication and proceeds even without further sonication.
Naturally occurring gels in the animal kingdom
Some species secrete gels that are effective in parasite control. For example, the long-finned pilot whale secretes an enzymatic gel that rests on the outer surface of this animal and helps prevent other organisms from establishing colonies on the surface of these whales' bodies.[9]
Applications
Many substances can form gels when a suitable thickener or gelling agent is added to their formula. This approach is common in manufacture of wide range of products, from foods to paints and adhesives.
In fiber optics communications, a soft gel resembling "hair gel" in viscosity is used to fill the plastic tubes containing the fibers. The main purpose of the gel is to prevent water intrusion if the buffer tube is breached, but the gel also buffers the fibers against mechanical damage when the tube is bent around corners during installation, or flexed. Additionally, the gel acts as a processing aid when the cable is being constructed, keeping the fibers central whilst the tube material is extruded around it.
Hydrogels existing naturally in the body include mucus, the vitreous humor of the eye, cartilage, tendons and blood clots. Their viscoelastic nature results in the soft tissue component of the body, disparate from the mineral-based hard tissue of the skeletal system. Researchers are actively developing synthetically derived tissue replacement technologies derived from hydrogels, for both temporary implants (degradable) and permanent implants (non-degradable). A review article on the subject discusses the use of hydrogels for nucleus pulposus replacement, cartilage replacement, and synthetic tissue models.[10]
See also
- 2-Acrylamido-2-methylpropane sulfonic acid
- Aerogel
- Hydrocolloid
- Gel electrophoresis, Agarose gel electrophoresis, 2-D electrophoresis, SDS-PAGE
- Gel filtration chromatography, Gel permeation chromatography
- Paste (rheology)
- Food rheology
- Silicone GEL
References
- ^ Ferry, John D. Viscoelastic Properties of Polymers. New York: Wiley, 1980.
- ^ Terech P. Low-molecular weight organogelators. In: Robb ID, editor. Specialist surfactants. Glasgow: Blackie Academic and Professional, p. 208–268 (1997).
- ^ van Esch J, Schoonbeek F, De Loos M, Veen EM, Kellog RM, Feringa BL. Low molecular weight gelators for organic solvents. In: Ungaro R, Dalcanale E, editors. Supramolecular science: where it is and where it is going. Kluwer Academic Publishers, p. 233–259 (1999).
- ^ Kumar R, Katare OP. Lecithin organogels as a potential phospholipid-structured system for topical drug delivery: A review. American Association of Pharmaceutical Scientists PharmSciTech 6, E298–E310 (2005).
- ^ Carretti E, Dei L, Weiss RG. Soft matter and art conservation. Rheoreversible gels and beyond. Soft Matter 1, 17–22 (2005).
- ^ Pernetti M, van Malssen KF, Flöter E, Bot A. Structuring of edible oil by alternatives to crystalline fat. Current Opinion in Colloid and Interface Science 12, 221–231 (2007).
- ^ Visintin RFG, Lapasin R, Vignati E, D'Antona P, Lockhart TP. Rheological behavior and structural interpretation of waxy crude oil gels. Langmuir 21, 6240–6249 (2005)
- ^ Naota T, Koori H. Molecules That Assemble by Sound: An Application to the Instant Gelation of Stable Organic Fluids. J. Am. Chem. Soc., 127 (26), 9324-9325 (2005) Online details
- ^ Eileen May Dee, Mark McGinley and C.Michael Hogan. 2010. Long-finned pilot whale. Encyclopedia of Earth. National Council for Science and the Environment. Washington DC. eds. Peter Saundry and Cutler Cleveland
- ^ https://www.orthoworld.com/site/index.php/publications/view_article/221558
10. I. Katime, O. Katime, D. Katime.Los materiales inteligentes de este milenio: Los hidrogeles macromoleculares. Síntesis, propiedades y applicaciones. Servicio Editorial de la Universidad del País Vasco (UPV/EHU).Bilbao. Septiembre de 2004
Further reading
- Ajayaghosh, A., Praveen, V.K. & Vijayakumar, C. Organogels as scaffolds for excitation energy transfer and light harvesting. Chem Soc Rev 37, 109-22(2008).
- Ajayaghosh, A. & Praveen, V.K. p-Organogels of Self-Assembled p-Phenylenevinylenes: Soft Materials with Distinct Size, Shape, and Functions. Acc. Chem. Res. 40, 644-656(2007).
- Estroff, L.A. & Hamilton, A.D. Water gelation by small organic molecules. Chem Rev 104, 1201-18(2004).
- Fairclough, J.P.A. & Norman, A.I. Structure and rheology of aqueous gels. Annu. Rep. Prog. Chem., Sect. C 99, 243-276(2003).
- Pich, A.Z. & Adler, H.P. Composite aqueous microgels: an overview of recent advances in synthesis, characterization and application. Polymer International 56, 291-307(2007).
- Viscoelastic Characterization of Agarose Gel Scaffolds
External links
Routes of administration / Dosage forms Oral Buccal / Sublabial / Sublingual- Mouthwash
- Toothpaste
- Ointment
- Oral spray
- Oxygen mask
- Oxygen concentrator
- Anaesthetic machine
- Relative analgesia machine





Ocular / Otologic / Nasal - Nasal spray
- Ear drops
- Eye drops
- Ointment
- Hydrogel
- Nanosphere suspension
- Mucoadhesive microdisc (microsphere tablet)
Urogenital - Ointment
- Pessary (vaginal suppository)
- Vaginal ring
- Vaginal douche
- Intrauterine device (IUD)
- Extra-amniotic infusion
- Intravesical infusion
Rectal (enteral) - Ointment
- Suppository
- Enema (Solution • Hydrogel)
- Murphy drip
- Nutrient enema
Dermal Injection / Infusion
(into tissue/blood)- Intracavernous
- Intravitreal
- Intra-articular or intrasynovial injection
- Transscleral
- Intracerebral
- Intrathecal
- Epidural
Additional explanation: Mucous membranes are used by the human body to absorb the dosage for all routes of administration, except for "Dermal" and "Injection/Infusion".
Administration routes can also be grouped as Topical (local effect) or Systemic (defined as Enteral = Digestive tract/Rectal, or Parenteral = All other routes).Routes of administration by organ system Gastrointestinal Respiratory system Pulmonary • NasalVisual system / Auditory system Ocular (Ocular-topical / Intravitreal / Transscleral) • Otologic (Oto-topical)Reproductive system Intracavernous • Intravaginal • Intrauterine (Extra-amniotic)Urinary system IntravesicalPeritoneum Central nervous system Intracerebral • Intrathecal • EpiduralCirculatory system Musculoskeletal system Skin Epicutaneous • Intradermal • SubcutaneousCategories:- Physical chemistry
- Gels
- Drug delivery devices
- Dosage forms
Wikimedia Foundation. 2010.