- Corannulene
-
Corannulene 
 Dibenzo[ghi,mno]fluorantheneOther names[5]circulene
Dibenzo[ghi,mno]fluorantheneOther names[5]circuleneIdentifiers CAS number 5821-51-2 
PubChem 11831840 ChemSpider 10006487 
Jmol-3D images Image 1
Image 2- c16ccc2ccc3ccc5c4c(c1c2c34)c(cc5)cc6
c1cc2ccc3ccc4ccc5ccc1c6c2c3c4c56
Properties Molecular formula C20H10 Molar mass 250.29 g/mol  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Corannulene is a polycyclic aromatic hydrocarbon with chemical formula C20H10.[1] The molecule consists of a cyclopentane ring fused with 5 benzene rings, so another name for it is [5]circulene. It is of scientific interest because it is a geodesic polyarene and can be considered a fragment of buckminsterfullerene. Due to this connection and also its bowl shape, corannulene is also known as a buckybowl. Corannulene exhibits a bowl-to-bowl inversion with an inversion barrier of 10.2 kcal/mol (42.7 kJ/mol) at −64 °C.[2]
Contents
Synthesis
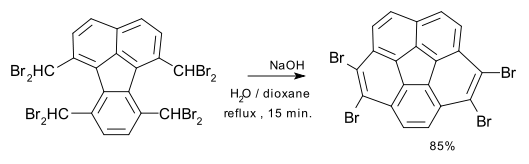
Several synthetic routes exist to corannulene. Flash vacuum pyrolysis techniques generally have lower chemical yields than solution-chemistry syntheses, but offer routes to more derivatives. Corannulane was first isolated in 1966 by multistep organic synthesis.[3] A flash vacuum pyrolysis method followed in 1991.[4] One synthesis based on solution chemistry[5] consists of a nucleophilic displacement–elimination reaction of an octabromide with potassium hydroxide:
The bromine substituents are removed with an excess of n-butyllithium.
Much effort is directed at functionalization of the corannulene ring with novel functional groups such as ethynyl groups,[2][6][7] ether groups,[8] thioether groups,[9] platinum function groups,[10] aryl groups,[11] phenalenyl fused [12] and indeno extensions.[13]
Aromaticity
The observed aromaticity for this compound is explained with a so-called annulene-within-an-annulene model. According to this model corannulene is made up of an aromatic 6 electron cyclopentadienyl anion surrounded by an aromatic 14 electron annulenyl cation. This model was suggested by Barth and Lawton in the first synthesis of corannulene in 1966.[3] They also suggested the trivial name 'corannulene', which is derived from the annulene-within-an-annulene model: core + annulene.
However, later theoretical calculations have disputed the validity of this approximation.[14][15]
Corannulene anions
Corannulene can be reduced up to a tetraanion in a series of one-electron reductions. This has been performed with alkali metals, electrochemically and with bases. The corannulene dianion is antiaromatic and tetraanion is again aromatic. With lithium as reducing agent two tetraanions form a supramolecular dimer with two bowls stacked into each other with 4 lithium ions in between and 2 pairs above and below the stack.[16]. This self-assembly motif was applied in the organization of fullerenes. Penta-substituted fullerenes (with methyl or phenyl groups) charged with five electrons form supramolecular dimers with a complementary corannulene tetraanion bowl, 'stitched' by interstitial lithium cations.[17] In a related system 5 lithium ions are sandwiched between two corannulene bowls [18]
In one cyclopenta[bc]corannulene a concave - concave aggregate is observed by NMR spectroscopy with 2 C–Li–C bonds connecting the tetraanions.[19]
Metals tend to bind to the convex face of the annulene. Concave binding has been reported for a cesium / crown ether system [20]
Corannulene cations
UV 193-nm photoionization effectively removes a π-electron from the twofold degenerate E1-HOMO located in the aromatic network of electrons yielding a corannulene radical cation.[21] Owing to the degeneracy in the HOMO orbital, the corannulene radical cation is unstable in its original C5v molecular arrangement, and therefore, subject to Jahn-Teller (JT) vibronic distortion.
Using electrospray ionization, a protonated corannulene cation has been produced in which the protonation site was observed to be on a peripheral sp2-carbon atom.[21]
Corannulene radicals
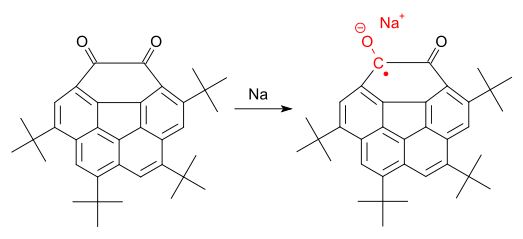
Corannulene-based free radicals have also been synthesised and studied.[22][23][24] A semiquinone radical anion obtained by reduction of the diketone by a sodium mirror (see below) has been reported stable in the solid state [25]
In this radical anion spin density is delocalized with a significant contribution from the central cyclopentadienyl radical.
Corannulene carbocations
Corannulene can react with electrophiles to form a corannulene carbocation. Reaction with chloromethane and aluminium chloride results in the formation of an AlCl4- salt with a methyl group situated at the center with the cationic center at the rim. X-ray diffraction analysis shows the that the new carbon-carbon bond is elongated (157 pm) [26]
Bicorannulenyl
Bicorannulenyl is the corannulene dimer, in which two corannulene units are connected through a single bond. The molecule's stereochemistry consists of two chiral elements: the asymmetry of a singly substituted corannulenyl, and the helical twist about the central bond. In the neutral state, bicorannulenyl exists as 12 conformers, which intercovert through multiple bowl-inversions and bond-rotations.[27] When bicorannulenyl is reduced to a dianion with potassium metal, the central bond assumes significant double-bond character. This is due to its orbital structure, which has a LUMO orbital localized on the central bond.[28] When bicorannulenyl is reduced to an octaanion with lithium metal, it self-assembles into supramolecular oligomers.[29] This is based on the "charged polyarene stacking" self-assembly motif.
Applications
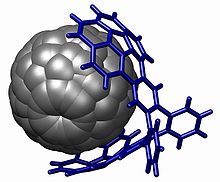
The corannulene group is used in host-guest chemistry with interactions based on pi stacking , notably with fullerenes (the buckycatcher) [30][31] but also with nitrobenzene [32]
With long aliphatic side chains corannulenes are reported forming a thermotropic hexagonal columnar liquid crystalline mesophase.[33] Corannulenes have also been used as the core group in a dendrimer [11] or as coordinating ligand to metals.[34][35][36][37][38][39][40] Corannulenes with ethynyl groups are investigated for their potential use as blue emitters.[7]
In space
Efforts to detect corannulene in space have thus far failed.[41][42]
See also
References
- ^ Scott, L. T.; Bronstein, H. E.; Preda, D. V.; Ansems, R. B. M.; Bratcher, M. S.; Hagen, S. (1999). "Geodesic polyarenes with exposed concave surfaces". Pure and Applied Chemistry 71 (2): 209. doi:10.1351/pac199971020209.
- ^ a b Scott, L. T.; Hashemi, M. M.; Bratcher, M. S. (1992). "Corannulene bowl-to-bowl inversion is rapid at room temperature". Journal of the American Chemical Society 114 (5): 1920. doi:10.1021/ja00031a079.
- ^ a b Barth, W. E.; Lawton, R. G. (1966). Journal of the American Chemical Society 88 (2): 380. doi:10.1021/ja00954a049.
- ^ Scott, L. T.; Hashemi, M. M.; Meyer, D. T.; Warren, H. B. (1991). "Corannulene. A convenient new synthesis". Journal of the American Chemical Society 113 (18): 7082. doi:10.1021/ja00018a082.
- ^ Sygula, A.; Rabideau, P. W. (2000). "A Practical, Large Scale Synthesis of the Corannulene System". Journal of the American Chemical Society 122 (26): 6323. doi:10.1021/ja0011461.
- ^ Wu, Y.; Bandera, D.; Maag, R.; Linden, A.; Baldridge, K.; Siegel, J. (2008). "Multiethynyl corannulenes: synthesis, structure, and properties". Journal of the American Chemical Society 130 (32): 10729–10739. doi:10.1021/ja802334n. PMID 18642812.
- ^ a b Mack, J.; Vogel, P.; Jones, D.; Kaval, N.; Sutton, A. (2007). "The development of corannulene-based blue emitters". Organic & biomolecular chemistry 5 (15): 2448–2452. doi:10.1039/b705621d. PMID 17637965.
- ^ Gershoni-Poranne, R.; Pappo, D.; Solel, E.; Keinan, E. (2009). "Corannulene ethers via Ullmann condensation". Organic letters 11 (22): 5146–5149. doi:10.1021/ol902352k. PMID 19905024.
- ^ Baldridge, K.; Hardcastle, K.; Seiders, T.; Siegel, J. (2010). "Synthesis, structure and properties of decakis(phenylthio)corannulene". Organic & biomolecular chemistry 8 (1): 53–55. doi:10.1039/b919616a. PMID 20024131.
- ^ Choi, H.; Kim, C.; Park, K. M.; Kim, J.; Kang, Y.; Ko, J. (2009). "Synthesis and structure of penta-platinum σ-bonded derivatives of corannulene". Journal of Organometallic Chemistry 694 (22): 3529. doi:10.1016/j.jorganchem.2009.07.015.
- ^ a b Pappo, D.; Mejuch, T.; Reany, O.; Solel, E.; Gurram, M.; Keinan, E. (2009). "Diverse Functionalization of Corannulene: Easy Access to Pentagonal Superstructure". Organic letters 11 (5): 1063. doi:10.1021/ol8028127. PMID 19193048.
- ^ Nishida, S.; Morita, Y.; Ueda, A.; Kobayashi, T.; Fukui, K.; Ogasawara, K.; Sato, K.; Takui, T. et al. (2008). "Curve-structured phenalenyl chemistry: synthesis, electronic structure, and bowl-inversion barrier of a phenalenyl-fused corannulene anion". Journal of the American Chemical Society 130 (45): 14954–14955. doi:10.1021/ja806708j. PMID 18937470.
- ^ Steinberg, B.; Jackson, E.; Filatov, A.; Wakamiya, A.; Petrukhina, M.; Scott, L. (2009). "Aromatic pi-systems more curved than C(60). The complete family of all indenocorannulenes synthesized by iterative microwave-assisted intramolecular arylations". Journal of the American Chemical Society 131 (30): 10537–10545. doi:10.1021/ja9031852. PMID 19722628.
- ^ Sygula, A. (1995). "Structure and inversion barriers of corannulene, its dianion and tetraanion. An ab initio study". Journal of Molecular Structure: THEOCHEM 333: 215–226. doi:10.1016/0166-1280(94)03961-J.
- ^ Monaco, G.; Scott, L.; Zanasi, R. (2008). "Magnetic euripi in corannulene". The journal of physical chemistry. A 112 (35): 8136–8147. doi:10.1021/jp8038779. PMID 18693706.
- ^ Ayalon, A.; Sygula, A.; Cheng, P.; Rabinovitz, M.; Rabideau, P.; Scott, L. (1994). "Stable High-Order Molecular Sandwiches: Hydrocarbon Polyanion Pairs with Multiple Lithium Ions Inside and out". Science 265 (5175): 1065–1067. Bibcode 1994Sci...265.1065A. doi:10.1126/science.265.5175.1065. PMID 17832895.
- ^ Aprahamian, I.; Eisenberg, D.; Hoffman, R.; Sternfeld, T.; Matsuo, Y.; Jackson, E.; Nakamura, E.; Scott, L. et al. (2005). "Ball-and-socket stacking of supercharged geodesic polyarenes: bonding by interstitial lithium ions". Journal of the American Chemical Society 127 (26): 9581–9587. doi:10.1021/ja0515102. PMID 15984885.
- ^ A Main Group Metal Sandwich: Five Lithium Cations Jammed Between Two Corannulene Tetraanion Decks Zabula, et al. Science 19 August 2011: 1008-1011. doi:10.1126/science.1208686
- ^ Aprahamian, I.; Preda, D.; Bancu, M.; Belanger, A.; Sheradsky, T.; Scott, L.; Rabinovitz, M. (2006). "Reduction of bowl-shaped hydrocarbons: dianions and tetraanions of annelated corannulenes". The Journal of organic chemistry 71 (1): 290–298. doi:10.1021/jo051949c. PMID 16388648.
- ^ Spisak, S. N., Zabula, A. V., Filatov, A. S., Rogachev, A. Y. and Petrukhina, M. A. (2011), Selective Endo and Exo Binding of Alkali Metals to Corannulene. Angewandte Chemie International Edition, 50: 8090–8094. doi:10.1002/anie.201103028
- ^ a b Galué, Héctor Alvaro; Rice, Corey A., Steill, Jeffrey D., Oomens, Jos (1 January 2011). "Infrared spectroscopy of ionized corannulene in the gas phase". The Journal of Chemical Physics 134 (5): 054310. doi:10.1063/1.3540661.
- ^ The First Bowl-Shaped Stable Neutral Radical with a Corannulene System: Synthesis and Characterization of the Electronic Structure Yasushi Morita, Shinsuke Nishida, Tadahiro Kobayashi, Kozo Fukui, Kazunobu Sato, Daisuke Shiomi, Takeji Takui, Kazuhiro Nakasuji Organic Letters 2004 6 (9), 1397-1400 doi: 10.1021/ol0497786
- ^ Morita, Y.; Ueda, A.; Nishida, S.; Fukui, K.; Ise, T.; Shiomi, D.; Sato, K.; Takui, T. et al. (2008). "Curved aromaticity of a corannulene-based neutral radical: crystal structure and 3 d unbalanced delocalization of spin". Angewandte Chemie (International ed. in English) 47 (11): 2035–2038. doi:10.1002/anie.200704752. PMID 18256997.
- ^ Ueda, A.; Nishida, S.; Fukui, K.; Ise, T.; Shiomi, D.; Sato, K.; Takui, T.; Nakasuji, K. et al. (2010). "Three-Dimensional Intramolecular Exchange Interaction in a Curved and Nonalternant pi-Conjugated System: Corannulene with Two Phenoxyl Radicals.". Angewandte Chemie (International ed. in English) 49 (9): 1678–1682. doi:10.1002/anie.200906666. PMID 20108294.
- ^ Ueda, A., Ogasawara, K., Nishida, S., Ise, T., Yoshino, T., Nakazawa, S., Sato, K., Takui, T., Nakasuji, K. and Morita, Y. (2010), A Bowl-Shaped ortho-Semiquinone Radical Anion: Quantitative Evaluation of the Dynamic Behavior of Structural and Electronic Features. Angewandte Chemie International Edition, 49: 6333–6337. doi:10.1002/anie.201002626
- ^ Zabula, A. V., Spisak, S. N., Filatov, A. S., Rogachev, A. Y. and Petrukhina, M. A. (2011), A Strain-Releasing Trap for Highly Reactive Electrophiles: Structural Characterization of Bowl-Shaped Arenium Carbocations. Angewandte Chemie International Edition, 50: 2971–2974. doi:10.1002/anie.201007762
- ^ Eisenberg, D.; Filatov, A.; Jackson, E.; Rabinovitz, M.; Petrukhina, M.; Scott, L.; Shenhar, R. (2008). "Bicorannulenyl: stereochemistry of a C40H18 biaryl composed of two chiral bowls". The Journal of organic chemistry 73 (16): 6073–6078. doi:10.1021/jo800359z. PMID 18505292.
- ^ Eisenberg, D., Quimby, J. M., Jackson, E. A., Scott, L. T. and Shenhar, R. (2010), The Bicorannulenyl Dianion: A Charged Overcrowded Ethylene. Angewandte Chemie International Edition, 49: 7538–7542. doi:10.1002/anie.201002515
- ^ Eisenberg, D., Quimby, J. M., Jackson, E. A., Scott, L. T. and Shenhar, R. (2010), Highly Charged Supramolecular Oligomers Based on the Dimerization of Corannulene Tetraanion. Chemical Communications, 46: 9010–9012. doi:10.1039/c0cc03965a
- ^ Sygula, A.; Fronczek, F.; Sygula, R.; Rabideau, P.; Olmstead, M. (2007). "A double concave hydrocarbon buckycatcher". Journal of the American Chemical Society 129 (13): 3842–3843. doi:10.1021/ja070616p. PMID 17348661.
- ^ Wong, B. M. (2009). "Noncovalent interactions in supramolecular complexes: a study on corannulene and the double concave buckycatcher". Journal of computational chemistry 30 (1): 51–56. doi:10.1002/jcc.21022. PMID 18504779.
- ^ Kobryn, L.; Henry, W. P.; Fronczek, F. R.; Sygula, R.; Sygula, A. (2009). "Molecular clips and tweezers with corannulene pincers". Tetrahedron Letters 50 (51): 7124. doi:10.1016/j.tetlet.2009.09.177.
- ^ Miyajima, D.; Tashiro, K.; Araoka, F.; Takezoe, H.; Kim, J.; Kato, K.; Takata, M.; Aida, T. (2009). "Liquid crystalline corannulene responsive to electric field". Journal of the American Chemical Society 131 (1): 44–45. doi:10.1021/ja808396b. PMID 19128171.
- ^ Hexahapto Metal Coordination to Curved Polyaromatic Hydrocarbon Surfaces: The First Transition Metal Corannulene Complex T. Jon Seiders, Kim K. Baldridge, Joseph M. O'Connor, and Jay S. Siegel J. Am. Chem. Soc., 1997, 119 (20), pp 4781–4782 doi:10.1021/ja964380t
- ^ d8 Rhodium and Iridium Complexes of Corannulene Jay S. Siegel, Kim K. Baldridge, Anthony Linden, and Reto Dorta J. Am. Chem. Soc., 2006, 128 (33), pp 10644–10645 doi:10.1021/ja062110x
- ^
- ^ Zhu, B.; Ellern, A.; Sygula, A.; Sygula, R.; Angelici, R. J. (2007). "η6-Coordination of the Curved Carbon Surface of Corannulene (C20H10) to (η6-arene)M2+(M = Ru, Os)". Organometallics 26 (7): 1721. doi:10.1021/om0610795.
- ^ Petrukhina, M. A.; Sevryugina, Y.; Rogachev, A. Y.; Jackson, E. A.; Scott, L. T. (2006). "Corannulene: A Preference forexo-Metal Binding. X-ray Structural Characterization of [Ru2(O2CCF3)2(CO)4·(η2-C20H10)2]". Organometallics 25 (22): 5492. doi:10.1021/om060350f.
- ^ Siegel, J.; Baldridge, K.; Linden, A.; Dorta, R. (2006). "D8 rhodium and iridium complexes of corannulene". Journal of the American Chemical Society 128 (33): 10644–10645. doi:10.1021/ja062110x. PMID 16910635.
- ^ Bandera, D., Baldridge, K. K., Linden, A., Dorta, R. and Siegel, J. S. (2011), Stereoselective Coordination of C5-Symmetric Corannulene Derivatives with an Enantiomerically Pure [RhI(nbd*)] Metal Complex. Angewandte Chemie International Edition, 50: 865–867. doi: 10.1002/anie.201006877
- ^ Pilleri, P.; Herberth, D.; Giesen, T. F.; Gerin, M.; Joblin, C.; Mulas, G.; Malloci, G.; Grabow, J. -U. et al. (2009). "Search for corannulene (C20H10) in the Red Rectangle". Monthly Notices of the Royal Astronomical Society 397 (2): 1053. Bibcode 2009MNRAS.397.1053P. doi:10.1111/j.1365-2966.2009.15067.x.
- ^ Rouillé, G.; Jäger, C.; Steglich, M.; Huisken, F.; Henning, T.; Theumer, G.; Bauer, I.; Knölker, H. (2008). "IR, Raman, and UV/Vis spectra of corannulene for use in possible interstellar identification". Chemphyschem : a European journal of chemical physics and physical chemistry 9 (14): 2085–2091. doi:10.1002/cphc.200800387. PMID 18798213.
2 rings 3 rings 4 rings 5 rings 6+ rings Anthanthrene · Benzo[ghi]perylene · Corannulene · Coronene · Dicoronylene · Diindenoperylene · Helicene · Heptacene · Hexacene · Kekulene · Ovalene · ZethreneCategories:- Geodesic polyarenes
- Polycyclic aromatic hydrocarbons
- c16ccc2ccc3ccc5c4c(c1c2c34)c(cc5)cc6
Wikimedia Foundation. 2010.

![Cyclopenta[bc]corannulene](/pictures/enwiki/49/100px-Cyclopenta-bc-corannulene.png)