- Intrauterine device
-
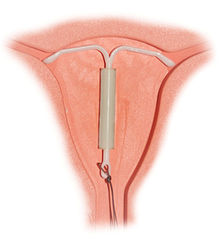
The intrauterine device (IUD) is a form of birth control; it is an object placed in the uterus to prevent pregnancy.[1] Among modern IUDs, the two types available are copper-containing devices, and a hormone-containing device that releases a progestogen. Currently, there are over 10 different kinds of copper IUD available in different parts of the world, and there is one hormonal device, called Mirena.
Contents
Modern IUDs
Names and descriptors of intrauterine contraceptive devices vary by location.
In the United States, there are two types of intrauterine contraceptive available: the copper Paragard and the hormonal Mirena. Both of these contraceptives are referred to as IUDs in the United States.[2]
In the United Kingdom, where over ten types of copper-containing IUDs are available, the term IUD only refers to inert or copper-containing devices. Hormonal intrauterine contraceptives are considered to be a different type of birth control, and they are distinguished with the term intrauterine system or IUS.[3][4]
The WHO/ATC name is IUD for both copper and hormonal devices.
Copper IUDs
Main article: Copper IUDCopper IUDs work by impairing the mobility of sperm and preventing them from joining with an egg. Additionally, the foreign body inside the uterus irritates the lining and wall making it hard for an embryo to implant.[medical citation needed]
Most non-hormonal IUDs have a plastic T-shaped frame that is wound around with pure electrolytic copper wire and/or has copper collars (sleeves). The Paragard T 380a is 32 mm (1.26") in the horizontal direction (top of the T), and 36 mm (1.42") in the vertical direction (leg of the T). In some IUDs, such as the Nova T 380, the pure copper wire has a silver core which has been shown to prevent breaking of the wire.[3][5] The arms of the frame hold the IUD in place near the top of the uterus.
Other shapes of IUD include the so-called U-shaped IUDs, such as the Load and Multiload. These are similar to the T-shaped IUDs, in that they have a plastic "leg" running vertically, but at the top, rather than two straight horizontal arms, they instead have two arms that curve downwards towards the "foot" of the device.
Another shape of IUD is the frameless IUD. It does not have a hard plastic shape of any kind. Rather, it has a plastic string (similar to fishing line) that holds several hollow cylindrical miniscule copper beads. It is held in place by a suture (knot) to the fundus of the uterus. It is mainly available in China, Europe, and Germany, although some clinics in Canada can provide it. It is removed like most other IUDs using forceps but is actually removed from the fundus.[citation needed]
All copper-containing IUDs have a number as part of their name. This is the surface area of copper (in square millimeters) the IUD provides.[citation needed]
Hormonal IUD - Mirena
Main article: Hormonal IUDHormonal uterine devices (sometimes called IntraUterine Systems) do not increase bleeding as copper-containing IUDs do. Rather, they reduce menstrual bleeding or prevent menstruation altogether, and can be used as a treatment for menorrhagia (heavy periods).
Although use of IntraUterine Systems results in much lower systemic progestogen levels than other very-low-dose progestogen-only hormonal contraceptives, they might possibly have some of the same side effects.
As of 2007, the LNG-20 IUS - marketed as Mirena by Bayer - is the only IntraUterine System available. First introduced in 1990, it releases levonorgestrel (a progestogen) and may be used for five years. With the use of the Mirena the hormones are localized to the uterine area unlike oral contraceptives.
Historical IUDs
Copper-containing devices
One historical device is Gräfenberg's ring, an early IUD designed by Ernst Gräfenberg, a German gynecologist in whose honor the G-spot was named.
Another historical IUD is the Dalkon Shield. Produced in the 1970s, the Dalkon Shield became infamous for its serious design flaw: a porous, multifilament string up which bacteria could swim into the uterus of users, leading to sepsis, injury, miscarriage, and death. Modern IUDs use monofilament strings which do not pose this grave risk to users.
Hormone-containing devices
Progestasert was the first hormonal uterine device, developed in 1976[6] and manufactured until 2001.[7] It released progesterone, was replaced annually, and had a failure rate of 2% per year.[8]
Future IUDs
A lower-dose T-shaped IntraUterine System named Femilis is being developed by Contrel, a Belgian company. Contrel also manufactures the FibroPlant-LNG, a frameless IUS. FibroPlant is anchored to the fundus of the uterus as the GyneFix IUD is. Although a number of trials have shown positive results, FibroPlant is not yet commercially available.[9]
References
- ^ "intrauterine device" at Dorland's Medical Dictionary
- ^ Treiman K, Liskin L, Kols A, Rinehart W (1995). "IUDs—an update" (PDF). Popul Rep B (6): 1–35. PMID 8724322. http://www.infoforhealth.org/pr/b6/b6.pdf. Retrieved 2006-01-01.
- ^ a b "Contraceptive coils (IUDs)". NetDoctor.co.uk. 2006. http://www.netdoctor.co.uk/sex_relationships/facts/contraceptivecoil.htm. Retrieved 2006-07-05.
- ^ French, R; Van Vliet H, Cowan F, et al. (2004). Sorhaindo, Annik M.. ed. "Hormonally impregnated intrauterine systems (IUSs) versus other forms of reversible contraceptives as effective methods of preventing pregnancy". Cochrane Database of Systematic Reviews (3): CD001776. doi:10.1002/14651858.CD001776.pub2. PMID 15266453.
- ^ Schering (May 13, 2003). "Nova T380 Patient information leaflet (PIL)". http://emc.medicines.org.uk/emc/assets/c/html/displaydoc.asp?documentid=3641. Retrieved 2007-04-27.
- ^ IUDs—An Update. Chapter 2: Types of IUDs.
- ^ Smith (pseudonym), Sydney (March 8, 2003). "Contraceptive Concerns". medpundit: Commentary on medical news by a practicing physician.. http://www.medpundit.blogspot.com/2003_03_02_medpundit_archive.html. Retrieved 2006-07-16.
- ^ "Birth Control Options: The Progestasert Intrauterine Device (IUD)". Wyoming Health Council. 2004. http://wyhc.org/birth_control_options/Progestasert-IUD.php. Retrieved 2006-07-16.
- ^ "New Contraceptive Choices". Population Reports, INFO Project, Center for Communication Programs (The Johns Hopkins School of Public Health) M (19). April 2005. http://www.infoforhealth.org/pr/m19/. Retrieved 2006-07-14. Chapter 9: Intrauterine Devices.
Birth control methods (G02B, G03A) Comparison Behavioral Avoiding vaginal intercourse: Abstinence • Anal sex • Masturbation • Non-penetrative sex • Oral sex
Including vaginal intercourse: Breastfeeding infertility (LAM) • Calendar-based methods (rhythm, etc.) • Fertility awareness • WithdrawalBarrier or
spermicidalHormonal
(formulations)Progestogen-onlyAnti-estrogen Ormeloxifene (Centchroman)Post-intercourse Intrauterine device Abortion Sterilization Routes of administration / Dosage forms Oral - Mouthwash
- Toothpaste
- Ointment
- Oral spray
- Oxygen mask
- Oxygen concentrator
- Anaesthetic machine
- Relative analgesia machine





Ocular / Otologic / Nasal - Nasal spray
- Ear drops
- Eye drops
- Ointment
- Hydrogel
- Nanosphere suspension
- Mucoadhesive microdisc (microsphere tablet)
Urogenital - Ointment
- Pessary (vaginal suppository)
- Vaginal ring
- Vaginal douche
- Intrauterine device (IUD)
- Extra-amniotic infusion
- Intravesical infusion
Rectal (enteral) - Ointment
- Suppository
- Enema (Solution • Hydrogel)
- Murphy drip
- Nutrient enema
Dermal Injection / Infusion
(into tissue/blood)- Intracavernous
- Intravitreal
- Intra-articular or intrasynovial injection
- Transscleral
- Intracerebral
- Intrathecal
- Epidural
Additional explanation: Mucous membranes are used by the human body to absorb the dosage for all routes of administration, except for "Dermal" and "Injection/Infusion".
Administration routes can also be grouped as Topical (local effect) or Systemic (defined as Enteral = Digestive tract/Rectal, or Parenteral = All other routes).Routes of administration by organ system Gastrointestinal Respiratory system Pulmonary • NasalVisual system / Auditory system Reproductive system Intracavernous • Intravaginal • Intrauterine (Extra-amniotic)Urinary system IntravesicalPeritoneum Central nervous system Circulatory system Musculoskeletal system Skin Categories:- Intrauterine contraception
- Drug delivery devices
- Dosage forms
Wikimedia Foundation. 2010.