- Merlin (bird)
-
Merlin 
Male Prairie Merlin (F. c. richardsoni) with prey in Alberta (Canada) Conservation status Scientific classification Kingdom: Animalia Phylum: Chordata Class: Aves Order: Falconiformes Family: Falconidae Genus: Falco Species: F. columbarius Binomial name Falco columbarius
Linnaeus, 1758Diversity 3–9 subspecies (see text) 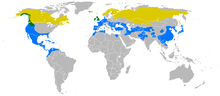
Yellow: summer only. Green: all year. Blue: winter only. Synonyms Aesalon columbarius (Linnaeus, 1758)
Falco aesalon Tunstall, 1771 (but see text)The Merlin (Falco columbarius) is a small species of falcon from the Northern Hemisphere. A bird of prey once known colloquially as a pigeon hawk in North America,[1] the Merlin breeds in the northern Holarctic; some migrate to subtropical and northern tropical regions in winter.
Contents
European and North American variants
Some regard the North American and Eurasian (F. columbarius) populations as two distinct species. The first modern taxonomist to describe the Merlin was Carl Linnaeus, a Swede who reported his type specimen came from America. Thirteen years after Linnaeus's description Marmaduke Tunstall recognized the Eurasian birds as a distinct taxon aesalon in his Ornithologica Britannica. If two species of Merlins are recognized, the Old World birds would thus bear the scientific name F. aesalon.[2]
Description
The Merlin is 24–33 cm (9.4–13 in) long with a 50–67 cm (20–26 in) wingspan. Compared to other small falcons, it is more robust and heavily built. Males average at about 165 g (5.8 oz) and females are typically about 230 g (8.1 oz). There is considerable variation, however, throughout the birds' range and—in particular in migratory populations—over the course of a year. Thus, adult males may weigh 150–210 g (5.3–7.4 oz), and females 190–255 g (6.7–9.0 oz). Such sexual dimorphism is common among raptors; it allows males and females to hunt different prey animals and decreases the territory size needed to feed a mated pair.[3]
The male Merlin has a blue-grey back, ranging from almost black to silver-grey in different subspecies. Its underparts are buff- to orange-tinted and more or less heavily streaked with black to reddish brown. The female and immature are brownish-grey to dark brown above, and whitish buff spotted with brown below. Besides a weak whitish supercilium and the faint dark malar stripe—which are barely recognizable in both the palest and the darkest birds—the face of the Merlin is less strongly patterned than in most other falcons. Nestlings are covered in pale buff down feathers, shading to whitish on the belly.[3]
 Upperside pattern of male (presumably F. c./a. pallidus) wintering in Little Rann of Kutch, Gujerat, India
Upperside pattern of male (presumably F. c./a. pallidus) wintering in Little Rann of Kutch, Gujerat, India
The remiges are blackish, and the tail usually has some 3–4 wide blackish bands, too. Very light males only have faint and narrow medium-grey bands, while in the darkest birds the bands are very wide, so that the tail appears to have narrow lighter bands instead. In all of them, however, the tail tip is black with a narrow white band at the very end, a pattern possibly plesiomorphic for all falcons. Altogether, the tail pattern is quite distinct though, resembling only that of the Aplomado Falcon (F. berigora) and (in light Merlins) some typical kestrels. The eye and beak are dark, the latter with a yellow cere. The feet are also yellow, with black claws.[3]
Light American males may resemble the American Kestrel (F. sparverius, not a typical kestrel), but merlin males have a grey back and tail rather than the reddish-brown of the kestrels. Light European males can be distinguished from kestrels by their mainly brown wings. In the north of South Asia, wintering males may be confused with the Red-necked Falcon (F. chicquera) if they fly away from the observer and the head (red on top in F. chicquera) and underside (finely barred with black in F. chicquera) are not visible.[3]
Systematics
The relationships of the Merlin are not resolved to satisfaction. In size, shape and coloration, it is fairly distinct among living falcons. The Red-necked Falcon is sometimes considered more closely related to the Merlin than other falcons, but this seems to be a coincidence due to similar hunting habits; it could not be confirmed in more recent studies. Indeed, the Merlin seems to represent a lineage distinct from other living falcons since at least the Early Pleistocene, some 5 Ma (million years ago). As suggested by biogeography and DNA sequence data, it might be part of an ancient non-monophyletic radiation of Falcos from Europe to North America, alongside the ancestors of forms such as the American Kestrel (F. sparvierus), and the Aplomado Falcon (F. femoralis) and its relatives. A relationship with the Red-necked Falcon (F. chicquera) was once proposed based on their phenetic similarity, but this is not considered likely today.[4]
In that regard, it is interesting to note a fossil falcon from the Early Blancan (4.3–4.8 Ma)[5] Rexroad Formation of Kansas. Known from an almost complete right coracoid (specimen UMMP V29107) and some tarsometatarsus, tibiotarsus and humerus pieces (V27159, V57508-V57510, V57513-V57514), this prehistoric falcon was slightly smaller than a Merlin and apparently a bit more stout-footed, but otherwise quite similar. It was part of the Fox Canyon and Rexroad Local Faunas, and may have been the ancestor of the living Merlins or its close relative. With its age quite certainly pre-dating the split between the Eurasian and North American Merlins, it agrees with the idea of the Merlin lineage originating in North America, or rather the colonization thereof. After adapting to its ecological niche, ancient Merlins would have spread to Eurasia again, with gene flow being interrupted as the Beringia and Greenland regions became icebound in the Quaternary glaciation.[6]
Subspecies
That the Merlin has a long-standing presence on both sides of the Atlantic is evidenced by the degree of genetic distinctness between Eurasian and North American populations. Arguably, they might be considered distinct species, with gene flow having ceased at least a million years ago, but probably more.[7]
By and large, color variation in either group independently follows Gloger's Rule. The Pacific temperate rain forest subspecies suckleyi's males are almost uniformly black on the upperside and have heavy black blotches on the belly, whereas those of the lightest subspecies, pallidus, have little non-dilute melanin altogether, with grey upperside and reddish underside pattern.[8]
American group[8]
- Falco columbarius columbarius Linnaeus, 1758 – Taiga Merlin, Tundra Merlin
- Canada and northernmost USA east of Rocky Mountains, except Great Plains. Migratory, winters in S North America, Central America, the Caribbean, and N South America from the Guyanas to the northern Andes foothills. Rarely winters in the northern USA.[9]
- Falco columbarius richardsoni Ridgway, 1871 – Prairie Merlin
- Falco columbarius suckleyi Ridgway, 1873 – Coastal Forest Merlin, Black Merlin
- Pacific coast of North America, from S Alaska to N Washington state. Resident (some altitudinal movements).
Eurasian group[8]
- Falco columbarius/aesalon aesalon Tunstall, 1771
- Northern Eurasia from British Isles through Scandinavia to central Siberia. Population of northern Britain shows evidence of gene flow from subaesalon. British Isles population resident, rest migratory; winters in Europe and the Mediterranean region to about Iran.
- Falco columbarius/aesalon subaesalon C.L. Brehm, 1827 – smyril (Faroese), smyrill (Icelandic)
- Iceland and Faroe Islands. Latter population has some gene flow with aesalon. Resident (some winter dispersal).
- Falco columbarius/aesalon pallidus (Sushkin, 1900)
- Asian steppes between Aral Sea and Altay Mountains. Migratory, winters in S Central Asia and N South Asia.
- Falco columbarius/aesalon insignis (Clark, 1907)
- Siberia between Yenisei and Kolyma Rivers. Migratory, winters in continental East Asia.
- Falco columbarius/aesalon lymani Bangs, 1913
- Mountains of eastern Kazakhstan and surrounding countries. Short-distance migrant.
- Falco columbarius/aesalon pacificus (Stegmann, 1929)
- Russian Far East to Sakhalin. Migratory, winters in Japan, Korea and nearby.
Ecology
Merlins inhabit fairly open country, such as willow or birch scrub, shrubland, but also taiga forest, parks, grassland such as steppe and prairies, or moorland. They are not very habitat-specific and can be found from sea level to the treeline. In general, they prefer a mix of low and medium-height vegetation with some trees, and avoid dense forests as well as treeless arid regions. During migration however, they will utilize almost any habitat.[8]
Most of its populations are migratory, wintering in warmer regions. Northern European birds move to southern Europe and North Africa, and North American populations to the southern USA to northern South America. In the milder maritime parts of its breeding range, such as Great Britain, the Pacific Northwest and western Iceland, as well as in Central Asia, it will merely desert higher ground and move to coasts and lowland during winter. The migration to the breeding grounds starts in late February, with most birds passing through the USA, Central Europe and southern Russia in March and April, and the last stragglers arriving in the breeding range towards the end of May. Migration to winter quarters at least in Eurasia peaks in August/September, while e.g. in Ohio, just south of the breeding range, F. c. columbarius is typically recorded as a southbound migrant as late as September/October.[10]
In Europe, Merlins will roost communally in winter, often with Hen Harriers (Circus cyaneus). In North America, communal roosting is rare, and Merlins are well known for fiercely attacking any birds of prey that they encounter, even adult eagles.
 F. c. columbarius hunting a Northern Blue Jay (Cyanocitta cristata bromia), Mount Auburn Cemetery, Massachusetts (USA)
F. c. columbarius hunting a Northern Blue Jay (Cyanocitta cristata bromia), Mount Auburn Cemetery, Massachusetts (USA)
Merlins rely on speed and agility to hunt their prey. They often hunt by flying fast and low, typically less than 1 metre above the ground, using trees and large shrubs to take prey by surprise. But they actually capture most prey in the air, and will "tail-chase" startled birds. Throughout its native range, the Merlin is one of the most able aerial predators of small to mid-sized birds, more versatile if anything than the larger hobbies (which prefer to attack in mid-air) and the more nimble sparrowhawks (which usually go for birds resting or sleeping in dense growth). Breeding pairs will frequently hunt cooperatively, with one bird flushing the prey toward its mate.[11]
The Merlin will readily take prey that is flushed by other causes, and can for example be seen tagging along Sharp-shinned Hawks (Accipiter striatus) to catch birds that escape from this ambush predator into the open air. It is quite unafraid, and will readily attack anything that moves conspicuously. Merlins have even been observed trying to "catch" automobiles and trains, and to feed on captive birds such as those snared in the mist nets used by ornithologists. Even under adverse conditions, one in 20 targets is usually caught, and under good conditions almost every other attack will be successful. Sometimes, Merlins cache food to eat it later.[12]
In particular during the breeding season, most of the prey are smallish birds weighing 10–40 g (0.35–1.4 oz). Almost any such species will be taken, with local preferences for whatever is most abundant—be it larks (Alaudidae), pipits (Anthus) or House Sparrows (Passer domesticus)—and inexperienced yearlings always a favorite. Smaller birds will generally avoid a hunting Merlin if possible. Even in the Cayman Islands (where it only occurs in winter), Bananaquits were noted to die of an apparent heart attack or stroke, without being physically harmed, when a Merlin went at them and they could not escape.[12]
Larger birds (e.g. sandpipers, flickers[13] and even Rock Pigeons[14] as heavy as the Merlin itself) and other animals—insects (especially dragonflies and moths), small mammals (especially bats and voles) and reptiles—complement its diet. These are more important outside the breeding season, when they can make up a considerable part of the Merlin's diet. But for example in Norway, while small birds are certainly the breeding Merlin's staple food, exceptional breeding success seems to require an abundance of Microtus voles.[8]
Corvids are the primary threat to eggs and nestlings. Adult Merlins may be preyed on by larger raptors, especially Peregrine Falcons (F. peregrinus), eagle-owls (e.g., Great Horned Owl, Bubo virginianus), and larger Accipiter hawks (e.g., Northern Goshawk, A. gentilis). In general however, carnivorous birds avoid Merlins due to their aggressiveness and agility.
Reproduction
Breeding occurs typically in May/June. Though the pairs are monogamous at least for a breeding season, extra-pair copulations have been recorded. Most nest sites have dense vegetative or rocky cover; the Merlin does not build a proper nest of its own. Most will use abandoned corvid (particularly Corvus crow and Pica magpie) or hawk nests which are in conifer or mixed tree stands. In moorland—particularly in the UK—the female will usually make a shallow scrape in dense heather to use as a nest. Others nest in crevices on cliff-faces and on the ground, and some may even use buildings.[8]
Three to six (usually 4 or 5) eggs are laid. The rusty brown eggs average at about 40 × 31.5 mm (1.6 × 1.24 in).[citation needed] The incubation period is 28 to 32 days. Incubation is performed by the female to about 90%; the male instead hunts to feed the family. Hatchlings weigh about 13 g (0.46 oz). The young fledge after another 30 days or so, and are dependent on their parents for up to 4 more weeks. Sometimes first-year Merlins (especially males) will serve as a "nest helper" for an adult pair. More than half—often all or almost all—eggs of a clutch survive to hatching, and at least two-thirds of the hatched young fledge. However, as noted above, in years with little supplementary food only one young in 3 may survive to fledging. The Merlin becomes sexually mature at one year of age and usually attempts to breed right away. The oldest wild bird known as of 2009 was recorded in its 13th winter.[15]
Relationship with humans
In medieval Europe, Merlins were popular in falconry: the Book of St. Albans listed it as " the falcon for a lady ". Today, they are still occasionally trained by falconers for hunting smaller birds, but due to conservation restrictions this is not very common any more.
John James Audubon illustrated the Merlin in the second edition of Birds of America (published in London, 1827–38) as Plate 75, under the title, "Le Petit Caporal – Falco temeratius". The image was engraved and colored by Robert Havell's London workshops. The original watercolor by Audubon was purchased by the New York History Society[verification needed], where it remains as of January 2009.
William Lewin illustrates the Merlin as Plate 22 in volume 1 of his Birds of Great Britain and their Eggs, published 1789 in London.
Status and conservation
Altogether, the Merlin is not particularly rare, and due to this and its wide range it is considered a Species of Least Concern by the IUCN. Its numbers are—except in the Asian part of its range, where the situation is less well determined—regularly censused. In about every major country it inhabits, many hundreds to many thousands are found, ranging from a "mere" 250-300 pairs in Belarus to perhaps as many as 30,000 pairs of aesalon in European Russia as determined in 1993. It is listed on CITES Appendix II and on a local level protected as other birds of prey; while some countries allow to capture Merlins, e.g. for falconry, international trade requires an export permit.[16]
By far the most serious long-term threat to these birds is habitat destruction, especially in their breeding areas. Ground-nesting populations in moorland have a preference for tall heather, and are thus susceptible to overmanagement by burning vast tracts instead of creating a habitat mosaic containing old and new growth. Still, the Merlin is rather euryoecious and will even live in settled areas, provided they have the proper mix of low and high vegetation, as well as sufficient prey (which is usually the case) and nesting sites (which is a common limiting factor).[8]
In North America, the species seems to have been more widespread in the past, or perhaps its range has shifted northwards: F. c. columbarius was an uncommon breeding bird in Ohio before the 20th century, but e.g. in Seneca County, as early as the 1900s even single adults were rarely seen in the breeding season. It is encountered in Ohio as a passage migrant and rarely as a winter guest, though two recent nestings have been confirmed. Changing land-use in Ohio mainly turned forest into agricultural land and thus is not very likely to have rendered the region inhospitable to the Merlin; global warming on the other hand cannot be dismissed as a reason, given that the Merlin is essentially a subarctic species that barely ranges even into temperate climes. Also, it may be that the number of Merlins wintering in the northern USA has increased during the 20th century.[17]
Perhaps the most frequent cause of accidental death for individuals is collision with man-made objects, particularly during attacks. This may account for almost half of all premature deaths of Merlins. In the 1960s and 1970s, organochlorine pesticides were responsible for declines—particularly in Canada—due to eggshell thinning and subsequent brood failure, and compromising the immune system of adults. This has since been remedied with restrictions on the use of DDT and similar chemicals, and numbers have rebounded. Overall, Merlin stocks appear globally stable; while they may decline temporarily in places, they will usually increase again eventually, suggesting that this phenomenon is due to the fluctuations of supplementary food stocks discussed above.[16]
Footnotes
- ^ E.g. Henninger (1906)
- ^ White (1994), Wink et al. (1998)
- ^ a b c d White (1994), White et al. (1994)
- ^ Helbig et al. (1994), White (1994), Wink et al. (1998), Griffiths (1999), Groombridge et al. (2002).
- ^ See Martin et al. (2000) for dating.
- ^ Feduccia (1970), Helbig et al. (1994), Wink et al. (1998)
- ^ Wink et al. (1998)
- ^ a b c d e f g White (1994)
- ^ Henninger (1906), OOS (2004)
- ^ White (1994), OOS (2004)
- ^ del Hoyo et al. (1994), White (1994)
- ^ a b Olson et al. (1981), White (1994)
- ^ http://www.carolinaraptorcenter.org/merlin.php
- ^ http://elibrary.unm.edu/sora/Wilson/v100n01/p0137-p0139.pdf
- ^ White (1994), AnAge [2009]
- ^ a b White (1994), BLI (2008)
- ^ Henninger (1906), White (1994), OOS (2004)
References
- AnAge [2009]: Falco columbarius life history data. Retrieved 2009-OCT-28.
- BirdLife International (BLI) (2008). Falco columbarius. In: IUCN 2008. IUCN Red List of Threatened Species. Downloaded on 28 October 2009.
- del Hoyo, Josep; Elliott, Andrew & Sargatal, Jordi (eds.): Handbook of Birds of the World (Volume 2: New World vultures to Guineafowl). Lynx Edicions, Barcelona. ISBN 84-87334-15-6
- Feduccia, J. Alan (1970): Some birds of prey from the Upper Pliocene of Kansas. Auk 87(4): 795-797. DjVu fulltext PDF fulltext
- Griffiths, Carole S. (1999): Phylogeny of the Falconidae inferred from molecular and morphological data. Auk 116(1): 116–130. DjVu fulltext PDF fulltext
- Groombridge, Jim J.; Jones, Carl G.; Bayes, Michelle K.; van Zyl, Anthony J.; Carrillo, José; Nichols, Richard A. & Bruford, Michael W. (2002): A molecular phylogeny of African kestrels with reference to divergence across the Indian Ocean. Mol. Phylogenet. Evol. 25(2): 267–277. doi:10.1016/S1055-7903(02)00254-3 PDF fulltext
- Helbig, A.J.; Seibold, I.; Bednarek, W.; Brüning, H.; Gaucher, P.; Ristow, D.; Scharlau, W.; Schmidl, D. & Wink, Michael (1994): Phylogenetic relationships among falcon species (genus Falco) according to DNA sequence variation of the cytochrome b gene. In: Meyburg, B.-U. & Chancellor, R.D. (eds.): Raptor conservation today: 593-599. PDF fulltext
- Henninger, W.F. (1906): A preliminary list of the birds of Seneca County, Ohio. Wilson Bull. 18(2): 47-60. DjVu fulltext PDF fulltext
- Martin, R.A.; Honey, J.G. & Pelaez-Campomanes, P. (2000): The Meade Basin Rodent Project; a progress report. Kansas Geologial Survey Open-file Report 2000-61[verification needed]. Paludicola 3(1): 1-32.
- Ohio Ornithological Society (OOS) (2004): Annotated Ohio state checklist. Version of April 2004. PDF fulltext
- Olson, Storrs L.; James, Helen F. & Meister, Charles A. (1981): Winter field notes and specimen weights of Cayman Island Birds. Bull. B.O.C. 101(3): 339-346. PDF fulltext
- White, Clayton M. (1994): 44. Merlin. In: del Hoyo, Josep; Elliott, Andrew & Sargatal, Jordi (eds.): Handbook of Birds of the World (Volume 2: New World vultures to Guineafowl): 267, plate 27. Lynx Edicions, Barcelona. ISBN 84-87334-15-6
- White, Clayton M.; Olsen, Penny D. & Kiff, Lloyd F. (1994): Family Falconidae. In: del Hoyo, Josep; Elliott, Andrew & Sargatal, Jordi (eds.): Handbook of Birds of the World (Volume 2: New World vultures to Guineafowl): 216-275, plates 24-28. Lynx Edicions, Barcelona. ISBN 84-87334-15-6
- Wink, Michael; Seibold, I.; Lotfikhah, F. & Bednarek, W. (1998): Molecular systematics of holarctic raptors (Order Falconiformes). In: Chancellor, R.D., Meyburg, B.-U. & Ferrero, J.J. (eds.): Holarctic Birds of Prey: 29-48. Adenex & WWGBP. PDF fulltext
External links
- Merlin Falcon Foundation A non-profit organization that researches the Coastal Forest Merlin's life history and educates scientists and citizens about this raptor's importance to the Northwest environment.
- Cornell Lab of Ornithology - Merlin
- USGS Patuxent - Merlin
- South Dakota Birds - Merlin Information and Photos
- Picture of Black Merlin (Falco columbarius suckleyi subspecies)
- Oiseaux Photos.
- Identification guide (PDF) by Javier Blasco-Zumeta
- Sounds of Merlin by Martyn Stewart
- Merlin, Canadian Peregrine Foundation
Categories:- IUCN Red List least concern species
- Falco
- Birds of Pakistan
- Birds of Europe
- Birds of Turkey
- Birds of the Faroe Islands
- British Isles coastal fauna
- Birds of Japan
- Birds of Puerto Rico
Wikimedia Foundation. 2010.









