- Dodecahedrane
-
Dodecahedrane 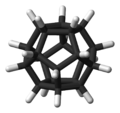
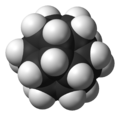 [5]fullerane-C20-Ih
[5]fullerane-C20-IhIdentifiers CAS number 4493-23-6 PubChem 123218 ChemSpider 109833 
ChEBI CHEBI:33013 
Jmol-3D images Image 1
Image 2- C12C3C4C5C1C6C7C2C8C3C9C4C1C5C6C2C7C8C9C12
C31C%10C2C5C%11C6C8C(C1C9C4C7C(C2C34)C5C6C7C89)C%10%11
Properties Molecular formula C20H20 Molar mass 260.37 g mol−1 Related compounds Related hydrocarbons Cubane
Tetrahedrane
Pagodane (an isomer of dodecahedrane)
Prismane (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dodecahedrane is a chemical compound (C20H20) first synthesised by Leo Paquette of Ohio State University in 1982, primarily for the "aesthetically pleasing symmetry of the dodecahedral framework".[1][2]
In this molecule,[3] each vertex is a carbon atom that bonds to three neighbouring carbon atoms. The 108° angle of each regular pentagon is close to the ideal bond angle of 109.5° for an sp3 hybridised atom. Each carbon atom is bonded to a hydrogen atom as well. The molecule, like fullerene, has Ih symmetry, evidenced by its proton NMR spectrum in which all hydrogen atoms appear at a single chemical shift of 3.38 ppm. Dodecahedrane is one of the platonic hydrocarbons, the others being cubane and tetrahedrane, and does not occur in nature.
Contents
Total synthesis
For over 30 years several research groups have actively pursued the total synthesis of dodecahedrane. A review article published in 1978 just dealt with the different strategies that existed up to then.[4] The first attempt was initiated in 1964 by R.B. Woodward with the synthesis of the compound triquinacene which was thought to be able to simply dimerize to dodecahedrane. Other groups joined in the race, for example that of Philip Eaton and Paul von Ragué Schleyer
Paquette's 1982 organic synthesis takes about 29 steps with raw materials cyclopentadiene (2 equivalents 10 carbon atoms), dimethyl acetylenedicarboxylate (4 carbon atoms) and allyltrimethylsilane (2 equivalents, 6 carbon atoms).
In the first leg of the procedure [5] two molecules of cyclopentadiene 1 are coupled together by reaction with elemental sodium (forming the cyclopentadienyl complex) and iodine to dihydrofulvalene 2. Next up is a tandem Diels-Alder reaction with dimethyl acetylenedicarboxylate 3 with desired sequence pentadiene-acetylene-pentadiene as in symmetrical adduct 4. An equal amount of asymmetric pentadiene-pentadiene-acetylene compound (4b) is formed and discarded.
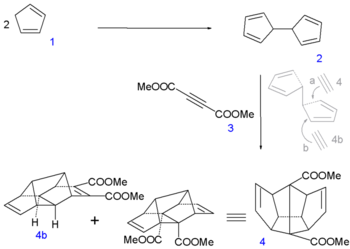
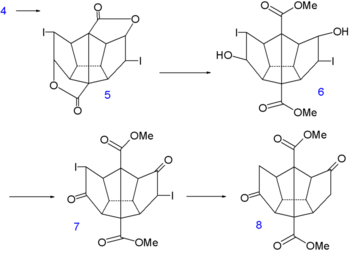
Dodecahedrane synthesis part I Dodecahedrane synthesis part II In the next step of the sequence [6] iodine is temporarily introduced via an iodolactonization of the diacid of 4 to dilactone 5. The ester group is cleaved next by methanol to the halohydrin 6, the alcohol groups converted to ketone groups in 7 by Jones oxidation and the iodine groups reduced by a zinc-copper couple in 8.
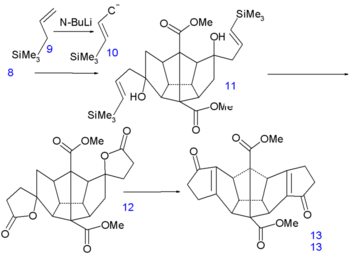
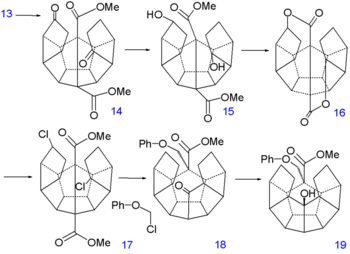
Dodecahedrane synthesis part III Dodecahedrane synthesis part IV The final 6 carbon atoms are inserted in a nucleophilic addition to the ketone groups of the carbanion 10 generated from allyltrimethylsilane 9 and n-butyllithium. In the next step the vinyl silane 11 reacts with peracetic acid in acetic acid in a radical substitution to the dilactone 12 followed by an intramolecular Friedel-Crafts alkylation with phosphorus pentoxide to diketone 13. This molecule contains all required 20 carbon atoms and is also symmetrical which facilitates the construction of the remaining 5 carbon-carbon bonds.
Reduction of the double bonds in 13 to 14 is accomplished with hydrogenation with palladium on carbon and that of the ketone groups to alcohol groups in 15 by sodium borohydride. Replacement of hydroxyl by chlorine in 17 via nucleophilic aliphatic substitution takes place through the dilactone 16 (tosyl chloride). The first C-C bond forming reaction is a kind of Birch alkylation (lithium, ammonia) with the immediate reaction product trapped with chloromethyl phenyl ether,[2] the other chlorine atom in 17 is simply reduced. This temporary appendix will in a later stage prevent unwanted enolization. The newly formed ketone group then forms another C-C bond by photochemical Norrish reaction to 19 whose alcohol group is induced to eliminate with TsOH to alkene 20.
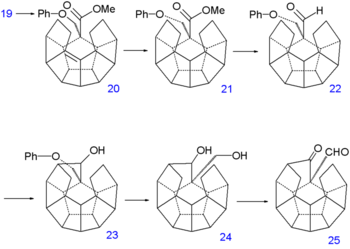
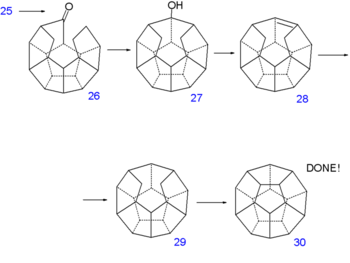
Dodecahedrane synthesis part V Dodecahedrane synthesis part VI The double bond is reduced with hydrazine and sequential diisobutylaluminum hydride reduction and pyridinium chlorochromate oxidation of 21 forms the aldehyde 22. A second Norrish reaction then adds another C-C bond to alcohol 23 and having served its purpose the phenoxy tail is removed in several steps: a Birch reduction to diol 24, oxidation with pyridinium chlorochromate to ketoaldehyde 25 and a reverse Claisen condensation to ketone 26. A third Norrish reaction produces alcohol 27 and a second dehydration 28 and another reduction 29 at which point the synthesis is left completely without functional groups. The missing C-C bond is put in place by hydrogen pressurized dehydrogenation with palladium on carbon at 250°C to dodecahedrane 30.
Synthesis from pagodane
In 1987 an alternative synthesis route was found by W.-D. Fessner and others, through the isomer pagodane.[7][8]
The world's smallest helium balloons
By shooting helium ions (He+) at a film of C20H20, Cross, Saunders and Prinsbach managed to obtain a few micrograms of He@C20H20 — dodecahedrane with a helium atom trapped inside each molecule. This substance was described as quite stable, and the authors claimed to have produced the world's smallest helium balloons.[9]
Derivatives
A variety of dodecahedrane derivatives have been synthesized and reported in the literature. Substitution of all 20 hydrogens by fluorine atoms yields the relatively unstable perfluorododecahedrane C20F20, which was obtained in milligram quantities by Wahl and others (2006).[10] Trace amounts of the analogous perchlorododecahedrane C20Cl20 were obtained, among other partially chlorinated derivatives, by reacting C20H20 dissolved in liquid chlorine under pressure at about 140 °C and under intense light for five days. Complete replacement by heavier halogens seems increasingly difficult due to their larger size. Half or more of the hydrogen atoms can be substituted by hydroxyl groups to yield polyols, but the extreme compound C20(OH)20 remained elusive as of 2006.[10]
References
- ^ Dodecahedrane Robert J. Ternansky, Douglas W. Balogh, and Leo A. Paquette J. Am. Chem. Soc.; 1982; 104(16) pp 4503 - 4504; doi:10.1021/ja00380a040
- ^ a b Leo A. Paquette, Robert J. Ternansky, Douglas W. Balogh, and Gary Kentgen (1983). "Total synthesis of dodecahedrane". Journal of the American Chemical Society 105 (16): 5446–5450. doi:10.1021/ja00354a043.
- ^ IUPAC name: hexadecahydro-5,2,1,6,3,4-[2,3]butanediyl[1,4]diylidenedipentaleno[2,1,6-cde:2',1',6'-gha]pentalene CAS number: 4493-23-6
- ^ Towards dodecahedrane Tetrahedron, Volume 35, Issue 19, 1979, Pages 2189-2223 Philip E. Eaton doi:10.1016/0040-4020(79)80114-3
- ^ Domino Diels-Alder reactions. I. Applications to the rapid construction of polyfused cyclopentanoid systems Leo A. Paquette and Matthew J. Wyvratt J. Am. Chem. Soc.; 1974; 96(14) pp 4671 - 4673; doi:10.1021/ja00821a052
- ^ Topologically spherical molecules. Synthesis of a pair of C2-symmetric hexaquinane dilactones and insights into their chemical reactivity. An efficient .pi.-mediated 1,6-dicarbonyl reduction Leo A. Paquette, Matthew J. Wyvratt, Otto Schallner, Jean L. Muthard, William J. Begley, Robert M. Blankenship, and Douglas Balogh J. Org. Chem.; 1979; 44(21) pp 3616 - 3630; doi: 10.1021/jo01335a003
- ^ Wolf-Dieter Fessner, Bulusu A. R. C. Murty, Horst Prinzbach (1987), The Pagodane Route to Dodecahedranes - Thermal, Reductive, and Oxidative Transformations of Pagodanes Angewandte Chemie International Edition in English Volume 26, Issue 5, Pages 451-452 doi:10.1002/anie.198704511
- ^ Wolf-Dieter Fessner, Bulusu A. R. C. Murty, Jürgen Wörth, Dieter Hunkler, Hans Fritz, Horst Prinzbach, Wolfgang D. Roth, Paul von Ragué Schleyer, Alan B. McEwen, Wilhelm F. Maier (1987), Dodecahedranes from [1.1.1.1]Pagodanes. Angewandte Chemie International Edition in English, Volume 26, Issue 5, Pages 452-454 doi:10.1002/anie.198704521
- ^ R. James Cross, Martin Saunders, and Horst Prinzbach (1999): Putting Helium Inside Dodecahedrane. Org. Lett. 1 (9), pp 1479–1481. doi: 10.1021/ol991037v
- ^ a b Fabian Wahl, Andreas Weiler, Peter Landenberger, Emmerich Sackers, Torsten Voss, Alois Haas, Max Lieb, Dieter Hunkler, Jürgen Wörth, Lothar Knothe, and Horst Prinzbach (2006) Towards Perfunctionalized Dodecahedranes—En Route to C20 Fullerene. Chemistry - A European Journal, volume 12, pages 6255 – 6267. doi:10.1002/chem.200501618
External links
Categories:- Hydrocarbons
- Total synthesis
- C12C3C4C5C1C6C7C2C8C3C9C4C1C5C6C2C7C8C9C12
Wikimedia Foundation. 2010.
