- Fullerene
-
Buckminsterfullerene C60 (left) and carbon nanotubes (right) are two examples of structures in the fullerene family.
Part of a series of articles on
Fullerenes Carbon nanotubes
Buckminsterfullerene
Fullerene chemistry
Applications
In popular culture
Timeline
Carbon allotropesNanoparticles Quantum dots
Nanostructures
Colloidal gold
Silver nanoparticles
Iron nanoparticles
Platinum nanoparticlesSee also
Nanotechnology
A fullerene is any molecule composed entirely of carbon, in the form of a hollow sphere, ellipsoid, or tube. Spherical fullerenes are also called buckyballs, and they resemble the balls used in association football. Cylindrical ones are called carbon nanotubes or buckytubes. Fullerenes are similar in structure to graphite, which is composed of stacked graphene sheets of linked hexagonal rings; but they may also contain pentagonal (or sometimes heptagonal) rings.[1]
The first fullerene to be discovered, and the family's namesake, buckminsterfullerene (C60), was prepared in 1985 by Richard Smalley, Robert Curl, James Heath, Sean O'Brien, and Harold Kroto at Rice University. The name was an homage to Buckminster Fuller, whose geodesic domes it resembles. The structure was also identified some five years earlier by Sumio Iijima, from an electron microscope image, where it formed the core of a "bucky onion."[2] Fullerenes have since been found to occur in nature.[3] More recently, fullerenes have been detected in outer space.[4] According to astronomer Letizia Stanghellini, "It’s possible that buckyballs from outer space provided seeds for life on Earth.”[5]
The discovery of fullerenes greatly expanded the number of known carbon allotropes, which until recently were limited to graphite, diamond, and amorphous carbon such as soot and charcoal. Buckyballs and buckytubes have been the subject of intense research, both for their unique chemistry and for their technological applications, especially in materials science, electronics, and nanotechnology.
Contents
History
 The icosahedral fullerene C540, another member of the family of fullerenes.
The icosahedral fullerene C540, another member of the family of fullerenes.
The icosahedral C60H60 cage was mentioned in 1965 as a possible topological structure.[6] The existence of C60 was predicted by Eiji Osawa of Toyohashi University of Technology in 1970.[7][8] He noticed that the structure of a corannulene molecule was a subset of football shape, and he hypothesised that a full ball shape could also exist. His idea was reported in Japanese scientific journals, but did not reach Europe or the Americas.
Also in 1970, R. W. Henson (then of the Atomic Energy Research Establishment) proposed the structure and made a model of C60. Unfortunately, the evidence for this new form of carbon was very weak and was not accepted, even by his colleagues. The results were never published but were acknowledged in Carbon in 1999.[9][10]
Independently from Henson in 1973 the group of scientists from USSR directed by Prof.Bochvar made the quantum-chemical analysis of stability of C60 and calculated electronic structure of the molecule. As in the last cases the theoretical prediction wasn't accepted by scientific community. The paper was published in 1973 in Proceedings of the USSR Academy of Sciences (in Russian).[11]
In mass spectrometry, discrete peaks appeared corresponding to molecules with the exact mass of sixty or seventy or more carbon atoms. In 1985, Harold Kroto (then of the University of Sussex), James R. Heath, Sean O'Brien, Robert Curl and Richard Smalley, from Rice University, discovered C60, and shortly thereafter came to discover the fullerenes.[12] Kroto, Curl, and Smalley were awarded the 1996 Nobel Prize in Chemistry for their roles in the discovery of this class of molecules. C60 and other fullerenes were later noticed occurring outside the laboratory (e.g., in normal candle soot). By 1991, it was relatively easy to produce gram-sized samples of fullerene powder using the techniques of Donald Huffman, Wolfgang Krätschmer and Konstantinos Fostiropoulos. Fullerene purification remains a challenge to chemists and to a large extent determines fullerene prices. So-called endohedral fullerenes have ions or small molecules incorporated inside the cage atoms. Fullerene is an unusual reactant in many organic reactions such as the Bingel reaction discovered in 1993. Carbon nanotubes were recognized in 1991.[13]
Minute quantities of the fullerenes, in the form of C60, C70, C76, and C84 molecules, are produced in nature, hidden in soot and formed by lightning discharges in the atmosphere.[14] In 1992, fullerenes were found in a family of minerals known as Shungites in Karelia, Russia.[3]
In 2010, fullerenes (C60) have been discovered in a cloud of cosmic dust surrounding a distant star 6500 light years away. Using NASA's Spitzer infrared telescope the scientists spotted the molecules' unmistakable infrared signature. Sir Harry Kroto, who shared the 1996 Nobel Prize in Chemistry for the discovery of buckyballs commented: "This most exciting breakthrough provides convincing evidence that the buckyball has, as I long suspected, existed since time immemorial in the dark recesses of our galaxy." [15]
Naming
Buckminsterfullerene (C60) was named after Richard Buckminster Fuller, a noted architectural modeler who popularized the geodesic dome. Since buckminsterfullerenes have a shape similar to that sort of dome, the name was thought appropriate. As the discovery of the fullerene family came after buckminsterfullerene, the shortened name 'fullerene' is used to refer to the family of fullerenes. The suffix “ene” indicates that each C atom is covalently bonded to three others (instead of the maximum of four), a situation that classically would correspond to the existence of bonds involving two pairs of electrons (“double bonds”).
Types of fullerene
Since the discovery of fullerenes in 1985, structural variations on fullerenes have evolved well beyond the individual clusters themselves. Examples include:[16]
- buckyball clusters: smallest member is C20 (unsaturated version of dodecahedrane) and the most common is C60;
- nanotubes: hollow tubes of very small dimensions, having single or multiple walls; potential applications in electronics industry;
- megatubes: larger in diameter than nanotubes and prepared with walls of different thickness; potentially used for the transport of a variety of molecules of different sizes;[17]
- polymers: chain, two-dimensional and three-dimensional polymers are formed under high-pressure high-temperature conditions
- nano"onions": spherical particles based on multiple carbon layers surrounding a buckyball core; proposed for lubricants;[18]
- linked "ball-and-chain" dimers: two buckyballs linked by a carbon chain;[19]
- fullerene rings.[20]
Buckyballs
 C60 with isosurface of ground state electron density as calculated with DFT
C60 with isosurface of ground state electron density as calculated with DFT
Buckminsterfullerene
Main article: BuckminsterfullereneBuckminsterfullerene is the smallest fullerene molecule in which no two pentagons share an edge (which can be destabilizing, as in pentalene). It is also the most common in terms of natural occurrence, as it can often be found in soot.
The structure of C60 is a truncated (T = 3) icosahedron, which resembles an association football ball of the type made of twenty hexagons and twelve pentagons, with a carbon atom at the vertices of each polygon and a bond along each polygon edge.
The van der Waals diameter of a C60 molecule is about 1.1 nanometers (nm).[21] The nucleus to nucleus diameter of a C60 molecule is about 0.71 nm.
The C60 molecule has two bond lengths. The 6:6 ring bonds (between two hexagons) can be considered "double bonds" and are shorter than the 6:5 bonds (between a hexagon and a pentagon). Its average bond length is 1.4 angstroms.
Silicon buckyballs have been created around metal ions.
Boron buckyball
A type of buckyball which uses boron atoms, instead of the usual carbon, was predicted and described in 2007. The B80 structure, with each atom forming 5 or 6 bonds, is predicted to be more stable than the C60 buckyball.[22] One reason for this given by the researchers is that the B-80 is actually more like the original geodesic dome structure popularized by Buckminster Fuller, which uses triangles rather than hexagons. However, this work has been subject to much criticism by quantum chemists[23][24] as it was concluded that the predicted Ih symmetric structure was vibrationally unstable and the resulting cage undergoes a spontaneous symmetry break, yielding a puckered cage with rare Th symmetry (symmetry of a volleyball).[23] The number of six-member rings in this molecule is 20 and number of five-member rings is 12. There is an additional atom in the center of each six-member ring, bonded to each atom surrounding it.
Other buckyballs
Another fairly common fullerene is C70,[25] but fullerenes with 72, 76, 84 and even up to 100 carbon atoms are commonly obtained.
In mathematical terms, the structure of a fullerene is a trivalent convex polyhedron with pentagonal and hexagonal faces. In graph theory, the term fullerene refers to any 3-regular, planar graph with all faces of size 5 or 6 (including the external face). It follows from Euler's polyhedron formula, V − E + F = 2, (where V, E, F are the numbers of vertices, edges, and faces), that there are exactly 12 pentagons in a fullerene and V/2 − 10 hexagons.
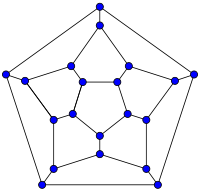
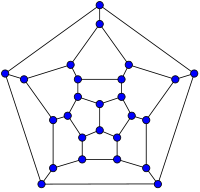
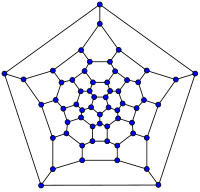
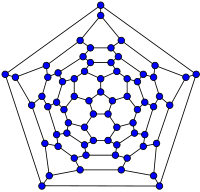
20-fullerene
(dodecahedral graph)26-fullerene graph 60-fullerene
(truncated icosahedral graph)70-fullerene graph The smallest fullerene is the dodecahedral C20. There are no fullerenes with 22 vertices.[26] The number of fullerenes C2n grows with increasing n = 12, 13, 14, ..., roughly in proportion to n9 (sequence A007894 in OEIS). For instance, there are 1812 non-isomorphic fullerenes C60. Note that only one form of C60, the buckminsterfullerene alias truncated icosahedron, has no pair of adjacent pentagons (the smallest such fullerene). To further illustrate the growth, there are 214,127,713 non-isomorphic fullerenes C200, 15,655,672 of which have no adjacent pentagons.
Trimetasphere carbon nanomaterials were discovered by researchers at Virginia Tech and licensed exclusively to Luna Innovations. This class of novel molecules comprises 80 carbon atoms (C80) forming a sphere which encloses a complex of three metal atoms and one nitrogen atom. These fullerenes encapsulate metals which puts them in the subset referred to as metallofullerenes. Trimetaspheres have the potential for use in diagnostics (as safe imaging agents), therapeutics and in organic solar cells.[citation needed]
Carbon nanotubes
 This rotating model of a carbon nanotube shows its 3D structure.Main article: Carbon nanotube
This rotating model of a carbon nanotube shows its 3D structure.Main article: Carbon nanotubeNanotubes are cylindrical fullerenes. These tubes of carbon are usually only a few nanometres wide, but they can range from less than a micrometer to several millimeters in length. They often have closed ends, but can be open-ended as well. There are also cases in which the tube reduces in diameter before closing off. Their unique molecular structure results in extraordinary macroscopic properties, including high tensile strength, high electrical conductivity, high ductility, high heat conductivity, and relative chemical inactivity (as it is cylindrical and "planar" — that is, it has no "exposed" atoms that can be easily displaced). One proposed use of carbon nanotubes is in paper batteries, developed in 2007 by researchers at Rensselaer Polytechnic Institute.[27] Another highly speculative proposed use in the field of space technologies is to produce high-tensile carbon cables required by a space elevator.
Carbon nanobuds
Main article: Carbon nanobudNanobuds have been obtained by adding buckminsterfullerenes to carbon nanotubes.
Fullerite
Main article: Aggregated diamond nanorodFullerites are the solid-state manifestation of fullerenes and related compounds and materials.
"Ultrahard fullerite" is a coined term frequently used to describe material produced by high-pressure high-temperature (HPHT) processing of fullerite. Such treatment converts fullerite into a nanocrystalline form of diamond which has been reported to exhibit remarkable mechanical properties.[28]
Properties
For the past decade, the chemical and physical properties of fullerenes have been a hot topic in the field of research and development, and are likely to continue to be for a long time. Popular Science has published articles about the possible uses of fullerenes in armor.[citation needed] In April 2003, fullerenes were under study for potential medicinal use: binding specific antibiotics to the structure to target resistant bacteria and even target certain cancer cells such as melanoma. The October 2005 issue of Chemistry & Biology contains an article describing the use of fullerenes as light-activated antimicrobial agents.[29]
In the field of nanotechnology, heat resistance and superconductivity are some of the more heavily studied properties.
A common method used to produce fullerenes is to send a large current between two nearby graphite electrodes in an inert atmosphere. The resulting carbon plasma arc between the electrodes cools into sooty residue from which many fullerenes can be isolated.
There are many calculations that have been done using ab-initio quantum methods applied to fullerenes. By DFT and TD-DFT methods one can obtain IR, Raman and UV spectra. Results of such calculations can be compared with experimental results.
Aromaticity
Researchers have been able to increase the reactivity of fullerenes by attaching active groups to their surfaces. Buckminsterfullerene does not exhibit "superaromaticity": that is, the electrons in the hexagonal rings do not delocalize over the whole molecule.
A spherical fullerene of n carbon atoms has n pi-bonding electrons, free to delocalize. These should try to delocalize over the whole molecule. The quantum mechanics of such an arrangement should be like one shell only of the well-known quantum mechanical structure of a single atom, with a stable filled shell for n = 2, 8, 18, 32, 50, 72, 98, 128, etc.; i.e. twice a perfect square number; but this series does not include 60. This 2(N + 1)2 rule (with N integer) for spherical aromaticity is the three-dimensional analogue of Hückel's rule. The 10+ cation would satisfy this rule, and should be aromatic. This has been shown to be the case using quantum chemical modelling, which showed the existence of strong diamagnetic sphere currents in the cation.[30]
As a result, C60 in water tends to pick up two more electrons and become an anion. The nC60 described below may be the result of C60 trying to form a loose metallic bond.
Chemistry
Main article: Fullerene chemistryFullerenes are stable, but not totally unreactive. The sp2-hybridized carbon atoms, which are at their energy minimum in planar graphite, must be bent to form the closed sphere or tube, which produces angle strain. The characteristic reaction of fullerenes is electrophilic addition at 6,6-double bonds, which reduces angle strain by changing sp2-hybridized carbons into sp3-hybridized ones. The change in hybridized orbitals causes the bond angles to decrease from about 120° in the sp2 orbitals to about 109.5° in the sp3 orbitals. This decrease in bond angles allows for the bonds to bend less when closing the sphere or tube, and thus, the molecule becomes more stable.
Other atoms can be trapped inside fullerenes to form inclusion compounds known as endohedral fullerenes. An unusual example is the egg shaped fullerene Tb3N@C84, which violates the isolated pentagon rule.[31] Recent evidence for a meteor impact at the end of the Permian period was found by analyzing noble gases so preserved.[32] Metallofullerene-based inoculates using the rhonditic steel process are beginning production as one of the first commercially-viable uses of buckyballs.
Solubility
Fullerenes are sparingly soluble in many solvents. Common solvents for the fullerenes include aromatics, such as toluene, and others like carbon disulfide. Solutions of pure buckminsterfullerene have a deep purple color. Solutions of C70 are a reddish brown. The higher fullerenes C76 to C84 have a variety of colors. C76 has two optical forms, while other higher fullerenes have several structural isomers. Fullerenes are the only known allotrope of carbon that can be dissolved in common solvents at room temperature.
Solvent C60 C70 1-chloronaphthalene 51 mg/mL * 1-methylnaphthalene 33 mg/mL * 1,2-dichlorobenzene 24 mg/mL 36.2 mg/mL 1,2,4-trimethylbenzene 18 mg/mL * tetrahydronaphthalene 16 mg/mL * carbon disulfide 8 mg/mL 9.875 mg/mL 1,2,3-tribromopropane 8 mg/mL * xylene 5 mg/mL 3.985 mg/mL(p-xylene) bromoform 5 mg/mL * cumene 4 mg/mL * toluene 3 mg/mL 1.406 mg/mL benzene 1.5 mg/mL 1.3 mg/mL carbon tetrachloride 0.447 mg/mL 0.121 mg/mL chloroform 0.25 mg/mL * n-hexane 0.046 mg/mL 0.013 mg/mL cyclohexane 0.035 mg/mL 0.08 mg/mL tetrahydrofuran 0.006 mg/mL * acetonitrile 0.004 mg/mL * methanol 0.000 04 mg/mL * water 1.3×10−11 mg/mL * pentane 0.004 mg/mL 0.002 mg/mL heptane * 0.047 mg/mL octane 0.025 mg/mL 0.042 mg/mL isooctane 0.026 mg/mL * decane 0.070 mg/mL 0.053 mg/mL dodecane 0.091 mg/mL 0.098 mg/mL tetradecane 0.126 mg/mL * acetone * 0.0019 mg/mL isopropanol * 0.0021 mg/mL dioxane 0.0041 mg/mL * mesitylene 0.997 mg/mL 1.472 mg/mL dichloromethane 0.254 mg/mL 0.080 mg/mL * : Solubility not measured Some fullerene structures are not soluble because they have a small band gap between the ground and excited states. These include the small fullerenes C28,[33] C36 and C50. The C72 structure is also in this class, but the endohedral version with a trapped lanthanide-group atom is soluble due to the interaction of the metal atom and the electronic states of the fullerene. Researchers had originally been puzzled by C72 being absent in fullerene plasma-generated soot extract, but found in endohedral samples. Small band gap fullerenes are highly reactive and bind to other fullerenes or to soot particles.
Solvents that are able to dissolve buckminsterfullerene (C60 and C70) are listed at left in order from highest solubility. The solubility value given is the approximate saturated concentration.[34] [35][36][37]
Solubility of C60 in some solvents shows unusual behaviour due to existence of solvate phases (analogues of crystallohydrates). For example, solubility of C60 in benzene solution shows maximum at about 313 K. Crystallization from benzene solution at temperatures below maximum results in formation of triclinic solid solvate with four benzene molecules C60·4C6H6 which is rather unstable in air. Out of solution, this structure decomposes into usual fcc C60 in few minutes' time. At temperatures above solubility maximum the solvate is not stable even when immersed in saturated solution and melts with formation of fcc C60. Crystallization at temperatures above the solubility maximum results in formation of pure fcc C60. Millimeter-sized crystals of C60 and C70 can be grown from solution both for solvates and for pure fullerenes.[38][39]Hydrated Fullerene (HyFn)
Hydrated fullerene C60HyFn is a stable, highly hydrophilic, supra-molecular complex consisting of С60 fullerene molecule enclosed into the first hydrated shell that contains 24 water molecules: C60@(H2O)24. This hydrated shell is formed as a result of donor-acceptor interaction between lone-electron pairs of oxygen, water molecules and electron-acceptor centers on the fullerene surface. Meanwhile, the water molecules which are oriented close to the fullerene surface are interconnected by a three-dimensional network of hydrogen bonds. The size of C60HyFn is 1.6–1.8 nm. The maximal concentration of С60 in the form of C60HyFn achieved by 2010 is 4 mg/mL.[40] [41][42][43]
Quantum mechanics
In 1999, researchers from the University of Vienna demonstrated that wave-particle duality applied to molecules such as fullerene.[44] One of the co-authors of this research, Julian Voss-Andreae, has since created several sculptures symbolizing wave-particle duality in fullerenes (see Fullerenes in popular culture for more detail).
Science writer Marcus Chown stated on the CBC radio show Quirks and Quarks in May 2006 that scientists are trying to make buckyballs exhibit the quantum behavior of existing in two places at once (quantum superposition).[45]
Safety and toxicity
Moussa et al. (1996-7)[46][47] studied the in vivo toxicity of C60 after intra-peritoneal administration of large doses. No evidence of toxicity was found and the mice tolerated a dose of 5 000 mg/kg of body weight (BW). Mori et al. (2006) [48] could not find toxicity in rodents for C60 and C70 mixtures after oral administration of a dose of 2 000 mg/kg BW and did not observe evidence of genotoxic or mutagenic potential in vitro. Other studies could not establish the toxicity of fullerenes: on the contrary, the work of Gharbi et al. (2005)[49] suggested that aqueous C60 suspensions failing to produce acute or subacute toxicity in rodents could also protect their livers in a dose-dependent manner against free-radical damage.
A comprehensive and recent review on fullerene toxicity is given by Kolosnjaj et al. (2007a,b, c).[50][51] These authors review the works on fullerene toxicity beginning in the early 1990s to present, and conclude that very little evidence gathered since the discovery of fullerenes indicate that C60 is toxic.
With reference to nanotubes, a recent study by Poland et al. (2008)[52] on carbon nanotubes introduced into the abdominal cavity of mice led the authors to suggest comparisons to "asbestos-like pathogenicity". It should be noted that this was not an inhalation study, though there have been several performed in the past, therefore it is premature to conclude that nanotubes should be considered to have a toxicological profile similar to asbestos. Conversely, and perhaps illustrative of how the various classes of molecules which fall under the general term fullerene cover a wide range of properties, Sayes et al. found that in vivo inhalation of C60(OH)24 and nano-C60 in rats gave no effect, whereas in comparison quartz particles produced an inflammatory response under the same conditions.[53] As stated above, nanotubes are quite different in chemical and physical properties to C60, i.e., molecular weight, shape, size, physical properties (such as solubility) all are very different, so from a toxicological standpoint, different results for C60 and nanotubes are not suggestive of any discrepancy in the findings.
When considering toxicological data, care must be taken to distinguish as necessary between what are normally referred to as fullerenes: (C60, C70, ...); fullerene derivatives: C60 or other fullerenes with covalently bonded chemical groups; fullerene complexes (e.g., water-solubilized with surfactants, such as C60-PVP; host-guest complexes, such as with cyclodextrin), where the fullerene is physically bound to another molecule; C60 nanoparticles, which are extended solid-phase aggregates of C60 crystallites; and nanotubes, which are generally much larger (in terms of molecular weight and size) molecules, and are different in shape to the spheroidal fullerenes C60 and C70, as well as having different chemical and physical properties.
The above different molecules span the range from insoluble materials in either hydrophilic or lipophilic media, to hydrophilic, lipophilic, or even amphiphilic molecules, and with other varying physical and chemical properties. Therefore any broad generalization extrapolating for example results from C60 to nanotubes or vice versa is not possible, though technically all are fullerenes, as the term is defined as a close-caged all-carbon molecule. Any extrapolation of results from one molecule to other molecules must take into account considerations based on a quantitative structural analysis relationship study (QSARS), which mostly depends on how close the molecules under consideration are in physical and chemical properties.
Superconductivity
After the synthesis of macroscopic amounts of fullerenes,[54] their physical properties could be investigated. Haddon et al.[55] found that intercalation of alkali-metal atoms in solid C60 leads to metallic behavior.[56] In 1991, it was revealed that potassium-doped C60 becomes superconducting at 18 K.[57] This was the highest transition temperature for a molecular superconductor. Since then, superconductivity has been reported in fullerene doped with various other alkali metals.[58][59] It has been shown that the superconducting transition temperature in alkaline-metal-doped fullerene increases with the unit-cell volume V.[60][61] As caesium forms the largest alkali ion, caesium-doped fullerene is an important material in this family. Recently, superconductivity at 38 K has been reported in bulk Cs3C60,[62] but only under applied pressure. The highest superconducting transition temperature of 33 K at ambient pressure is reported for Cs2RbC60.[63]
The increase of transition temperature with the unit-cell volume had been believed to be evidence for the BCS mechanism of C60 solid superconductivity, because inter C60 separation can be related to an increase in the density of states on the Fermi level, N(εF). Therefore, there have been many efforts to increase the interfullerene separation, in particular, intercalating neutral molecules into the A3C60 lattice to increase the interfullerene spacing while the valence of C60 is kept unchanged. However, this ammoniation technique has revealed a new aspect of fullerene intercalation compounds: the Mott-Hubbard transition and the correlation between the orientation/orbital order of C60 molecules and the magnetic structure.[64]
The C60 molecules compose a solid of weakly bound molecules. The fullerites are therefore molecular solids, in which the molecular properties still survive. The discrete levels of a free C60 molecule are only weakly broadened in the solid, which leads to a set of essentially nonoverlapping bands with a narrow width of about 0.5 eV.[56] For an undoped C60 solid, the 5-fold hu band is the HOMO level, and the 3-fold t1u band is the empty LUMO level, and this system is a band insulator. But when the C60 solid is doped with metal atoms, the metal atoms give electrons to the t1u band or the upper 3-fold t1g band.[65] This partial electron occupation of the band leads to sometimes metallic behavior. However, A4C60 is an insulator, although the t1u band is only partially filled and it should be a metal according to band theory.[66] This unpredicted behavior may be explained by the Jahn-Teller effect, where spontaneous deformations of high-symmetry molecules induce the splitting of degenerate levels to gain the electronic energy. The Jahn-Teller type electron-phonon interaction is strong enough in C60 solids to destroy the band picture for particular valence states.[64]
A narrow band or strongly correlated electronic system and degenerated ground states are important points to understand in explaining superconductivity in fullerene solids. When the inter-electron repulsion U is greater than the bandwidth, an insulating localized electron ground state is produced in the simple Mott-Hubbard model. This explains the absence of superconductivity at ambient pressure in caesium-doped C60 solids.[62] Electron-correlation-driven localization of the t1u electrons exceeds the critical value, leading to the Mott insulator. The application of high pressure decreases the interfullerene spacing, therefore caesium-doped C60 solids turn to metallic and superconducting.
A fully developed theory of C60 solids superconductivity is still lacking, but it has been widely accepted that strong electronic correlations and the Jahn-Teller electron-phonon coupling[67] produce local electron-pairings that show a high transition temperature close to the insulator-metal transition.[68]
Chirality
Some fullerenes (e.g. C76, C78, C80, and C84) are inherently chiral because they are D2-symmetric, and have been successfully resolved. Research efforts are ongoing to develop specific sensors for their enantiomers.
Popular culture
Main article: Fullerenes in popular cultureExamples of fullerenes in popular culture are numerous. Fullerenes appeared in fiction well before scientists took serious interest in them. In New Scientist there used to be a weekly column called "Daedalus" written by David Jones, which contained humorous descriptions of unlikely technologies. In 1966 [69] Jones suggested that it may be possible to create giant hollow carbon molecules by distorting a plane hexagonal net by the addition of impurity atoms.
On 4 September 2010, Google used an interactively rotatable fullerene C60 as the second 'o' in their logo to celebrate the 25th anniversary of the discovery of the fullerenes.[70][71]
See also
References
- ^ Fullerene, Encyclopedia Britannica on-line
- ^ Iijima, S (1980). "Direct observation of the tetrahedral bonding in graphitized carbon black by high resolution electron microscopy". Journal of Crystal Growth 50 (3): 675. Bibcode 1980JCrGr..50..675I. doi:10.1016/0022-0248(80)90013-5.
- ^ a b Buseck, P.R.; Tsipursky, S.J.; Hettich, R. (1992). "Fullerenes from the Geological Environment". Science 257 (5067): 215–7. Bibcode 1992Sci...257..215B. doi:10.1126/science.257.5067.215. PMID 17794751.
- ^ Cami, J; Bernard-Salas, J.; Peeters, E.; Malek, S. E. (2010-09-02). "Detection of C60 and C70 in a Young Planetary Nebula". Science 329 (5996): 1180–2. Bibcode 2010Sci...329.1180C. doi:10.1126/science.1192035. PMID 20651118.
- ^ Atkinson, Nancy (2010-10-27). "Buckyballs Could Be Plentiful in the Universe". Universe Today. http://www.universetoday.com/76732/buckyballs-could-be-plentiful-in-the-universe. Retrieved 2010-10-28.
- ^ Schultz, H.P. (1965). "Topological Organic Chemistry. Polyhedranes and Prismanes". Journal of Organic Chemistry 30 (5): 1361. doi:10.1021/jo01016a005.
- ^ Osawa, E. (1970). Kagaku 25: 854.
- ^ Halford, B. (9 October 2006). "The World According to Rick". Chemical & Engineering News 84 (41): 13. http://pubs.acs.org/cen/coverstory/84/8441cover.html.
- ^ Thrower, P.A. (1999). "Editorial". Carbon 37 (11): 1677. doi:10.1016/S0008-6223(99)00191-8.
- ^ Henson, R.W.. "The History of Carbon 60 or Buckminsterfullerene". http://www.solina.demon.co.uk/c60.htm. Retrieved 2010-07-04.
- ^ Bochvar, D.A.; Galpern, E.G. (1973). Dokl. Acad. Nauk SSSR 209: 610.
- ^ Kroto, H.W.; et al. (1985). "C60: Buckminsterfullerene". Nature 318 (6042): 162–163. Bibcode 1985Natur.318..162K. doi:10.1038/318162a0.
- ^ Mraz, S.J. (14 April 2005). "A new buckyball bounces into town". Machine Design. http://machinedesign.com/ContentItem/60618/Anewbuckyballbouncesintotown.aspx.
- ^ "The allotropes of carbon". Interactive Nano-Visualization in Science & Engineering Education. http://invsee.asu.edu/nmodules/Carbonmod/everywhere.html. Retrieved 2010-08-29.
- ^ Stars reveal carbon 'spaceballs' July 22, 2010
- ^ Miessler, G.L.; Tarr, D.A. (2004). Inorganic Chemistry (3rd ed.). Pearson Education. ISBN 0-13-120198-0.
- ^ Mitchel, D.R.; et al. (2001). "The Synthesis of Megatubes: New Dimensions in Carbon Materials". Inorganic Chemistry 40 (12): 2751–5. doi:10.1021/ic000551q. PMID 11375691.
- ^ Sano, N.; et al. (2001). "Synthesis of carbon 'onions' in water". Nature 414 (6863): 506–7. Bibcode 2001Natur.414..506S. doi:10.1038/35107141. PMID 11734841.
- ^ Shvartsburg, A.A. (1999). Journal of Physical Chemistry 103: 5275.
- ^ Li, Y.; et al. (2001). "Structures and stabilities of C60-rings". Chemical Physics Letters 335 (5–6): 524. Bibcode 2001CPL...335..524L. doi:10.1016/S0009-2614(01)00064-1.
- ^ Qiao, Rui; Roberts, Aaron P.; Mount, Andrew S.; Klaine, Stephen J.; Ke, Pu Chun. "Translocation of C60 and Its Derivatives Across a Lipid Bilayer". Nano Letters. http://pubs.acs.org/doi/abs/10.1021/nl062515f. Retrieved 4 September 2010.
- ^ Gonzalez Szwacki, N.; Sadrzadeh, A.; Yakobson, B. (2007). "B80 Fullerene: An Ab Initio Prediction of Geometry, Stability, and Electronic Structure". Physical Review Letters 98 (16): 166804. Bibcode 2007PhRvL..98p6804G. doi:10.1103/PhysRevLett.98.166804. PMID 17501448.
- ^ a b Gopakumar, G.; Nguyen, M.T.; Ceulemans, A. (2008). "The boron buckyball has an unexpected Th symmetry". Chemical Physics Letters 450 (4–6): 175. Bibcode 2008CPL...450..175G. doi:10.1016/j.cplett.2007.11.030.
- ^ Prasad, D.; Jemmis, E. (2008). "Stuffing Improves the Stability of Fullerenelike Boron Clusters". Physical Review Letters 100 (16): 165504. Bibcode 2008PhRvL.100p5504P. doi:10.1103/PhysRevLett.100.165504. PMID 18518216.
- ^ Locke, W. (13 October 1996). "Buckminsterfullerene: Molecule of the Month". Imperial College. http://www.bristol.ac.uk/Depts/Chemistry/MOTM/buckyball/c60a.htm. Retrieved 2010-07-04.
- ^ Meija, J. (2006). "Goldberg Variations Challenge". Analytical and Bioanalytical Chemistry 385: 6–7. doi:10.1007/s00216-006-0358-9. http://www.springer.com/cda/content/document/cda_downloaddocument.pdf?SGWID=0-0-45-275900-0.
- ^ Pushparaj, V.L.; et al. (2007). "Flexible energy storage devices based on nanocomposite paper". Proceedings of the National Academy of Sciences 104 (34): 13574–7. Bibcode 2007PNAS..10413574P. doi:10.1073/pnas.0706508104. PMC 1959422. PMID 17699622. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1959422.
- ^ Blank, V. (1998). "Ultrahard and superhard phases of fullerite C60: Comparison with diamond on hardness and wear". Diamond and Related Materials 7 (2–5): 427. doi:10.1016/S0925-9635(97)00232-X. http://nanoscan.info/wp-content/publications/article_03.pdf.
- ^ Tegos, G.; et al. (2005). "Cationic Fullerenes Are Effective and Selective Antimicrobial Photosensitizers". Chemistry & Biology 12 (10): 1127–1135. doi:10.1016/j.chembiol.2005.08.014. PMC 3071678. PMID 16242655. http://linkinghub.elsevier.com/retrieve/pii/S1074-5521(05)00270-X.
- ^ Johansson, M.P.; Jusélius, J.; Sundholm, D. (2005). "Sphere Currents of Buckminsterfullerene". Angewandte Chemie International Edition 44 (12): 1843–6. doi:10.1002/anie.200462348. PMID 15706578.
- ^ Beavers, C.M.; et al. (2006). "Tb3N@C84: An improbable, egg-shaped endohedral fullerene that violates the isolated pentagon rule". Journal of the American Chemical Society 128 (35): 11352–3. doi:10.1021/ja063636k. PMID 16939248.
- ^ Luann, B.; et al. (2007). "Impact Event at the Permian-Triassic Boundary: Evidence from Extraterrestrial Noble Gases in Fullerenes". Science 291 (5508): 1530–3. Bibcode 2001Sci...291.1530B. doi:10.1126/science.1057243. PMID 11222855.
- ^ Guo, T.; Smalley, R.E.; Scuseria, G.E. (1993). "Ab initio theoretical predictions of C28, C28H4, C28F4, (Ti at C28)H4, and M at C28 (M = Mg, Al, Si, S, Ca, Sc, Ti, Ge, Zr, and Sn)". Journal of Chemical Physics 99 (1): 352. Bibcode 1993JChPh..99..352G. doi:10.1063/1.465758.
- ^ Beck, Mihály T.; Mándi, Géza (1997). "Solubility of C60". Fullerenes, Nanotubes and Carbon Nanostructures 5 (2): 291. doi:10.1080/15363839708011993.
- ^ Bezmel'nitsyn, V.N.; Eletskii, A.V.; Okun', M.V. (1998). "Fullerenes in solutions". Physics-Uspekhi 41 (11): 1091. Bibcode 1998PhyU...41.1091B. doi:10.1070/PU1998v041n11ABEH000502.
- ^ Ruoff, R.S.; et al. (1993). "Solubility of fullerene (C60) in a variety of solvents". Journal of Physical Chemistry 97 (13): 3379. doi:10.1021/j100115a049.
- ^ N. Sivaraman; R. Dhamodaran; I. Kaliappan et. al., "SOLUBILITY OF C-70 IN ORGANIC-SOLVENTS", Fullerene Science and Technology, 1994, 2, 233.
- ^ Talyzin, A.V. (1997). "Phase Transition C60−C60*4C6H6in Liquid Benzene". Journal of Physical Chemistry B 101 (47): 9679. doi:10.1021/jp9720303.
- ^ Talyzin, A.V.; Engström, I. (1998). "C70 in Benzene, Hexane, and Toluene Solutions". Journal of Physical Chemistry B 102 (34): 6477. doi:10.1021/jp9815255.
- ^ Fullerene Hydration
- ^ Andrievsky, G; Klochkov, V.K.; Bordyuh, A.B.; Dovbeshko, G.I. (2002). "Comparative analysis of two aqueous-colloidal solutions of C60 fullerene with help of FTIR reflectance and UV–Vis spectroscopy". Chemical Physics Letters 364: 8. Bibcode 2002CPL...364....8A. doi:10.1016/S0009-2614(02)01305-2.
- ^ Andrievsky, G.; Klochkov, V.; Derevyanchenko, L. (2005). "Is the C 60 Fullerene Molecule Toxic?!". Fullerenes, Nanotubes, and Carbon Nanostructures 13 (4): 363. doi:10.1080/15363830500237267.
- ^ Andrievsky, GV; Bruskov, VI; Tykhomyrov, AA; Gudkov, SV (2009). "Peculiarities of the antioxidant and radioprotective effects of hydrated C60 fullerene nanostuctures in vitro and in vivo". Free radical biology & medicine 47 (6): 786–93. doi:10.1016/j.freeradbiomed.2009.06.016. PMID 19539750.
- ^ Arndt, M.; et al. (1999). "Wave-particle duality of C60". Nature 401 (6754): 680–2. Bibcode 1999Natur.401..680A. doi:10.1038/44348. PMID 18494170.
- ^ The radio show
- ^ Moussa, F.; et al. (1996). "Early effects of C60Administration in Swiss Mice: A Preliminary Account forIn VivoC60Toxicity". Fullerene Science and Technology 4: 21. doi:10.1080/10641229608001534.
- ^ Moussa, F.; et al. (1997). Proceedings of the Electrochemical Society 5: 332.
- ^ Mori, T.; et al. (2006). "Preclinical studies on safety of fullerene upon acute oral administration and evaluation for no mutagenesis". Toxicology 225 (1): 48–54. doi:10.1016/j.tox.2006.05.001. PMID 16782258.
- ^ Gharbi, N.; et al. (2005). "[60]fullerene is a powerful antioxidant in vivo with no acute or subacute toxicity". Nano Letters 5 (12): 2578–85. Bibcode 2005NanoL...5.2578G. doi:10.1021/nl051866b. PMID 16351219.
- ^ Kolosnjaj, J.; et al. (2007). "Toxicity studies of fullerenes and derivatives". In Chan, W.C.. Bioapplications of Nanoparticles. Landes Bioscience. pp. 168–180.
- ^ Kolosnjaj, J.; Szwarc, H.; Moussa, F. (2007). "Toxicity studies of carbon nanotubes". Advances in Experimental Medicine and Biology. Advances in Experimental Medicine and Biology 620: 181–204. doi:10.1007/978-0-387-76713-0_14. ISBN 978-0-387-76712-3. PMID 18217344.
- ^ Poland, C.A.; et al. (2008). "Carbon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study". Nature Nanotechnology 3 (7): 423–8. doi:10.1038/nnano.2008.111. PMID 18654567.
- ^ Sayes, C.M.; et al. (2007). "Comparative Pulmonary Toxicity Assessments of C60Water Suspensions in Rats: Few Differences in Fullerene Toxicity in Vivo in Contrast to in Vitro Profiles". Nano Letters 7 (8): 2399–406. Bibcode 2007NanoL...7.2399S. doi:10.1021/nl0710710. PMID 17630811.
- ^ Krätschmer, W.; et al. (1990). "Solid C60: a new form of carbon". Nature 347 (6291): 354. Bibcode 1990Natur.347..354K. doi:10.1038/347354a0.
- ^ Haddon, R.C.; et al. (1991). "Conducting films of C60 and C70 by alkali-metal doping". Nature 350 (6316): 320. Bibcode 1991Natur.350..320H. doi:10.1038/350320a0.
- ^ a b Gunnarsson, O. (1997). "Superconductivity in fullerides". Reviews of Modern Physics 69 (2): 575. arXiv:cond-mat/9611150. Bibcode 1997RvMP...69..575G. doi:10.1103/RevModPhys.69.575.
- ^ Hebard, A.F.; et al. (1991). "Superconductivity at 18 K in potassium-doped C60". Nature 350 (6319): 600. Bibcode 1991Natur.350..600H. doi:10.1038/350600a0.
- ^ Rosseinsky, M.J.; et al. (1991). "Superconductivity at 28 K in Rb_{x}C_{60}". Physical Review Letters 66: 2830. Bibcode 1991PhRvL..66.2830R. doi:10.1103/PhysRevLett.66.2830.
- ^ Chen, C.-C.; et al. (1991). "(RbxK1-x)3C60 Superconductors: Formation of a Continuous Series of Solid Solutions". Science 253 (5022): 886–8. Bibcode 1991Sci...253..886C. doi:10.1126/science.253.5022.886. PMID 17751824.
- ^ Zhou, O.; et al. (1992). "Compressibility of M3C60 Fullerene Superconductors: Relation Between Tc and Lattice Parameter". Science 255 (5046): 833–5. Bibcode 1992Sci...255..833Z. doi:10.1126/science.255.5046.833. PMID 17756430.
- ^ Brown, C.M.; et al. (1999). "Pressure dependence of superconductivity in the Na_{2}Rb_{0.5}Cs_{0.5}C_{60} fulleride". Physical Review B 59: 4439. Bibcode 1999PhRvB..59.4439B. doi:10.1103/PhysRevB.59.4439.
- ^ a b Ganin, A.Y.; et al. (2008). "Bulk superconductivity at 38 K in a molecular system". Nature 7 (5): 367–71. Bibcode 2008NatMa...7..367G. doi:10.1038/nmat2179. PMID 18425134.
- ^ Tanigaki, K.; et al. (1991). "Superconductivity at 33 K in CsxRbyC60". Nature 352 (6332): 222–223. Bibcode 1991Natur.352..222T. doi:10.1038/352222a0.
- ^ a b Iwasa, Y.; et al. (2003). "Superconductivity, Mott Hubbard states, and molecular orbital order in intercalated fullerides". Journal of Physics: Condensed Matter 15 (13): R495. Bibcode 2003JPCM...15R.495I. doi:10.1088/0953-8984/15/13/202.
- ^ Erwin, S.C.; et al. (1991). "Electronic structure of crystalline K_{6}C_{60}". Physical Review Letters 67: 1610. Bibcode 1991PhRvL..67.1610E. doi:10.1103/PhysRevLett.67.1610.
- ^ Erwin, S.C.; et al. (1993). "Electronic structure of superconducting Ba_{6}C_{60}". Physical Review B 47: 14657. arXiv:cond-mat/9301006. Bibcode 1993PhRvB..4714657E. doi:10.1103/PhysRevB.47.14657.
- ^ Han, J.E.; et al. (2003). "Strong Superconductivity with Local Jahn-Teller Phonons in C_{60} Solids". Physical Review Letters 90: 167006. Bibcode 2003PhRvL..90p7006H. doi:10.1103/PhysRevLett.90.167006. PMID 12731998.
- ^ Capone, M.; et al. (2002). "Strongly Correlated Superconductivity". Science 296 (5577): 2364–6. arXiv:cond-mat/0207058. Bibcode 2002Sci...296.2364C. doi:10.1126/science.1071122. PMID 12089436.
- ^ Jones, D. (1966). New Scientist 32: 245.
- ^ 25th anniversary of the Buckyball celebrated by interactive Google Doodle, Telegraph.co.uk 4 September 2010
- ^ Google celebrates 25 years of the 'buckyball' Guardian.co.uk, 4 September 2010
External links
- Properties of C60 fullerene
- Richard Smalley's autobiography at Nobel.se
- Sir Harry Kroto's webpage
- Simple model of Fullerene.
- Rhonditic Steel
- Introduction to fullerites
- Bucky Balls, a short video explaining the structure of C60 by the Vega Science Trust
- Giant Fullerenes, a short video looking at Giant Fullerenes
Allotropes of carbon sp3 forms sp2 forms sp forms Linear acetylenic carbonmixed sp3/sp2 forms other forms related Categories:- Fullerenes
Wikimedia Foundation. 2010.






