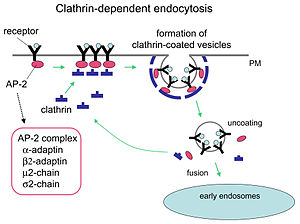
- Clathrin
-
clathrin, light polypeptide (Lca) Identifiers Symbol CLTA Entrez 1211 HUGO 2090 OMIM 118960 RefSeq NM_007096 UniProt P09496 Other data Locus Chr. 12 q23-q24 clathrin, light polypeptide (Lcb) Identifiers Symbol CLTB Entrez 1212 HUGO 2091 OMIM 118970 RefSeq NM_001834 UniProt P09497 Other data Locus Chr. 4 q Clathrin light chain Identifiers Symbol Clathrin_lg_ch Pfam PF01086 InterPro IPR000996 PROSITE PDOC00196 Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary clathrin, heavy polypeptide (Hc) Identifiers Symbol CLTC Alt. symbols CLTCL2 Entrez 1213 HUGO 2092 OMIM 118955 RefSeq NM_004859 UniProt Q00610 Other data Locus Chr. 17 q11-qter clathrin, heavy polypeptide-like 1 Identifiers Symbol CLTCL1 Alt. symbols CLTCL Entrez 8218 HUGO 2093 OMIM 601273 RefSeq NM_001835 UniProt P53675 Other data Locus Chr. 22 q11.2 Clathrin propeller repeat 
clathrin terminal domain complexed with tlpwdlwtt Identifiers Symbol Clathrin_propel Pfam PF01394 Pfam clan CL0020 InterPro IPR022365 SCOP 1bpo Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary Clathrin heavy-chain linker 
clathrin terminal domain complexed with tlpwdlwtt Identifiers Symbol Clathrin-link Pfam PF09268 Pfam clan CL0020 InterPro IPR015348 SCOP 1utc Available protein structures: Pfam structures PDB RCSB PDB; PDBe PDBsum structure summary Clathrin is a protein that plays a major role in the formation of coated vesicles. Clathrin was first isolated and named by Barbara Pearse in 1975.[1] It forms a triskelion shape composed of three clathrin heavy chains and three light chains. When the triskelia interact they form a polyhedral lattice that surrounds the vesicle. Coat-proteins, like clathrin, are used to build small vesicles in order to safely transport molecules between cells. The endocytosis and exocytosis of vesicles allows cells to transfer nutrients, to import signaling receptors, to mediate an immune response after sampling the extracellular world, and to clean up the cell debris left by tissue inflammation. On occasion, this mechanism also provides a pathway for raiding pathogens or toxins.
Contents
Structure
Clathrin coat structureThe clathrin triskelion is composed of three clathrin heavy chains and three light chains interacting at their C-termini. The three heavy chains provide the structural backbone of the clathrin lattice, and the three light chains are thought to regulate the formation and disassembly of a clathrin lattice. Clathrin heavy chain is, in concept, broken down into multiple subdomains, starting with the N-terminal domain, followed by the ankle, distal leg, knee, proximal leg, and trimerization domains. The N-terminal domain consists of a seven-bladed β-propeller structure. The other domains form a super-helix of short alpha helices. This was originally determined from the structure of the proximal leg domain that identified and is composed of a smaller structural module referred to as clathrin heavy chain repeat motifs. The light chains bind primarily to the proximal leg portion of the heavy chain with some interaction near the trimerization domain. When triskelia assemble together in solution, they can interact with enough flexibility to form 6-sided rings that yield a flatter lattice, or 5-sided rings that are necessary for curved lattice formation. When many triskelions connect, they can form a basket-like structure.
The structure shown above, is built of 36 triskelia, one of which is highlighted in green. When triskelia snap together in solution, they can interact with enough flexibility to form either 6-sided rings that yield a flatter surface or 5-sided rings with higher curvature. In a cell, a triskelion floating in the cytoplasm binds to an adaptor protein (shown on the next page), linking one of its three feet to the membrane at a time. This triskelion will bind to other membrane-attached triskelia to form a rounded lattice of hexagons and pentagons, reminiscent of the panels on a soccer ball, that pulls the membrane into a bud. By constructing different combinations of 5-sided and 6-sided rings, vesicles of different sizes may assemble. The structure shown here represents the second-smallest possible cage structure, which is actually too small to contain a functional vesicle. It was created in the laboratory by reconstituting triskelions without a lipid vesicle. The smallest clathrin cage commonly photographed, called a mini-coat, has 12 pentagons and only two hexagons. Even smaller cages with zero hexagons probably do not form from the native protein, because the feet of the triskelia are too bulky.
Function
Like many proteins, clathrin represents a perfect case of form following function; it performs critical roles in shaping rounded vesicles in the cytoplasm for intracellular trafficking. Clathrin-coated vesicles (CCV) selectively sort cargo at the cell membrane, trans-Golgi network, and endosomal compartments for multiple membrane traffic pathways. After a vesicle buds into the cytoplasm, the coat rapidly disassembles, allowing the clathrin to recycle while the vesicle gets transported to a variety of locations. Adaptor molecules are responsible for self-assembly and recruitment. Two examples of adaptor proteins are AP180[2] and epsin.[3][4][5] AP180 is used in synaptic vesicle formation. It recruits clathrin to membranes and also promotes its polymerization. Epsin also recruits clathrin to membranes and promotes its polymerization, and can help deform the membrane, and thus clathrin-coated vesicles can bud. In a cell, a triskelion floating in the cytoplasm binds to an adaptor protein, linking one of its feet to the membrane at a time. The skelion will bind to other ones attached to the membrane to form a polyhedral lattice, skelion, which pulls the membrane into a bud. The skelion does not bind directly to the membrane, but binds to the adaptor proteins that recognize the molecules on the membrane surface.
Clathrin has another function aside from the coating of organelles. In non-dividing cells, the formation of clathrin-coated vesicles occurs continuously. Formation of clathrin-coated vesicles is shut down in cells undergoing mitosis. During mitosis, clathrin binds to the spindle apparatus. Clathrin aids in the congression of chromosomes by stabilizing fibres of the mitotic spindle. Clathrin is bound directly through the amino-terminal domain of the clathrin heavy chain. During mitosis the clathrin binds directly to the microtubules or microtubule-associated proteins. The stabilization of kinetochore fibres requires the trimetric structure of clathrin in order to strengthen the spindle fibres.[6]
Clathrin-mediated endocytosis (CME) regulates many cellular physiological processes such as the internalization of growth factors and receptors, entry of pathogens, and synaptic transmission. It is believed that cellular invaders use the nutrient pathway to gain access to a cell's replicating mechanisms. Certain signalling molecules open the nutrients pathway. Two chemical compounds called Pitstop 1 and Pitstop 2, selective clathrin inhibitors, can interfere with the pathogenic activity, and thus protect the cells against invasion. These two compounds selectively block the endocytic ligand association with the clathrin terminal domain. [7]
See also
References
- ^ Pearse BM (April 1976). "Clathrin: a unique protein associated with intracellular transfer of membrane by coated vesicles". Proceedings of the National Academy of Sciences of the United States of America 73 (4): 1255–9. doi:10.1073/pnas.73.4.1255. PMC 430241. PMID 1063406. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=430241.
- ^ McMahon HT. "Clathrin and its interactions with AP180.". MRC Laboratory of Molecular Biology. http://www.endocytosis.org/AP180/Clathrin.html. Retrieved 2009-04-17. "micrographs of clathrin assembly"
- ^ McMahon HT. "Epsin 1 EM gallery". MRC Laboratory of Molecular Biology,. http://www.endocytosis.org/epsin/EM/MonolayerEMs.html. Retrieved 2009-04-17. "micrographs of vesicle budding"
- ^ Ford MG, Pearse BM, Higgins MK, Vallis Y, Owen DJ, Gibson A, Hopkins CR, Evans PR, McMahon HT (February 2001). "Simultaneous binding of PtdIns(4,5)P2 and clathrin by AP180 in the nucleation of clathrin lattices on membranes". Science 291 (5506): 1051–5. doi:10.1126/science.291.5506.1051. PMID 11161218. http://www.endocytosis.org/epsin/EM/ford.pdf.
- ^ Higgins MK, McMahon HT (May 2002). "Snap-shots of clathrin-mediated endocytosis". Trends in Biochemical Sciences 27 (5): 257–63. doi:10.1016/S0968-0004(02)02089-3. PMID 12076538. http://www.endocytosis.org/epsin/EM/mcmahon.pdf.
- ^ Royle SJ, Bright NA, Lagnado L (April 2005). "Clathrin is required for the function of the mitotic spindle". Nature 434 (7037): 1152–1157. doi:10.1038/nature03502. PMID 15858577. http://www.nature.com/nature/journal/v434/n7037/full/nature03502.html.
- ^ Role of the Clathrin Terminal Domain in Regulating Coated Pit Dynamics Revealed by Small Molecule Inhibition|Cell, Volume 146, Issue 3, 471-484, 5 August 2011 Abstract
Further reading
- Wakeham DE, Chen CY, Greene B, Hwang PK, Brodsky FM (October 2003). "Clathrin self-assembly involves coordinated weak interactions favorable for cellular regulation". The EMBO Journal 22 (19): 4980–90. doi:10.1093/emboj/cdg511. PMC 204494. PMID 14517237. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=204494.
- Ford MG, Mills IG, Peter BJ, Vallis Y, Praefcke GJ, Evans PR, McMahon HT (September 2002). "Curvature of clathrin-coated pits driven by epsin". Nature 419 (6905): 361–6. doi:10.1038/nature01020. PMID 12353027.
- Fotin A, Cheng Y, Sliz P, Grigorieff N, Harrison SC, Kirchhausen T, Walz T (December 2004). "Molecular model for a complete clathrin lattice from electron cryomicroscopy". Nature 432 (7017): 573–9. doi:10.1038/nature03079. PMID 15502812.
- Mousavi SA, Malerød L, Berg T, Kjeken R (January 2004). "Clathrin-dependent endocytosis". The Biochemical Journal 377 (Pt 1): 1–16. doi:10.1042/BJ20031000. PMC 1223844. PMID 14505490. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1223844.
- Smith CJ, Grigorieff N, Pearse BM (September 1998). "Clathrin coats at 21 A resolution: a cellular assembly designed to recycle multiple membrane receptors". The EMBO Journal 17 (17): 4943–53. doi:10.1093/emboj/17.17.4943. PMC 1170823. PMID 9724631. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1170823. (Model of Clathrin assembly)
- Pérez-Gómez J, Moore I (March 2007). "Plant endocytosis: it is clathrin after all". Current Biology : CB 17 (6): R217–9. doi:10.1016/j.cub.2007.01.045. PMID 17371763. (Review on involvement of clathrin in plant endocytosis - proven recently)
- Royle SJ, Bright NA, Lagnado L (April 2005). "Clathrin is required for the function of the mitotic spindle". Nature 434 (7037): 1152–7. doi:10.1038/nature03502. PMID 15858577.
- Knuehl C, Chen CY, Manalo V, Hwang PK, Ota N, Brodsky FM (December 2006). "Novel binding sites on clathrin and adaptors regulate distinct aspects of coat assembly". Traffic (Copenhagen, Denmark) 7 (12): 1688–700. doi:10.1111/j.1600-0854.2006.00499.x. PMID 17052248.
- Edeling MA, Smith C, Owen D (January 2006). "Life of a clathrin coat: insights from clathrin and AP structures". Nature Reviews. Molecular Cell Biology 7 (1): 32–44. doi:10.1038/nrm1786. PMID 16493411.
External links
- Eukaryotic Linear Motif resource motif class LIG_Clathr_ClatBox_1.html LIG_Clathr_ClatBox_1
- Eukaryotic Linear Motif resource motif class LIG_Clathr_ClatBox_2.html LIG_Clathr_ClatBox_2
- Clathrin structure
- Membrane Dynamics
- Clathrin Dynamics ASCB Image & Video Library
Synaptic vesicle OtherCOPI COPII RME/Clathrin Caveolae Other/ungrouped Vesicle formationAdaptor protein complex 1: AP1AR · AP1B1 · AP1G1 · AP1G2 · AP1M1 · AP1M2 · AP1S1 · AP1S2 · AP1S3
Adaptor protein complex 2: AP2A1 · AP2A2 · AP2B1 · AP2M1 · AP2S1
Adaptor protein complex 3: AP3B1 · AP3B2 · AP3D1 · AP3M1 · AP3M2 · AP3S1 · AP3S2
Adaptor protein complex 4: AP4B1 · AP4E1 · AP4M1 · AP4S1
Coats: Retromer · TIP47OtherCategories:- Genes on chromosome 12
- Genes on chromosome 4
- Genes on chromosome 17
- Genes on chromosome 22
- Proteins
- Cell biology
- Peripheral membrane proteins
Wikimedia Foundation. 2010.