- Froth flotation
-
 Diagram of a cylindrical froth flotation cell with camera and light used in image analysis of the froth surface.
Diagram of a cylindrical froth flotation cell with camera and light used in image analysis of the froth surface.
Froth flotation is a process for selectively separating hydrophobic materials from hydrophilic. This is used in several processing industries. Historically this was first used in the mining industry.
Contents
History
William Haynes in 1869 patented a process for separating sulfide and gangue minerals using oil and called it bulk-oil flotation.
The first successful commercial flotation process for mineral sulphides was invented by Frank Elmore[1] who worked on the development with his brother, Stanley. The Glasdir copper mine at Llanelltyd, near Dolgellau in North Wales was bought in 1896 by the Elmore brothers in conjunction with their father, William. In 1897, the Elmore brothers installed the world's first industrial size commercial flotation process for mineral beneficiation at the Glasdir mine. The process was not froth flotation but used oil to agglomerate pulverised sulphides and buoy them to the surface, and was patented in 1898 with a description of the process published in 1903 in the Engineering and Mining Journal. By this time they had recognized the importance of air bubbles in assisting the oil to carry away the mineral particles. The Elmores had formed a company known as the Ore Concentration Syndicate Ltd to promote the commercial use of the process worldwide. However developments elsewhere, particularly in Australia by Minerals Separation Ltd., led to decades of hard fought legal battles and litigations which, ultimately, were lost as the process was superseded by more advanced techniques.
The modern froth flotation process was independently invented in the early 1900s in Australia by C.V Potter and around the same time by G.D Delprat.[2] Initially, naturally occurring chemicals such as fatty acids and oils were used as flotation reagents in a large quantity to increase the hydrophobicity of the valuable minerals. Since then, the process has been adapted and applied to a wide variety of materials to be separated, and additional collector agents, including surfactants and synthetic compounds have been adopted for various applications.
In the 1960s the froth flotation technique was adapted for deinking recycled paper.
Industries
Mining
 Froth flotation to separate plastics, Argonne National Laboratory
Froth flotation to separate plastics, Argonne National Laboratory
Froth flotation is a process for separating minerals from gangue by taking advantage of differences in their hydrophobicity. Hydrophobicity differences between valuable minerals and waste gangue are increased through the use of surfactants and wetting agents. The selective separation of the minerals makes processing complex (that is, mixed) ores economically feasible. The flotation process is used for the separation of a large range of sulfides, carbonates and oxides prior to further refinement. Phosphates and coal are also upgraded by flotation technology.
Waste water treatment
The flotation process is also widely used in industrial waste water treatment plants, where it removes fats, oil, grease and suspended solids from waste water. These units are called Dissolved air flotation (DAF) units.[3] In particular, dissolved air flotation units are used in removing oil from the wastewater effluents of oil refineries, petrochemical and chemical plants, natural gas processing plants and similar industrial facilities.
Paper recycling
Main article: DeinkingFroth flotation is one of the processes used to recover recycled paper. In the paper industry this step is called deinking or just flotation. The target is to release and remove the hydrophobic contaminants from the recycled paper. The contaminants are mostly printing ink and stickies. Normally the setup is a two stage system with 3,4 or 5 flotation cells in series.[4]
Principle of operation
Froth flotation commences by comminution (that is, crushing and grinding), which is used to increase the surface area of the ore for subsequent processing and break the rocks into the desired mineral and gangue in a process known as liberation, which then has to be separated from the desired mineral. The ore is ground into a fine powder and mixed with water to form a slurry. The desired mineral is rendered hydrophobic by the addition of a surfactant or collector chemical. The particular chemical depends on which mineral is being refined. As an example, SEX is added as a collector in the slective flotation of galena and sphalerite, after the addition of other flotation reagents.. This slurry (more properly called the pulp) of hydrophobic particles and hydrophilic particles is then introduced to a water bath which is aerated, creating bubbles. The hydrophobic particles attach to the air bubbles, which rise to the surface, forming a froth. The froth is removed and the concentrate(con) is further refined.
Science of flotation
To be effective on a given ore slurry, the collectors are chosen based upon their selective wetting of the types of particles to be separated. A good collector will adsorb, physically or chemically, with one of the types of particles. This provides the thermodynamic requirement for the particles to bind to the surface of a bubble. The wetting activity of a surfactant on a particle can be quantified by measuring the contact angles that the liquid/bubble interface makes with it. Another important measure for attachment of bubbles to particles is induction time. The induction time is the time required for the particle and bubble to rupture the thin film separating the particle and bubble. This rupturing is achieved by the surface forces between the particle and bubble.
The mechanisms for the bubble-particle attachment is very complex and consists of three steps, collision, attachment and detachment. The collision is achieved by particles being within the collision tube of a bubble and this is affected by the velocity of the bubble and radius of the bubble. The collision tube corresponds to the region in which a particle will collide with the bubble, with the perimeter of the collision tube corresponding to the grazing trajectory.
The attachment of the particle to the bubble is controlled by the induction time of the particle and bubble. The particle and bubble need to bind and this occurs if the time in which the particle and bubble are in contact with each other is larger than the required induction time. This induction time is effected by the fluid viscosity, particle and bubble size and the forces between the particle and bubbles.
The detachment of a particle and bubble occurs when the force exerted by the surface tension is exceeded by shear forces and gravitational forces. These forces are complex and vary within the cell. High shear will be experienced close to the impeller of a mechanical flotation cell and mostly gravitational force in the collection and cleaning zone of a flotation column.
Significant issues of entrainment of fine particles occurs as these particles experience low collision efficiencies as well as sliming and degradation of the particle surfaces. Coarse particles show a low recovery of the valuable mineral due to the low liberation and high detachment efficiencies.
Flotation equipment
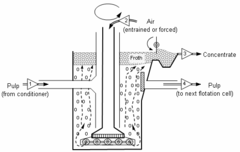
Diagram of froth flotation cell. Numbered triangles show direction of stream flow. A mixture of ore and water called pulp [1] enters the cell from a conditioner, and flows to the bottom of the cell. Air [2] or nitrogen is passed down a vertical impeller where shearing forces break the air stream into small bubbles. The mineral concentrate froth is collected from the top of the cell [3], while the pulp [4] flows to another cell.
Flotation can be performed in rectangular or cylindrical mechanically agitated cells or tanks, flotation columns, Jameson cells or deinking flotation machines.
Mechanical cells use a large mixer and diffuser mechanism at the bottom of the mixing tank to introduce air and provide mixing action. Flotation columns use air spargers to introduce air at the bottom of a tall column while introducing slurry above. The countercurrent motion of the slurry flowing down and the air flowing up provides mixing action. Mechanical cells generally have a higher throughput rate, but produce material that is of lower quality, while flotation columns generally have a low throughput rate but produce higher quality material.
The Jameson cell uses neither impellers nor spargers, instead combining the slurry with air in a downcomer where high shear creates the turbulent conditions required for bubble particle contacting.
Mechanics of flotation
The following steps are followed, following grinding to liberate the mineral particles:
- Reagent conditioning to achieve hydrophobic surface charges on the desired particles
- Collection and upward transport by bubbles in an intimate contact with air or nitrogen
- Formation of a stable froth on the surface of the flotation cell
- Separation of the mineral laden froth from the bath (flotation cell)
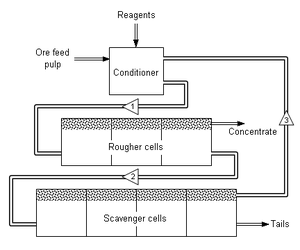
Simple flotation circuit for mineral concentration. Numbered triangles show direction of stream flow, Various flotation reagents are added to a mixture of ore and water (called pulp) in a conditioning tank. The flow rate and tank size are designed to give the minerals enough time to be activated. The conditioner pulp [1] is fed to a bank of rougher cells which remove most of the desired minerals as a concentrate. The rougher pulp [2] passes to a bank of scavenger cells where additional reagents may be added. The scavenger cell froth [3] is usually returned to the rougher cells for additional treatment, but in some cases may be sent to special cleaner cells. The scavenger pulp is usually barren enough to be discarded as tails. More complex flotation circuits have several sets of cleaner and re-cleaner cells, and intermediate re-grinding of pulp or concentrate.
Chemicals of flotation
Collectors
Collectors either chemically bond (chemisorption) on a hydrophobic mineral surface, or adsorb onto the surface in the case of, for example, coal flotation through physisorption. Collectors increase the natural hydrophobicity of the surface, increasing the separability of the hydrophobic and hydrophilic particles.
- Potassium amyl xanthate (PAX)
- Potassium isobutyl xanthate (PIBX)
- Potassium ethyl xanthate (KEX)
- Sodium isobutyl xanthate (SIBX)
- Sodium isopropyl xanthate (SIPX)
- Sodium ethyl xanthate (SEX)
Dithiophosphates
- Thiocarbamates
- Xanthogen Formates
- Thionocarbamates
- Thiocarbanilide
Frothers
- Pine oil
- Alcohols (methyl isobutyl carbinol (MIBC))
- Polyglycols
- Polyoxyparafins|
- Cresylic Acid (Xylenol)
Modifiers
pH modifiers such as:
- Lime CaO
- Soda ash Na2CO3
- Caustic soda NaOH
- Acid H2SO4, HCl
Cationic modifiers:
- Ba2+, Ca2+, Cu+, Pb2+, Zn2+, Ag+
Anionic modifiers:
- SiO32-, PO43-, CN-, CO32-, S2-
Organic modifers:
Chemicals for deinking of recycled paper
- pH control: sodium silicate and sodium hydroxide
- Calcium ion source: hard water, lime or calcium chloride
- Collector: fatty acid, fatty acid emulsion, fatty acid soap and/or organo-modified siloxane[5]
Specific ore applications
Sulfide ores - Copper (see copper extraction)
- Copper-Molybdenum
- Lead-Zinc
- Lead-Zinc-Iron
- Copper-Lead-Zinc-Iron
- Gold-Silver
- Oxide Copper and Lead
- Nickel
- Nickel-Copper
Nonsulfide ores Processes Absorption · Acid-base extraction · Adsorption · Chromatography · Cross-flow filtration · Crystallization · Cyclonic separation · Dialysis (biochemistry) · Dissolved air flotation · Distillation · Drying · Electrochromatography · Electrofiltration · Filtration · Flocculation · Froth flotation · Gravity separation · Leaching (chemical science) · Liquid-liquid extraction · Microfiltration · Osmosis · Precipitation (chemistry) · Recrystallization · Reverse osmosis · Sedimentation · Solid Phase Extraction · Sublimation · Ultrafiltration (industrial)
Devices Multiphase systems Concepts See also
People
- James M. Hyde, inventor and metallurgist
References
- ^ "Wales - The birthplace of Flotation". http://www.maelgwyn.com/birthplaceflotation.html#top. Retrieved 2010-01-13.
- ^ "Historical Note". Minerals Separation Ltd. http://www.austehc.unimelb.edu.au/guides/mine/historicalnote.htm. Retrieved 2007-12-30.
- ^ Beychok, Milton R. (1967). Aqueous Wastes from Petroleum and Petrochemical Plants (1st ed.). John Wiley & Sons Ltd.. LCCN 67019834.
- ^ Voith EcoCell flotation plant http://www.voithpaper.com/applications/productsearch/files/594_VPR-PB-07-0001-GB-07.pdf
- ^ METHOD OF DEINKING. 2004 (published 05.02.2004).
Categories:- Chemical engineering
- Industrial processes
- Unit operations
- Water treatment
- Metallurgical processes
Wikimedia Foundation. 2010.