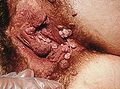
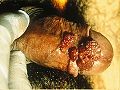
- Genital wart
-
Genital wart(s) Classification and external resources 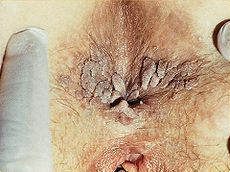
Severe case of genital warts around the anusICD-10 A63.0 ICD-9 078.11 DiseasesDB 29120 eMedicine derm/454 med/1037 MeSH C02.256.650.810.217 Genital warts (or Condylomata acuminata, venereal warts, anal warts and anogenital warts) is a highly contagious sexually transmitted disease caused by some sub-types of human papillomavirus (HPV). It is spread through direct skin-to-skin contact during oral, genital, or anal sex with an infected partner. Warts are the most easily recognized symptom of genital HPV infection, where types 6 and 11 are responsible for 90% of genital warts cases.[1] Whilst of those infected with genital HPV it is estimated that only a "small percentage"[2] (between 1%[3] and 5%[4]) develop genital warts, those infected can still transmit the virus. Other types of HPV also cause cervical cancer and probably most anal cancers, however it is important to underline that the types of HPV that cause the overwhelming majority of genital warts are not the same as those that can potentially increase the risk of genital or anal cancer.[5][6][7][8][9] HPV prevalence at any one time has been observed in some studies at 27% over all sexually active people, rising to 45% between the ages of 14 and 19.[1]
Contents
Signs and symptoms
Genital warts often occur in clusters and can be very tiny or can spread into large masses in the genital or penis area. In other cases they look like small stalks. In women they occur on the outside and inside of the vagina, on the opening (cervix) to the womb (uterus), or around (or inside) the anus. They are approximately as prevalent in men but the symptoms may be less obvious. When present, they usually are seen on the tip of the penis. They also may be found on the shaft of the penis, on the scrotum, or around (or inside) the anus. Rarely, genital warts also can develop in the mouth or throat of a person who has had oral sex with an infected person.[10]
The viral particles are able to penetrate the skin and mucosal surfaces through microscopic abrasions in the genital area, which occur during sexual activity. Once cells are invaded by HPV, a latency (quiet) period of months to years (decades) may occur. HPV can last for several years without a symptom.[11] Having sex with a partner whose HPV infection is latent and demonstrates no outward symptoms still leaves one vulnerable to becoming infected. If an individual has unprotected sex with an infected partner, there is a 70% chance that he or she will also become infected. The immune system eventually clears the virus through interleukins, which recruit interferons, which slow viral replication.[12]
Diagnosis
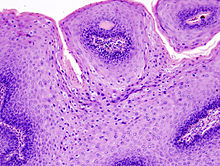 Micrograph of a genital wart with the characteristic changes (parakeratosis, koilocytes, papillomatosis). H&E stain.
Micrograph of a genital wart with the characteristic changes (parakeratosis, koilocytes, papillomatosis). H&E stain.
Genital warts, histopathologically, characteristically rise above the skin surface due to enlargement of the dermal papillae, have parakeratosis and the characteristic nuclear changes typical of HPV infections (nuclear enlargement with perinuclear clearing).
Prevention
See also: HPV vaccineGardasil (sold by Merck & Co.) is a vaccine that protects against human papillomavirus types 16, 18, 6, and 11. Types 6 and 11 cause genital warts, while 16 and 18 cause cervical cancer. The vaccine is preventive, not therapeutic, and must be given before exposure to the virus type to be effective, ideally before the beginning of sexual activity. The vaccine is widely approved for use by young women, it is being tested for young men,[13][14] and has been approved for males in some areas, such as the UK, the US and Canada.
Management
There is no cure for HPV, but there are methods to treat visible warts, which could reduce infectivity, although there are no trials studying the effectiveness of removing visible warts in reducing transmission.[15] Genital warts may disappear without treatment, but sometimes eventually develop a fleshy, small raised growth. There is no way to predict whether they will grow or disappear. Warts can sometimes be identified because they show up as white when acetic acid is applied, but this method is not recommended on the vulva because microtrauma and inflammation can also show up as acetowhite.[12] Magnifying glasses or colposcope may also be used to aid in identifying small warts.[12]
Depending on the sizes and locations of warts (as well as other factors), a doctor will offer one of several ways to treat them. Podofilox is the first-line treatment due to its low cost.[16] Almost all treatments can potentially cause depigmentation or scarring.[15]
- A 0.15% – 0.5% podophyllotoxin (also called podofilox) solution in a gel or cream. Marketed as Condylox (0.5%), Wartec (0.15%) and Warticon (0.15%),[17] it can be applied by the patient to the affected area and is not washed off. It is the purified and standardized active ingredient of the podophyllin (see below). Podofilox is safer and more effective than podophyllin.[17] Skin erosion and pain are more commonly reported than with imiquimod and sinecatechins.[18] Its use is cycled (2 times per day for 3 days then 4–7 days off); one review states that it should only be used for four cycles.[19]
- Imiquimod (Aldara) is a topical immune response cream, applied to the affected area. It causes less local irritation than podofilox but may cause fungal infections (11% in package insert) and flu-like symptoms (less than 5% disclosed in package insert).[18]
- Sinecatechins (marketed as Veregen and Polyphenon E) is an ointment of catechins (55% epigallocatechin gallate[12]) extracted from green tea and other components. Mode of action is undetermined.[20] It appears to have higher clearance rates than podophyllotoxin and imiquimod and causes less local irritation, but clearance takes longer than with imiquimod.[18]
- Liquid nitrogen cryosurgery is safe for pregnancy. It kills warts 71–79% of the time, but recurrence is 38% to 73% 6 months after treatment.[12] Local infections have been reported.[12]
- Trichloroacetic acid (TCA) is less effective than cryosurgery,[19] and is not recommended for use in the vagina, cervix, or urinary meatus.[12]
- Surgical excision is best for large warts, and has a greater risk of scarring.[15]
- Laser ablation does not seem to be any more effective than other physician-applied methods,[3] but is often used as a last resort and is extremely expensive.[12]
- A 20% podophyllin anti-mitotic solution , applied to the affected area and later washed off. However, this crude herbal extract is not recommended for use on vagina, urethra, perianal area, or cervix,[12] and must be applied by a physician.[17] Reported reactions include nausea, vomiting, fever, confusion, coma, renal failure, ileus, and leukopenia; death has been reported with extensive topical application, or application on mucous membranes.[12]
- Interferon can be used; it is effective, but it is also expensive and its effect is inconsistent.[19]
- Electrocauterization can be used; it is an older procedure but recovery time is generally longer. In severe cases of genital warts, treatment may require general or spinal anesthesia. This is a surgical procedure. More effective than cryosurgery and recurrence is at a much lower rate.
- Discontinued
- A 5% 5-fluorouracil (5-FU) cream was used, but it is no longer considered an acceptable treatment due to the side-effects.[12]
Podophyllin and podofilox should not be used during pregnancy, as they are absorbed by the skin and could cause birth defects in the fetus.
Epidemiology
Genital HPV infections have an estimated prevalence in the US of 10–20% and clinical manifestations in 1% of the sexually active adult population.[19] US incidence of HPV infection has increased between 1975 and 2006.[19] About 80% of those infected are between the ages of 17–33.[19] Although treatments can remove the warts, they do not remove the HPV, so warts can recur after treatment (about 50–73% of the time[21]). Warts can also spontaneously regress (with or without treatment).[19] Traditional theories postulated that the virus remained in the body for a lifetime. However, new studies using sensitive DNA techniques have shown that through immunological response the virus can either be cleared or suppressed to levels below what polymerase chain reaction (PCR) tests can measure. One study testing genital skin for subclinical HPV using PCR found a prevalence of 10%.[19]
References
- ^ a b http://www.webmd.com/sexual-conditions/hpv-genital-warts/hpv-virus-information-about-human-papillomavirus
- ^ http://www.aad.org/public/publications/pamphlets/viral_genital.html
- ^ a b O'Mahony C (2005). "Genital warts: current and future management options". Am J Clin Dermatol 6 (4): 239–243. PMID 16060711.
- ^ http://www.mckinley.illinois.edu/Handouts/genital_warts.html
- ^ http://96.10.7.92/hpv_diagnosing.shtml
- ^ http://dermatology.cdlib.org/126/original/HPV/payal.html
- ^ http://www.cdc.gov/std/hpv/stdfact-hpv.htm
- ^ http://www.cancerhelp.org.uk/type/cervical-cancer/about/cervical-cancer-risks-and-causes#hpv
- ^ http://brown.edu/Student_Services/Health_Services/Health_Education/sexual_health/sexually_transmitted_infections/hpv.php
- ^ Genkins, Gill. "Genital warts". BBC. http://www.bbc.co.uk/health/physical_health/sexual_health/stis_warts.shtml. Retrieved 17 November 2011.
- ^ "American Cancer Society: "Can Anal Cancer Be Prevented?"". http://www.cancer.org/docroot/CRI/content/CRI_2_2_2x_Can_Anal_Cancer_Be_Prevented.asp?sitearea=. Retrieved 2008-09-10.[dead link]
- ^ a b c d e f g h i j k Mayeaux EJ, Dunton C (July 2008). "Modern management of external genital warts". J Low Genit Tract Dis 12 (3): 185–192. doi:10.1097/LGT.0b013e31815dd4b4. PMID 18596459.
- ^ Cortez, Michelle Fay and Pettypiece, Shannon. "Merck Cancer Shot Cuts Genital Warts, Lesions in Men". Bloomberg News. (Bloomberg.com) 13 Nov 2008.
- ^ HPV Vaccine Gardasil May Help Boys, Men by WebMD Health News, Nov 13, 2008
- ^ a b c Kodner CM, Nasraty S (December 2004). "Management of genital warts". Am Fam Physician 70 (12): 2335–2342. PMID 15617297. http://www.aafp.org/afp/20041215/2335.html.
- ^ Fox PA, Tung MY (2005). "Human papillomavirus: burden of illness and treatment cost considerations". Am J Clin Dermatol 6 (6): 365–381. PMID 16343025.
- ^ a b c von Krogh G, Longstaff E (December 2001). "Podophyllin office therapy against condyloma should be abandoned". Sex Transm Infect 77 (6): 409–412. doi:10.1136/sti.77.6.409. PMC 1744412. PMID 11714936. http://sti.bmj.com/cgi/content/full/77/6/409.
- ^ a b c Meltzer SM, Monk BJ, Tewari KS (March 2009). "Green tea catechins for treatment of external genital warts". Am. J. Obstet. Gynecol. 200 (3): 233.e1–7. doi:10.1016/j.ajog.2008.07.064. PMID 19019336.
- ^ a b c d e f g h Scheinfeld N, Lehman DS (2006). "An evidence-based review of medical and surgical treatments of genital warts". Dermatol. Online J. 12 (3): 5. PMID 16638419. http://dermatology.cdlib.org/123/reviews/warts/scheinfeld.html.
- ^ "Veragen package insert" (PDF). http://www.veregenrx.com/IL331%20Veregen%20PI%20170mm_x_420mm.pdf. Retrieved 2008-08-18.[dead link]
- ^ CDC. (2004). REPORT TO CONGRESS: Prevention of Genital Human Papillomavirus Infection.
External links
- [1]-warts Condyloma] - [Genital Warts]
- Genital warts at the Open Directory Project
- HPV information from the US CDC
- CDC Report to Congress
- An evidence-based review of medical and surgical treatments of genital warts. Noah Scheinfeld and Daniel S Lehman. Dermatology Online Journal (2006) 12 (3): 5.
Sexually transmitted diseases and infections (STD/STI) (primarily A50–A64, 090–099) Bacterial Protozoal Parasitic Viral AIDS (HIV-1/HIV-2) · Cervical cancer, vulvar cancer & Genital warts (condyloma), Penile cancer, Anal cancer (Human papillomavirus (HPV)) · Hepatitis B (Hepatitis B virus) · Herpes simplex (HSV1/HSV2) · Molluscum contagiosum (MCV)General
inflammationInfectious skin disease: Viral cutaneous conditions, including viral exanthema (B00–B09, 050–059) Ungrouped unknown/multiple: Asymmetric periflexural exanthem of childhood · Post-vaccination follicular eruption · Lipschütz ulcer · Eruptive pseudoangiomatosis · Viral-associated trichodysplasia · Gianotti–Crosti syndromePapillomavirus – Human papillomavirus Related
diseasesCervical cancer ·
Warts (genital, plantar, flat, Laryngeal papillomatosis), Epidermodysplasia verruciformis, Focal epithelial hyperplasia, Papilloma
Factor in other cancers (Anal, Vaginal, Vulvar, Penile, Head and neck cancer (HPV-positive oropharyngeal cancer)) ·Vaccine Screening Pap test (stain) - Cytopathology/Cytotechnology results Bethesda System
Experimental techniques (Speculoscopy, Cervicography)Colposcopy Biopsy histology Treatment History Categories:- Infections with a predominantly sexual mode of transmission
- Papillomavirus
- Viral diseases
- Virus-related cutaneous conditions
Wikimedia Foundation. 2010.