- Minoxidil
-
Minoxidil 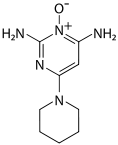
Systematic (IUPAC) name 6-piperidin-1-ylpyrimidine-2,4-diamine 3-oxide Clinical data Trade names Rogaine AHFS/Drugs.com monograph Pregnancy cat. C Legal status P(UK) for topical use, otherwise POM. Cannot be prescribed on the NHS. Routes Oral / topical Pharmacokinetic data Metabolism Primarily hepatic Half-life 4.2 hours Excretion renal Identifiers CAS number 38304-91-5 
ATC code C02DC01 D11AX01 PubChem CID 4201 DrugBank DB00350 ChemSpider 10438564 
UNII 5965120SH1 
KEGG D00418 
ChEBI CHEBI:6942 
ChEMBL CHEMBL802 
Chemical data Formula C9H15N5O Mol. mass 209.251 g/mol  (what is this?) (verify)
(what is this?) (verify)Minoxidil is an antihypertensive vasodilator medication which also slows or stops hair loss and promotes hair regrowth. Now off-patent, it is available over-the-counter for the treatment of androgenic alopecia. Minoxidil must be used indefinitely for continued support of existing hair follicles and the maintenance of any experienced hair regrowth. It is marketed under many trade names, including Rogaine/Regaine, Vanarex,[1] , Mintop and Loniten (oral), and Avacor Physician's Formulation (Avacor also produce a number of hair loss treatments that do not include minoxidil).
Contents
History
Minoxidil was first used exclusively as an oral drug (with the trade name 'Loniten') to treat high blood pressure. However, it was discovered to have an interesting side effect:[2] Minoxidil may cause increased growth or darkening of fine body hairs, or in some cases, significant hair growth. When the medication is discontinued, the hair loss will return to normal rate within 30 to 60 days. Upjohn Corporation produced a topical solution that contained 2% minoxidil to be used to treat baldness and hair loss, under the brand name Rogaine in the United States and Canada, and Regaine in Europe and the Asia-Pacific. Treatments usually include a 5% concentration solution that is designed for men, whereas the 2% concentration solutions are designed for women. The patent on minoxidil expired February 11, 1996.[3] While the drug is available in the United Kingdom, it cannot be prescribed on the NHS, so patients must either buy it over-the-counter or have a private prescription for it.[4]
In 2007, a new foam-based formulation of 5% minoxidil, which is claimed to be as effective as the liquid-based formulation, was introduced by Upjohn and marketed under the brand name Rogaine/Regaine Foam.[5]
Mechanism
The mechanism by which minoxidil promotes hair growth is not fully understood. Minoxidil contains the nitric oxide chemical moiety and may act as a nitric oxide agonist.[6] Similarly, minoxidil is a potassium channel opener, causing hyperpolarization of cell membranes. Minoxidil is less effective when there is a large area of hair loss. In addition, its effectiveness has largely been demonstrated in younger men who have experienced hair loss for less than 5 years. Minoxidil use is indicated for central (vertex) hair loss only.[7] Minoxidil is also a vasodilator.[8] It is speculated that by widening blood vessels and opening potassium channels, it allows more oxygen, blood, and nutrients to the follicle. This may cause follicles in the telogen phase to shed, which are then replaced by thicker hairs in a new anagen phase.
Side effects
Common side effects of minoxidil include burning or irritation of the eye, itching, redness or irritation at the treated area, as well as unwanted hair growth elsewhere on the body. Users should stop treatment and seek medical attention right away if they experience any of the following serious side effects: Severe allergic reactions (rash; hives; itching; difficulty breathing; tightness in the chest; swelling of the mouth, face, lips, or tongue); chest pain; dizziness; fainting; fast heartbeat; sudden, unexplained weight gain; swollen hands or feet.[9]
Alcohol present in topical preparations may dry the scalp, resulting in dandruff.[10] Side effects of oral minoxidil may include swelling of the face and extremities, rapid and irregular heartbeat, lightheadedness, cardiac lesions, and focal necrosis of the papillary muscle and subendocardial areas of the left ventricle.[11] There have been cases of allergic reactions to minoxidil or the non-active ingredient propylene glycol, which is found in some topical minoxidil formulations. Pseudoacromegaly is an extremely rare side effect reported with large doses of oral minoxidil.[12]
Paradoxically, hair loss is a common side effect of minoxidil treatment.[13] Manufacturers note that minoxidil-induced hair loss is a common side effect and describe the process as "shedding". Although this phenomenon demonstrates that minoxidil is indeed affecting hair follicles, manufacturers offer no guarantee that the new hair loss will be replaced with hair growth. The speculated reason for this shedding is the encouragement of hairs already in the telogen phase to shed early, before often beginning a fresh, healthier anagen phase.
Minoxidil is highly toxic to cats and may cause death with inadvertent skin contact.[14][15]
Application
Minoxidil needs to be applied once or the recommended twice daily, and must be used indefinitely for continued support of existing hair follicles and the maintenance of any experienced hair regrowth. To achieve maximum effect, the solution should be in contact with the scalp for at least 4 hours before allowing hair to get wet.[16] Minoxidil stimulates hair follicles and growth, but does not reduce DHT or the enzyme responsible for its accumulation around the hair follicle, 5-alpha reductase, which is the primary mediator of male pattern baldness in genetically susceptible individuals.[17] Therefore, when treatment is stopped, the DHT has its expected effect of shrinking and ultimately destroying the genetically predisposed hair follicles.
Minoxidil solutions are sold under many brand names. Many high priced as well as generic brands of minoxidil regrowth solutions exist and do not differ in their active ingredient or concentration. Minoxidil products involve contact with hair strands which may cause problems with hair styling as minoxidil must be in an alcohol-based solution. If the solution does not pose any issues, hair styling products may be applied after it has dried.
References
- ^ Vanarex | Official website
- ^ MINOXIDIL - ORAL (Loniten) side effects, medical uses, and drug interactions | MedicineNet
- ^ "Grant v. Pharmacia & Upjohn Co.". http://ca10.washburnlaw.edu/cases/2002/12/01-1509.htm. Retrieved 2009-01-17.
- ^ Drug Tariff, retrieved 2009/04/11.
- ^ Olsen EA, Whiting D, Bergfeld W, Miller J, Hordinsky M, Wanser R, Zhang P, Kohut B (2007). "A multicenter, randomized, placebo-controlled, double-blind clinical trial of a novel formulation of 5% minoxidil topical foam versus placebo in the treatment of androgenetic alopecia in men". J Am Acad Dermatol. 57 (5): 767–74. doi:10.1016/j.jaad.2007.04.012. PMID 17761356.
- ^ Alopecia & Free Radical "Redox" Signaling | Nitric Oxide and Superoxide | drproctor.com
- ^ Medical Treatments for Balding in Men American Family Physician April 15, 1999
- ^ Vasodilators | MayoClinic.com
- ^ Rogaine Side Effects | Drugs.com
- ^ "Dandruff and Seborrheic Dermatitis". Medscape.com. http://www.medscape.com/viewarticle/407641. Retrieved 2009-10-09.
- ^ "Minoxidil Official FDA information, side effects and uses". Drugs.com. http://www.drugs.com/pro/minoxidil.html.
- ^ Nguyen, K.; Marks Jr, J. (2003). "Pseudoacromegaly induced by the long-term use of minoxidil". Journal of the American Academy of Dermatology 48 (6): 962–965. doi:10.1067/mjd.2003.325. PMID 12789195.
- ^ FAQs | Rogaine
- ^ Camille DeClementi; Keith L. Bailey, Spencer C. Goldstein, and Michael Scott Orser (December 2004). "Suspected toxicosis after topical administration of minoxidil in 2 cats". Journal of Veterinary Emergency and Critical Care 14 (4): 287–292. doi:10.1111/j.1476-4431.2004.04014.x.
- ^ "Minoxidil Warning". ShowCatsOnline.com. http://www.showcatsonline.com/x/minoxidil.shtml. Retrieved 2007-01-18. "Very small amounts of Minoxidil can result [in] serious problems or death"
- ^ minoxidil (Rogaine) - drug class, medical uses, medication side effects, and drug interactions by MedicineNet.com
- ^ American Hair Loss Association | Men's Hair Loss | Causes of Hair Loss
External links
- ROGAINE® | Official website
- Minoxidil Topical | MedlinePlus Drug Information
- minoxidil - (min OX i dil) | Drug Guide of Brand Name and Generic Prescription Drugs | myOptumHealth
Nonsympatholytic vasodilatory antihypertensives (C02) Nitrovasodilator NO (arterioles and venules)N (arteriolar)(arteriolar) Potassium channel opener Minoxidil • Diazoxide(arteriolar) Calcium channel blocker see listOther dermatological preparations (D11) Medicated shampoos Cetrimide • Cadmium compounds • Selenium compounds • Povidone-iodine • Sulfur compounds • XenysalateOther dermatologicals Hair growth inhibitorsOtherCategories:- Amine oxides
- Piperidines
- Pyrimidines
- Vasodilators
- Potassium channel openers
Wikimedia Foundation. 2010.
