- Major urinary proteins
-
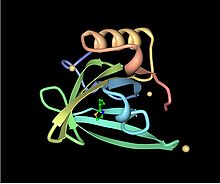
 Tertiary structure of a mouse major urinary protein. The protein has eight beta sheets (yellow) arranged in a beta barrel open at one end, with alpha helices (red) at both the amino- and carboxyl termini. The structure is resolved from Protein Data Bank entry 1i04.
Tertiary structure of a mouse major urinary protein. The protein has eight beta sheets (yellow) arranged in a beta barrel open at one end, with alpha helices (red) at both the amino- and carboxyl termini. The structure is resolved from Protein Data Bank entry 1i04.
Major urinary proteins (Mups), also known as α2u-globulins, are a subfamily of proteins found in abundance in the urine and other secretions of many animals. Mups provide a small range of identifying information about the donor animal, when detected by the vomeronasal organ of the receiving animal. They belong to a larger family of proteins known as lipocalins. Mups are encoded by a cluster of genes, located adjacent to each other on a single stretch of DNA, that varies greatly in number between species: from at least 21 functional genes in mice to none in humans. Mup proteins form a characteristic glove shape, encompassing a ligand-binding pocket that accommodates specific small, organic chemicals.
Urinary proteins were first reported in rodents in 1932, during studies by Thomas Addis into the cause of proteinuria. They are potent human allergens, and are largely responsible for a number of animal allergies, including to cats, horses and rodents. Their endogenous function within an animal is unknown, but may involve regulating energy expenditure. However, as secreted proteins they play multiple roles in chemical communication between animals, functioning as pheromone transporters and stabilizers in rodents and pigs. Mups can also act as protein pheromones themselves. They have been demonstrated to promote aggression in male mice, and one specific Mup protein found in male mouse urine is sexually attractive to female mice. Mups can also function as signals between different species: mice display an instinctive fear response on the detection of Mups derived from predators such as cats and rats.
Contents
Findings
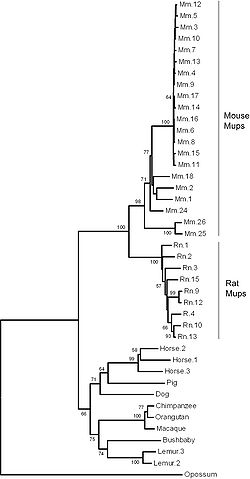 Phylogeny of Mup coding sequences in mammals.[1] The repeatability of the reconstruction was tested by bootstrapping. Interior branches with bootstrap support > 50% are shown.
Phylogeny of Mup coding sequences in mammals.[1] The repeatability of the reconstruction was tested by bootstrapping. Interior branches with bootstrap support > 50% are shown.
Humans in good health excrete urine that is largely free of protein. Therefore since 1827 physicians and scientists have been interested in proteinuria, the excess of protein in human urine, as an indicator of kidney disease.[2][3] To better understand the etiology of proteinuria, some scientists attempted to study the phenomenon in laboratory animals.[4] Between 1932 and 1933 a number of scientists, including Thomas Addis, independently reported the surprising finding that some healthy rodents have protein in their urine.[5][6][7] However, it was not until the 1960s that the major urinary proteins of mice and rats were first described in detail.[8][9] It was found that the proteins are primarily made in the liver of males and secreted through the kidneys into the urine in large quantities (milligrams per day).[8][9][10]
Since they were named, the proteins have been found to be differentially expressed in other glands that secrete products directly into the external environment. These include lacrimal, parotid, submaxillary, sublingual, preputial and mammary glands.[11][12][13] In some species, such as cats and pigs, Mups appear not to be expressed in urine at all and are mainly found in saliva.[14][15] Sometimes the term urinary Mups (uMups) is used to distinguish those Mups expressed in urine from those in other tissues.[16]
Isothermal titration calorimetry studies performed with MUP and associated ligands (pyrazines[17][18], alcohols[19][20], thiazolines[21][22], 6-hydroxy-6-methyl-3-heptanone[23][24], and N-phenylnapthylamine,[25][26]) revealed an unusual binding phenomena. The active site, a beta barrel consisting of 8 beta sheets and 1 alpha helix, has been found to be suboptimally hydrated, resulting in ligand binding being driven by enthalpic solute-solute dispersion forces. This is contrary to most other proteins which exhibit entropy driven binding forces from the reorganization of water molecules. The entropic contribution from protein and ligand desolvation in MUP was found to be almost zero. This enthalpy driven binding interaction has been termed the nonclassical hydrophobic effect. [27]
Mup genes
Between 1979 and 1981, it was estimated that Mups are encoded by a gene family of between 15 and 35 genes and pseudogenes in the mouse and by an estimated 20 genes in the rat.[28][29][30] In 2008 a more precise number of Mup genes in a range of species was determined by analyzing the DNA sequence of whole genomes.[1][31]
Rodents
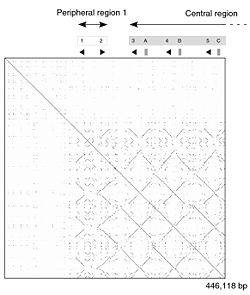 A dot plot showing self-similarity within the mouse Mup cluster.[32] The main diagonal represents the sequence's alignment with itself; lines off the main diagonal represent similar or repetitive patterns within the cluster. The pattern differs between the older, peripheral Class A and the newer, central Class B Mups.
A dot plot showing self-similarity within the mouse Mup cluster.[32] The main diagonal represents the sequence's alignment with itself; lines off the main diagonal represent similar or repetitive patterns within the cluster. The pattern differs between the older, peripheral Class A and the newer, central Class B Mups.
The mouse reference genome has at least 21 distinct Mup genes (with open reading frames), and a further 21 Mup pseudogenes (with reading frames disrupted by a nonsense mutation or an incomplete gene duplication). They are all clustered together, arrayed side by side across 1.92 megabases of DNA on chromosome 4. The 21 functional genes have been divided into two sub-classes based on position and sequence similarity: 6 peripheral Class A Mups, and 15 central Class B Mups.[1][32] The central Class B Mup gene cluster formed through a number of sequential duplications from one of the Class A Mups. As all the Class B genes are almost identical to each other, researchers have concluded that these duplications occurred very recently in mouse evolution. Indeed, the repetitive structure of these central Mup genes means they are likely to be unstable and may vary in number among wild mice.[32] The Class A Mups are more different from each other and are therefore likely to be more stable, older genes but what, if any, functional differences the classes have are unknown.[1] The similarity between the genes makes the region difficult to study using current DNA sequencing technology. Consequently the Mup gene cluster is one of the few parts of the mouse whole genome sequence with gaps remaining, and further genes may remain undiscovered.[1][32]
Rat urine also contains homologous urinary proteins; although they were originally given a different name, α2u-globulins,[9][10] they have since become known as rat Mups.[33][34] Rats have 9 distinct Mup genes and a further 13 pseudogenes clustered together across 1.1 megabases of DNA on chromosome 5. Like in mice, the cluster formed by multiple duplications. However, this occurred independently of the duplications in mice, meaning that both rodent species expanded their Mup gene families separately, but in parallel.[1][35]
Nonrodents
Most other mammals studied, including the pig, cow, cat, dog, bushbaby, macaque, chimpanzee and orangutan, have a single Mup gene. Some, however, have an expanded number: horses have three Mup genes and gray mouse lemurs have at least two. Insects, fish, amphibia, birds and marsupials appear to have disrupted synteny at the chromosomal position of the Mup gene cluster, suggesting the gene family may be specific to placental mammals.[1] Humans are the only placental mammals found not to have any active Mup genes; instead they have a single Mup pseudogene containing a mutation that causes missplicing, rendering it dysfunctional.[1]
Function
Transport proteins
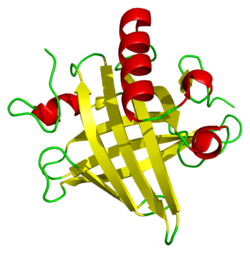
Mups are members of a large family of low-molecular weight (~19 kDa) proteins known as lipocalins.[37] They have a characteristic structure of eight beta sheets arranged in an anti-parallel beta barrel open on one face, with alpha helices at both ends.[37] Consequently, they form a characteristic glove shape, encompassing a cup-like pocket that binds small organic chemicals with high affinity.[1][38] A number of these ligands bind to mouse Mups, including 2-sec-butyl-4,5-dihydrothiazole (abbreviated as SBT or DHT), 6-hydroxy-6-methyl-3-heptanone (HMH) and 2,3 dihydro-exo-brevicomin (DHB).[39][40][41] These are all urine-specific chemicals that have been shown to act as pheromones—molecular signals excreted by one individual that trigger an innate behavioural response in another member of the same species.[39][42] Mouse Mups have also been shown to function as pheromone stabilizers, providing a slow release mechanism that extends the potency of volatile pheromones in male urine scent marks.[43] Given the diversity of Mups in rodents, it was originally thought that different Mups may have differently shaped binding pockets and therefore bind different pheromones. However, detailed studies found that most variable sites are located on the surface of the proteins, and appear to have little effect on ligand binding.[44]
Rat Mups bind different small chemicals. 1-Chlorodecane is the most common ligand, with 2-methyl-N-phenyl-2-propenamide, hexadecane and 2,6,11-trimethyl decane found to be less prominent.[45] Rat Mups also bind limonene-1,2-epoxide, resulting in a disease of the host's kidney, hyaline-droplet nephropathy, that progresses to cancer. Other species do not develop this disorder because their Mups do not bind that particular chemical.[46] Accordingly, when transgenic mice were engineered to express the rat Mup their kidneys developed the disease.[47] The Mup found in pigs, named salivary lipocalin (SAL), is expressed in the salivary gland of males where it tightly binds androstenone and androstenol, both pheromones that cause female pigs to assume a mating stance.[1][15]
Pheromones
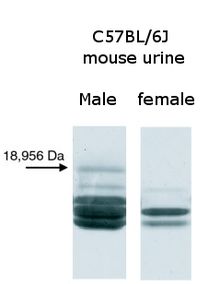 The Mups in C57BL/6J mouse urine analyzed by native gel electrophoresis
The Mups in C57BL/6J mouse urine analyzed by native gel electrophoresis
Studies have sought to find the precise function of Mups in pheromone communication. Mup proteins have been shown to promote puberty and accelerate the estrus cycle in female mice, inducing the Vandenbergh and Whitten effects.[48][49] However, in both cases the Mups had to be presented to the female dissolved in male urine, indicating that the protein requires some urinary context to function. In 2007 Mups normally found in male mouse urine were made in transgenic bacteria, and therefore created devoid of the chemicals they normally bind. These Mups were shown to be sufficient to promote aggressive behaviour in males, even in the absence of urine.[31] In addition, Mups made in bacteria were found to activate olfactory sensory neurons in the vomeronasal organ (VNO), a subsystem of the nose known to detect pheromones via specific sensory receptors, of mice and rats.[31][50] Together, this demonstrated that Mup proteins can act as pheromones themselves, independent of their ligands.[51]
 Fitzwilliam Darcy was the inspiration for the naming of darcin, the Mup that attracts female mice to male urine.
Fitzwilliam Darcy was the inspiration for the naming of darcin, the Mup that attracts female mice to male urine.
Consistent with a role in male-male aggression, adult male mice secrete significantly more Mups into their urine than females, juveniles or castrated male mice. The precise mechanism driving this difference between the sexes is complex, but at least three hormones—testosterone, growth hormone and thyroxine—are known to positively influence the production of Mups in mice.[52] Wild house mouse urine contains variable combinations of four to seven distinct Mup proteins per mouse.[53] Some inbred laboratory mouse strains, such as BALB/c and C57BL/6, also have different proteins expressed in their urine.[32] However, unlike wild mice, different individuals from the same strain express the same protein pattern, an artifact of many generations of inbreeding.[54][55] One unusual Mup is less variable than the others: it is consistently produced by a high proportion of wild male mice and is almost never found in female urine. When this Mup was made in bacteria and used in behavioural testing, it was found to attract female mice. Other Mups were tested but they did not have the same attractive qualities, suggesting the male-specific Mup acts as a sex pheromone.[56] Scientists named this Mup darcin as a humorous reference to Fitzwilliam Darcy, the romantic hero from Pride and Prejudice.[57][58] Taken together, the complex patterns of Mups produced has the potential to provide a range information about the donor animal, such as gender, fertility, social dominance, age, genetic diversity or kinship.[31][59][60] Wild mice (unlike laboratory mice that are genetically identical and which therefore also have identical patterns of Mups in the urine) have individual patterns of Mup expression in their urine that act as a "barcode" to uniquely identify the owner of a scent mark.[59]
Kairomones
In addition to serving as social cues between members of the same species, Mups can act as kairomones—chemical signals that transmit information between species.[61][62][63] Mice are instinctively afraid of the smell of their natural predators, including cats and rats. This occurs even in laboratory mice that have been isolated from predators for hundreds of generations.[64] When the chemical cues responsible for the fear response were purified from cat saliva and rat urine, two homologous protein signals were identified: Fel d 4 (Felis domesticus allergen 4), the product of the cat Mup gene, and Rat n 1 (Rattus norvegicus allergen 1), the product of the rat Mup13 gene.[62] Mice are fearful of these Mups even when they are made in bacteria, but mutant animals that are unable to detect the Mups showed no fear of rats, demonstrating their importance in initiating fearful behaviour.[61][65] It is not known exactly how Mups from different species initiate disparate behaviours, but mouse Mups and predator Mups have been shown to activate unique patterns of sensory neurons in the nose of recipient mice. This implies the mouse perceives them differently, via distinct neural circuits.[61][62] The pheromone receptors responsible for Mup detection are also unknown, though they are thought be members of the V2R receptor class.[31][62]
Allergens
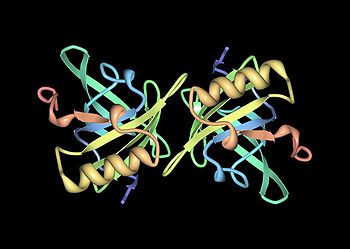 The three-dimensional structure of Equ c 1, shown in the crystallized dimeric form.[66] The structure is resolved from 1EW3.
The three-dimensional structure of Equ c 1, shown in the crystallized dimeric form.[66] The structure is resolved from 1EW3.
Along with other members of the lipocalin protein family, major urinary proteins can be potent allergens to humans.[67] The reason for this is not known, however molecular mimicry between Mups and structurally similar human lipocalins has been proposed as a possible explanation.[68] The protein product of the mouse Mup17 gene, known as Mus m 1, Ag1 or MA1, accounts for much of the allergenic properties of mouse urine.[1][69] The protein is extremely stable in the environment; studies have found 95% of inner city homes, and 82% of all types of homes in the United States have detectable levels in at least one room.[70][71] Similarly, Rat n 1 is a known human allergen.[67] A US study found its presence in 33% of inner city homes, and 21% percent of occupants were sensitized to the allergen.[72] Exposure and sensitization to rodent Mup proteins is considered a risk factor for childhood asthma, and is a leading cause of laboratory animal allergy (LAA)—an occupational disease of laboratory animal technicians and scientists.[73][74][75][76] One study found that two-thirds of laboratory workers who had developed asthmatic reactions to animals had antibodies to Rat n 1.[77]
Mup genes from other mammals also encode allergenic proteins, for example Fel d 4 is primarily produced in the submandibular salivary gland and is deposited onto dander as the cat grooms itself. A study found that 63% of cat allergic people have antibodies against the protein. Most had higher titres of antibodies against Fel d 4 than against Fel d 1, another prominent cat allergen.[14] Likewise, Equ c1 (Equus caballus allergen 1) is the protein product of a horse Mup gene that is found in the liver, sublingual and submaxillary salivary glands.[1][78] It is responsible for about 80% of the antibody response in patients who are chronically exposed to horse allergens.[78]
Metabolism
While the detection of Mups excreted by other animals has been well studied, the functional role in the producing animal is less clear. However, in 2009, Mups were shown to be associated with the regulation of energy expenditure in mice. Scientists found that genetically induced obese, diabetic mice produce thirty times less Mup RNA than their lean siblings.[79] When they delivered Mup protein directly into the bloodstream of these mice, they observed an increase in energy expenditure, physical activity and body temperature and a corresponding decrease in glucose intolerance and insulin resistance. They propose that Mups' beneficial effects on energy metabolism occurs by enhancing mitochondrial function in skeletal muscle.[79] A study found Mups were reduced in diet-induced obese mice. In this case, the presence of Mups in the bloodstream of mice restricted glucose production by directly inhibiting the expression of genes in the liver.[80]
See also
- Cis-vaccenyl acetate, an insect aggression pheromone.
- Major histocompatibility complex, peptides also implicated in individual recognition in mice.
- Proteins produced and secreted by the liver.
Notes
- ^ a b c d e f g h i j k l Logan DW, Marton TF, Stowers L (2008). Vosshall, Leslie B.. ed. "Species specificity in major urinary proteins by parallel evolution". PLoS ONE 3 (9): e3280. doi:10.1371/journal.pone.0003280. PMC 2533699. PMID 18815613. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2533699.
- ^ In that year Richard Bright first related kidney disease, later to become known as Bright's disease, with albuminous urine.
- ^ Comper WD, Hilliard LM, Nikolic-Paterson DJ, Russo LM (December 2008). "Disease-dependent mechanisms of albuminuria". Am. J. Physiol. Renal Physiol. 295 (6): F1589–600. doi:10.1152/ajprenal.00142.2008. PMID 18579704.
- ^ Lemley KV, Pauling L. (1994). "Thomas Addis: 1881–1949". Biographical Memoirs, National Academy of Sciences 63: 1–46. http://books.nap.edu/openbook.php?record_id=4560&page=3.
- ^ Addis T. (1932). "Proteinuria and cylinduria". Proceedings of the California Academy of Sciences 2: 38–52.
- ^ Bell ME. (September 1933). "Albuminuria in the normal male rat". Journal of Physiology 79 (2): 191–93. PMC 1394952. PMID 16994453. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1394952.
- ^ Parfentjev IA, Perlzweig WA. (1933). "The Composition of the Urine of White Mice". The Journal of Biological Chemistry 100 (2): 551–55. http://www.jbc.org/content/100/2/557.full.pdf+html.
- ^ a b Finlayson JS, Asofsky R, Potter M, Runner CC (August 1965). "Major urinary protein complex of normal mice: origin". Science 149 (3687): 981–82. doi:10.1126/science.149.3687.981. PMID 5827345.
- ^ a b c Roy AK, Neuhaus OW (March 1966). "Identification of rat urinary proteins by zone and immunoelectrophoresis". Proceedings of the Society for Experimental Biology and Medicine 121 (3): 894–99. PMID 4160706.
- ^ a b Roy AK, Neuhaus OW (September 1966). "Proof of the hepatic synthesis of a sex-dependent protein in the rat". Biochimica et Biophysica Acta 127 (1): 82–87. doi:10.1016/0304-4165(66)90478-8. PMID 4165835.
- ^ Held WA, Gallagher JF (April 1985). "Rat alpha 2u-globulin mRNA expression in the preputial gland". Biochemical Genetics 23 (3–4): 281–90. doi:10.1007/BF00504325. PMID 2409959.
- ^ Gubits RM, Lynch KR, Kulkarni AB, et al. (October 1984). "Differential regulation of alpha 2u globulin gene expression in liver, lachrymal gland, and salivary gland". The Journal of Biological Chemistry 259 (20): 12803–809. PMID 6208189. http://www.jbc.org/cgi/pmidlookup?view=long&pmid=6208189.
- ^ Shahan K, Denaro M, Gilmartin M, Shi Y, Derman E (May 1987). "Expression of six mouse major urinary protein genes in the mammary, parotid, sublingual, submaxillary, and lachrymal glands and in the liver". Molecular and Cellular Biology 7 (5): 1947–54. PMC 365300. PMID 3600653. http://mcb.asm.org/cgi/pmidlookup?view=long&pmid=3600653.
- ^ a b Smith W, Butler AJ, Hazell LA, et al. (November 2004). "Fel d 4, a cat lipocalin allergen". Clinical and Experimental Allergy 34 (11): 1732–38. doi:10.1111/j.1365-2222.2004.02090.x. PMID 15544598.
- ^ a b Loebel D, Scaloni A, Paolini S, et al. (September 2000). "Cloning, post-translational modifications, heterologous expression and ligand-binding of boar salivary lipocalin". The Biochemical Journal 350 (Pt 2): 369–79. doi:10.1042/0264-6021:3500369. PMC 1221263. PMID 10947950. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1221263.
- ^ Beynon RJ, Hurst JL (February 2003). "Multiple roles of major urinary proteins in the house mouse, Mus domesticus". Biochemical Society Transactions 31 (Pt 1): 142–46. doi:10.1042/BST0310142. PMID 12546672.
- ^ Bingham, R.; Findlay, J. B.; Hsieh, S. Y.; Kalverda, A. P.; Kjellberg, A.; Perazzolo, C.; Phillips, S. E.; Seshadri, K. C.; Trinh, H.; Turnbull, W. B.; Bodenhausen, G.; Homans, S. W. Thermodynamics of binding of 2-methoxy-3-isopropylpyrazine and 2-methoxy-3-isobutylpyrazine to the major urinary protein. J. Am. Chem. Soc., 2004, 126, 1675–1681.
- ^ Barratt, E.; Bingham, R. J.; Warner, D. J.; Laughton, C. A.; Phillips, S. E. V.; Homans, S. W. Van der Waals interactions dominate ligand-protein association in a protein binding site occluded from solvent water. J. Am. Chem. Soc., 2005, 127, 11827-11834.
- ^ Musignat-Caretta, C; Caretta, A.; Cavaggioni, A. Acceleration of puberty onset in female mice by male urinary proteins. J. Physiology 1995, 486, 517-522.
- ^ Malham, R.; Johnstone, S.; Bingham, R.J.; Barratt, E.; Phillips, S.E.; Laughton, C.A.; Homans, S.W. Strong solute-solute dispersive interactions in a protein-ligand complex. J. Am. Chem. Soc. 2005, 127, 17061-17067.
- ^ Sharrow, S.D.; Novotny, M.V.; Stone, M. J. Thermodynamic analysis of binding between mouse major urinary protein-I and the pheromone 2-sec- butyl-4,5-dihydrothiazole. Biochemistry, 2003, 42, 6302-6309.
- ^ Timm, D.E.; Baker, L.J.; Mueller, H.; Zidek, L.; Novotny, M.V. Structural basis of pheromone binding to mouse major urinary protein (MUP-I). Protein Sci. 2001, 10, 997-1004.
- ^ Sharrow, S. D.; Edmonds, K. A.; Goodman, M. A.; Novotny, M. V. ; Stone, M. J. Thermodynamic consequences of disrupting a water-mediated hydrogen bond network in a protein: pheromone complex. Protein Sci. 2005, 14, 249–256.
- ^ Sharrow, S. D.; Edmonds, K. A.; Goodman, M. A.; Novotny, M. V. ; Stone, M. J. Thermodynamic consequences of disrupting a water-mediated hydrogen bond network in a protein: pheromone complex. Protein Sci. 2005, 14, 249–256.
- ^ T. A;. Ferrari, E.; Casali, E.; Patel, J. A.; Spisni, A.; Smith, L.J. The binding cavity of mouse major urinary protein is optimised for a variety of ligand binding modes. Biochem. Bioph. Res. Co. 2009, 390, 1266-1271.
- ^ Homans, S. W. Water, water everywhere — except where it matters? Drug Discovery Today. 2007, 12, 534–539.
- ^ Homans SW (July 2007). "Water Water Everywhere - Except Where it Matters?". Drug Discovery Today 12 (13/14): 535-39. doi:10.1016/j.drudis.2007.05.004. PMID 17631247.
- ^ Kurtz DT (1981). "Rat alpha 2u globulin is encoded by a multigene family". Journal of Molecular and Applied Genetics 1 (1): 29–38. PMID 6180115.
- ^ Hastie ND, Held WA, Toole JJ (June 1979). "Multiple genes coding for the androgen-regulated major urinary proteins of the mouse". Cell 17 (2): 449–57. doi:10.1016/0092-8674(79)90171-5. PMID 88267.
- ^ Bishop JO, Clark AJ, Clissold PM, Hainey S, Francke U (1982). "Two main groups of mouse major urinary protein genes, both largely located on chromosome 4". The EMBO Journal 1 (5): 615–20. PMC 553096. PMID 6329695. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=553096.
- ^ a b c d e Chamero P, Marton TF, Logan DW, et al. (December 2007). "Identification of protein pheromones that promote aggressive behaviour". Nature 450 (7171): 899–902. doi:10.1038/nature05997. PMID 18064011.
- ^ a b c d e Mudge JM, Armstrong SD, McLaren K, et al. (2008). "Dynamic instability of the major urinary protein gene family revealed by genomic and phenotypic comparisons between C57 and 129 strain mice". Genome Biology 9 (5): R91. doi:10.1186/gb-2008-9-5-r91. PMC 2441477. PMID 18507838. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2441477.
- ^ Hurst J, Beynon RJ, Roberts SC, Wyatt TD. (October 2007). Urinary Lipocalins in Rodenta:is there a Generic Model?. Chemical Signals in Vertebrates 11. Springer New York. ISBN 978-0-387-73944-1.
- ^ Cavaggioni A, Mucignat-Caretta C (October 2000). "Major urinary proteins, alpha(2U)-globulins and aphrodisin". Biochimica et Biophysica Acta 1482 (1–2): 218–28. doi:10.1016/S0167-4838(00)00149-7. PMID 11058763.
- ^ McFadyen DA, Addison W, Locke J (May 1999). "Genomic organization of the rat alpha 2u-globulin gene cluster". Mammalian Genome 10 (5): 463–70. doi:10.1007/s003359901024. PMID 10337619.
- ^ Böcskei Z, Groom CR, Flower DR, et al. (November 1992). "Pheromone binding to two rodent urinary proteins revealed by X-ray crystallography". Nature 360 (6400): 186–88. doi:10.1038/360186a0. PMID 1279439.
- ^ a b Flower DR (August 1996). "The lipocalin protein family: structure and function". The Biochemical Journal 318 (1): 1–14. PMC 1217580. PMID 8761444. http://www.biochemj.org/bj/318/0001/bj3180001.htm.
- ^ Ganfornina MD, Gutiérrez G, Bastiani M, Sánchez D (January 2000). "A phylogenetic analysis of the lipocalin protein family". Molecular Biology and Evolution 17 (1): 114–26. PMID 10666711. http://mbe.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=10666711.
- ^ a b Halpern M, Martínez-Marcos A (June 2003). "Structure and function of the vomeronasal system: an update". Progress in Neurobiology 70 (3): 245–318. doi:10.1016/S0301-0082(03)00103-5. PMID 12951145.
- ^ Timm DE, Baker LJ, Mueller H, Zidek L, Novotny MV (May 2001). "Structural basis of pheromone binding to mouse major urinary protein (MUP-I)". Protein Science 10 (5): 997–1004. doi:10.1110/ps.52201. PMC 2374202. PMID 11316880. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2374202.
- ^ Armstrong SD, Robertson DH, Cheetham SA, Hurst JL, Beynon RJ (October 2005). "Structural and functional differences in isoforms of mouse major urinary proteins: a male-specific protein that preferentially binds a male pheromone". The Biochemical Journal 391 (Pt 2): 343–50. doi:10.1042/BJ20050404. PMC 1276933. PMID 15934926. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1276933.
- ^ Stowers L, Marton TF (June 2005). "What is a pheromone? Mammalian pheromones reconsidered". Neuron 46 (5): 699–702. doi:10.1016/j.neuron.2005.04.032. PMID 15924856.
- ^ Hurst JL, Robertson DHL, Tolladay U, Beynon RJ (May 1998). "Proteins in urine scent marks of male house mice extend the longevity of olfactory signals". Animal Behaviour 55 (5): 1289–97. doi:10.1006/anbe.1997.0650. PMID 9632512.
- ^ Darwish Marie A, Veggerby C, Robertson DH, et al. (February 2001). "Effect of polymorphisms on ligand binding by mouse major urinary proteins". Protein Science 10 (2): 411–17. doi:10.1110/ps.31701. PMC 2373947. PMID 11266626. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2373947.
- ^ Rajkumar R, Ilayaraja R, Mucignat C, Cavaggioni A, Archunan G (August 2009). "Identification of alpha2u-globulin and bound volatiles in the Indian common house rat (Rattus rattus)". Indian Journal of Biochemistry & Biophysics 46 (4): 319–24. PMID 19788064.
- ^ Lehman-McKeeman LD, Caudill D (February 1992). "Biochemical basis for mouse resistance to hyaline droplet nephropathy: lack of relevance of the alpha 2u-globulin protein superfamily in this male rat-specific syndrome". Toxicology and Applied Pharmacology 112 (2): 214–21. doi:10.1016/0041-008X(92)90190-4. PMID 1371614.
- ^ Lehman-McKeeman LD, Caudill D (November 1994). "d-Limonene induced hyaline droplet nephropathy in alpha 2u-globulin transgenic mice". Fundamental and Applied Toxicology 23 (4): 562–68. doi:10.1006/faat.1994.1141. PMID 7532604.
- ^ Mucignat-Caretta C, Caretta A, Cavaggioni A (July 1995). "Acceleration of puberty onset in female mice by male urinary proteins". J. Physiol. (Lond.). 486 (Pt 2): 517–22. PMC 1156539. PMID 7473215. http://www.jphysiol.org/cgi/pmidlookup?view=long&pmid=7473215.
- ^ Marchlewska-koj A, Caretta A, Mucignat-Caretta C, Olejniczak, P (October 2000). "Stimulation of estrus in female mice by male urinary proteins". Journal of Chemical Ecology 26 (10): 2355–65. doi:10.1023/A:1005578911652.
- ^ Krieger J, Schmitt A, Löbel D, et al. (February 1999). "Selective activation of G protein subtypes in the vomeronasal organ upon stimulation with urine-derived compounds". The Journal of Biological Chemistry 274 (8): 4655–62. doi:10.1074/jbc.274.8.4655. PMID 9988702.
- ^ "Aggression protein found in mice". BBC News. (5 December 2007). http://news.bbc.co.uk/2/hi/science/nature/7129176.stm. Retrieved 26 September 2009.
- ^ Knopf JL, Gallagher JF, Held WA (December 1983). "Differential, multihormonal regulation of the mouse major urinary protein gene family in the liver". Molecular and Cellular Biology 3 (12): 2232–40. PMC 370094. PMID 6656765. http://mcb.asm.org/cgi/pmidlookup?view=long&pmid=6656765.
- ^ Robertson DH, Hurst JL, Bolgar MS, Gaskell SJ, Beynon RJ (1997). "Molecular heterogeneity of urinary proteins in wild house mouse populations". Rapid Communications in Mass Spectrometry 11 (7): 786–90. doi:10.1002/(SICI)1097-0231(19970422)11:7<786::AID-RCM876>3.0.CO;2-8. PMID 9161047.
- ^ Robertson DH, Cox KA, Gaskell SJ, Evershed RP, Beynon RJ (May 1996). "Molecular heterogeneity in the Major Urinary Proteins of the house mouse Mus musculus". The Biochemical Journal 316 (Pt 1): 265–72. PMC 1217333. PMID 8645216. http://www.biochemj.org/bj/316/0265/bj3160265.htm.
- ^ Cheetham SA, Smith AL, Armstrong SD, Beynon RJ, Hurst JL (February 2009). "Limited variation in the major urinary proteins of laboratory mice". Physiology & Behavior 96 (2): 253–61. doi:10.1016/j.physbeh.2008.10.005. PMID 18973768.
- ^ Brennan PA (May 2010). "On the scent of sexual attraction". BMC Biol 8 (1): 71. doi:10.1186/1741-7007-8-71. PMC 2880966. PMID 20504292. http://www.biomedcentral.com/1741-7007/8/71.
- ^ Roberts SA, Simpson DM, Armstrong SD, et al. (June 2010). "Darcin: a male pheromone that stimulates female memory and sexual attraction to an individual male's odour". BMC Biol 8 (1): 75. doi:10.1186/1741-7007-8-75. PMC 2890510. PMID 20525243. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2890510.
- ^ Moskowitz, C (3 June 2010). "Biologists Learn Why Mice Go Gaga for Urine". FoxNews.com. FOX News Network. http://www.foxnews.com/scitech/2010/06/03/biologists-learn-mice-urine-darcin/. Retrieved 9 June 2010.
- ^ a b Hurst JL, Payne CE, Nevison CM, et al. (December 2001). "Individual recognition in mice mediated by major urinary proteins". Nature 414 (6864): 631–34. doi:10.1038/414631a. PMID 11740558.
- ^ Thom MD, Stockley P, Jury F, Ollier WE, Beynon RJ, Hurst JL (April 2008). "The direct assessment of genetic heterozygosity through scent in the mouse". Current Biology 18 (8): 619–23. doi:10.1016/j.cub.2008.03.056. PMID 18424142.
- ^ a b c Papes F, Logan DW, Stowers, L (May 2010). "The Vomeronasal Organ Mediates Interspecies Defensive Behaviors through Detection of Protein Pheromone Homologs". Cell 141 (4): 692–703. doi:10.1016/j.cell.2010.03.037. PMC 2873972. PMID 20478258. http://www.cell.com/abstract/S0092-8674%2810%2900355-7.
- ^ a b c d Rodriguez I (May 2010). "The chemical MUPpeteer". Cell 141 (4): 568–70. doi:10.1016/j.cell.2010.04.032. PMID 20478249.
- ^ "Why mice fear the smell of cats". BBC News. (17 May 2010). http://news.bbc.co.uk/1/hi/science_and_environment/10117428.stm. Retrieved 18 May 2010.
- ^ Ehrenberg, Rachel (5 June 2010). "Fight or flee, it's in the pee". Science News. http://www.sciencenews.org/view/generic/id/59205/title/Fight_or_flee,_it%E2%80%99s_in_the_pee.
- ^ Bhanoo, Sindya (17 May 2010). "When a Mouse Smells a Rat". The New York Times. http://www.nytimes.com/2010/05/18/science/18obmouse.html.
- ^ Lascombe MB, Grégoire C, Poncet P, et al. (July 2000). "Crystal structure of the allergen Equ c 1. A dimeric lipocalin with restricted IgE-reactive epitopes". The Journal of Biological Chemistry 275 (28): 21572–77. doi:10.1074/jbc.M002854200. PMID 10787420.
- ^ a b Lockey R, Ledford DK (2008). "Mammalian Allergens". Allergens and Allergen Immunotherapy. Volume 21 of Clinical allergy and immunology. Informa Health Care. pp. 201–218. ISBN 978-1-4200-6197-0.
- ^ Virtanen T, Zeiler T, Mäntyjärvi R (December 1999). "Important animal allergens are lipocalin proteins: why are they allergenic?". Int. Arch. Allergy Immunol. 120 (4): 247–58. doi:10.1159/000024277. PMID 10640908.
- ^ Lorusso JR, Moffat S, Ohman JL (November 1986). "Immunologic and biochemical properties of the major mouse urinary allergen (Mus m 1)". The Journal of Allergy and Clinical Immunology 78 (5 Pt 1): 928–37. doi:10.1016/0091-6749(86)90242-3. PMID 3097107.
- ^ Cohn RD, Arbes SJ, Yin M, Jaramillo R, Zeldin DC (June 2004). "National prevalence and exposure risk for mouse allergen in US households". The Journal of Allergy and Clinical Immunology 113 (6): 1167–71. doi:10.1016/j.jaci.2003.12.592. PMID 15208600.
- ^ Phipatanakul W, Eggleston PA, Wright EC, Wood RA (December 2000). "Mouse allergen. I. The prevalence of mouse allergen in inner-city homes. The National Cooperative Inner-City Asthma Study". The Journal of Allergy and Clinical Immunology 106 (6): 1070–74. doi:10.1067/mai.2000.110796. PMID 11112888.
- ^ Perry T, Matsui E, Merriman B, Duong T, Eggleston P (August 2003). "The prevalence of rat allergen in inner-city homes and its relationship to sensitization and asthma morbidity". The Journal of Allergy and Clinical Immunology 112 (2): 346–52. doi:10.1067/mai.2003.1640. PMID 12897741.
- ^ Wood RA (2001). "Laboratory animal allergens". ILAR Journal 42 (1): 12–16. PMID 11123185.
- ^ Gaffin JM, Phipatanakul W (April 2009). "The role of indoor allergens in the development of asthma". Current Opinion in Allergy and Clinical Immunology 9 (2): 128–35. doi:10.1097/ACI.0b013e32832678b0. PMC 2674017. PMID 19326507. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2674017.
- ^ Pongracic JA, Visness CM, Gruchalla RS, Evans R, Mitchell HE (July 2008). "Effect of mouse allergen and rodent environmental intervention on asthma in inner-city children". Annals of Allergy, Asthma & Immunology 101 (1): 35–41. doi:10.1016/S1081-1206(10)60832-0. PMID 18681082.
- ^ Gordon S, Preece R (September 2003). "Prevention of laboratory animal allergy". Occupational Medicine 53 (6): 371–77. doi:10.1093/occmed/kqg117. PMID 14514903.
- ^ Platts-Mills TA, Longbottom J, Edwards J, Cockroft A, Wilkins S (March 1987). "Occupational asthma and rhinitis related to laboratory rats: serum IgG and IgE antibodies to the rat urinary allergen". The Journal of Allergy and Clinical Immunology 79 (3): 505–15. doi:10.1016/0091-6749(87)90369-1. PMID 3819230.
- ^ a b Gregoire C, Rosinski-Chupin I, Rabillon J, Alzari PM, David B, Dandeu JP (December 1996). "cDNA cloning and sequencing reveal the major horse allergen Equ c1 to be a glycoprotein member of the lipocalin superfamily". The Journal of Biological Chemistry 271 (51): 32951–59. doi:10.1074/jbc.271.51.32951. PMID 8955138.
- ^ a b Hui X, Zhu W, Wang Y, et al. (May 2009). "Major urinary protein-1 increases energy expenditure and improves glucose intolerance through enhancing mitochondrial function in skeletal muscle of diabetic mice". The Journal of Biological Chemistry 284 (21): 14050–57. doi:10.1074/jbc.M109.001107. PMC 2682853. PMID 19336396. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2682853.
- ^ Zhou Y, Jiang L, Rui L (April 2009). "Identification of MUP1 as a regulator for glucose and lipid metabolism in mice". The Journal of Biological Chemistry 284 (17): 11152–59. doi:10.1074/jbc.M900754200. PMC 2670120. PMID 19258313. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2670120.
External links
- Scent of a Rodent, The Why Files – The Science Behind The News
- Fear Signals from Predators on YouTube, a video describing the research that determined Mups were kairomones
Fatty acid Hormone peptide hormone: Follistatin · Growth hormone binding protein · Insulin-like growth factor binding protein (IGFBP1, IGFBP2, IGFBP3, IGFBP4, IGFBP5, IGFBP6, IGFBP7) · Neurophysins (Neurophysin I, II)
steroid hormone: Sex hormone binding globulin/Androgen binding protein · Transcortin · Thyroxine-binding globulin · TransthyretinMetal/element calcium (Calcium-binding protein, Calmodulin-binding proteins) · copper (Ceruloplasmin) · iron (Iron-binding proteins, Transferrin receptor)Vitamin Other Acyl carrier protein · Adaptor protein · Cholesterylester transfer protein · F-box protein · GTP-binding protein · Latent TGF-beta binding protein · Light-harvesting complex · Major urinary proteins · Membrane transport protein · Odorant binding proteinB proteins: BY STRUCTURE: membrane, globular (en, ca, an), fibrous Categories:- Pheromones
- Allergology
- Protein families
- Urine
Wikimedia Foundation. 2010.