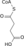
- Malic acid
-
Not to be confused with maleic acid or malonic acid."Malate" redirects here. For the district in Manila, see Malate, Manila.
Malic acid 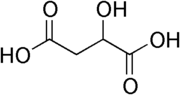
 hydroxybutanedioic acidOther namesL-Malic acid
hydroxybutanedioic acidOther namesL-Malic acid
D-Malic acid
(-)-Malic acid
(+)-Malic acid
(S)-Hydroxybutanedioic acid
(R)-Hydroxybutanedioic acidIdentifiers CAS number 6915-15-7 
PubChem 525 ChemSpider 510  , 83793 D-(+)-malic acid
, 83793 D-(+)-malic acid  , 193317 L-(-)-malic acid
, 193317 L-(-)-malic acid 
UNII 817L1N4CKP 
EC number 230-022-8 KEGG D04843 
ChEBI CHEBI:6650 
Jmol-3D images Image 1 - O=C(O)CC(O)C(=O)O
Properties Molecular formula C4H6O5 Molar mass 134.09 g mol−1 Density 1.609 g cm−3 Melting point 130 °C, 403 K, 266 °F
Solubility in water 558 g/L (at 20 °C)[1] Acidity (pKa) pKa1 = 3.40, pKa2 = 5.20 [2] Related compounds Other anions malate Related carboxylic acids succinic acid
tartaric acid
fumaric acidRelated compounds butanol
butyraldehyde
crotonaldehyde
sodium malate acid (verify) (what is:
acid (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Malic acid is an organic compound with the formula HO2CCH2CHOHCO2H. It is a dicarboxylic acid which is made by all living organisms, contributes to the pleasantly sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms (L- and D-enantiomers), though only the L-isomer exists naturally. The salts and esters of malic acid are known as malates. The malate anion is an intermediate in the citric acid cycle.
Contents
Biochemistry
L-Malic acid is the naturally occurring form, whereas a mixture of L- and D-malic acid is produced synthetically.
Malate plays an important role in biochemistry. In the C4 carbon fixation process, malate is a source of CO2 in the Calvin cycle. In the citric acid cycle, (S)-malate is an intermediate, formed by the addition of an -OH group on the si face of fumarate. It can also be formed from pyruvate via anaplerotic reactions.
Malate is also synthesized by the carboxylation of phosphoenolpyruvate in the guard cells of plant leaves. Malate, as a double anion, often accompanies potassium cations during the uptake of solutes into the guard cells in order to maintain electrical balance in the cell. The accumulation of these solutes within the guard cell decreases the solute potential, allowing water to enter the cell and promote aperture of the stomata.
Malic acid in food
Malic acid was first isolated from apple juice by Carl Wilhelm Scheele in 1785. Antoine Lavoisier in 1787 proposed the name acide malique which is derived from the Latin word for apple, mālum.[3] Malic acid contributes to the sourness of green apples. It is present in grapes and in most wines with concentrations sometimes as high as 5 g/l.[4] It confers a tart taste to wine, although the amount decreases with increasing fruit ripeness. The process of malolactic fermentation converts malic acid to much milder lactic acid.
Malic acid, when added to food products, is denoted by E number E296. Malic acid is the source of extreme tartness in USA-produced confectionery, the so-called extreme candy. It is also used with or in place of the less sour citric acid in sour sweets. These sweets are sometimes labeled with a warning stating that excessive consumption can cause irritation of the mouth. It is approved for use as a food additive in the EU,[5] USA[6] and Australia and New Zealand[7] (where it is listed by its INS number 296).
Production and main reactions
Malic acid is produced industrially by the double hydration of maleic anhydride.[8]
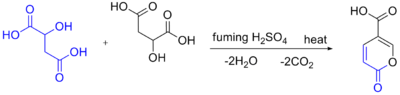
Self-condensation of malic acid with fuming sulfuric acid gives the pyrone coumalic acid:[9]
Malic acid was important in the discovery of the Walden inversion and the Walden cycle, in which (-)-malic acid first is converted into (+)-chlorosuccinic acid by action of phosphorus pentachloride. Wet silver oxide then converts the chlorine compound to (+)-malic acid, which then reacts with PCl5 to the (-)-chlorosuccinic acid. The cycle is completed when silver oxide takes this compound back to (-)-malic acid.
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles.[10]
Citric_acid_cycle edit
See also
References
- ^ chemBlink Online Database of Chemicals from Around the World
- ^ Dawson, R. M. C. et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ^ The Origin of the Names Malic, Maleic, and Malonic Acid Jensen, William B. J. Chem. Educ. 2007, 84, 924. Abstract
- ^ "Methods For Analysis of Musts and Wines", Ough and Amerine, John Wiley and Sons, 2nd Edition, 1988, page 67
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". http://www.food.gov.uk/safereating/chemsafe/additivesbranch/enumberlist. Retrieved 2011-10-27.
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part II". http://www.fda.gov/Food/FoodIngredientsPackaging/FoodAdditives/ucm191033.htm#ftnT. Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 - Labelling of ingredients". http://www.comlaw.gov.au/Details/F2011C00827. Retrieved 2011-10-27.
- ^ Karlheinz Miltenberger "Hydroxycarboxylic Acids, Aliphatic" in Ullmann's Encyclopedia of Industrial Chemistry,2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a13 507
- ^ Richard H. Wiley and Newton R. Smith (1963), "Coumalic acid", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv4p0201; Coll. Vol. 4: 201
- ^ The interactive pathway map can be edited at WikiPathways: "TCACycle_WP78". http://www.wikipathways.org/index.php/Pathway:WP78.
External links
Citric Acid Cycle Metabolic Pathway Oxaloacetate Malate Fumarate Succinate Succinyl-CoA 

Acetyl-CoA NADH + H+ NAD+ H2O FADH2 FAD CoA + ATP(GTP) Pi + ADP(GDP) 
+ H2O 
NADH + H+ + CO2 CoA NAD+ 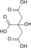
H2O 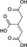
H2O 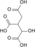
NAD(P)+ NAD(P)H + H+ 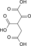
CO2 
Citrate cis-Aconitate Isocitrate Oxalosuccinate α-Ketoglutarate Categories:- Citric acid cycle
- Cellular respiration
- Citric acid cycle compounds
- Cosmetics chemicals
- Dicarboxylic acids
- Food acidity regulators
- Hydroxy acids
- Acids in wine
Wikimedia Foundation. 2010.