- Coordination polymer
-
A coordination polymer is an inorganic or organometallic polymer structure containing metal cation centers linked by ligands, extending in an array. It can also be described as a polymer whose repeat units are coordination complexes. Similar supramolecular architectures are also called Metal-organic frameworks (MOFs), and coordination networks, with some inconsistency in the distinctions between the terms.[1] (An IUPAC project was initiated in 2009 to address the terminology issues in this area and will deliver its final report in 2011.[2][3]) Coordination polymers span scientific fields such as organic and inorganic chemistry, biology, materials science, electrochemistry, and pharmacology, having many potential applications.[4] This interdisciplinary nature has led to extensive study in the past few decades.[5]
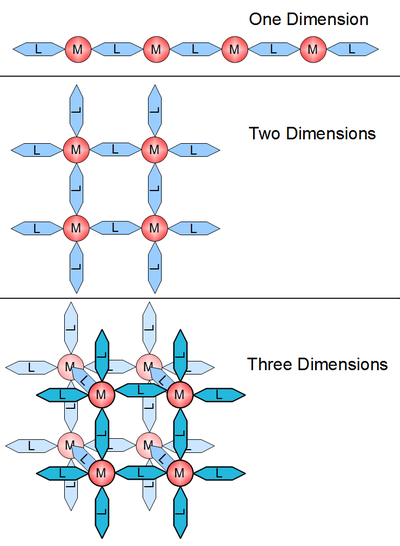
Coordination polymers can be classified in a number of different ways due to particular aspects of structure and composition. One important classification is referred to as dimensionality. A structure can be determined to be one, two or three dimensional, depending on the number of directions in space the array extends in. A one dimensional structure extends in a straight line (along the x axis); a two dimensional structure extends in a plane (two directions, x and y axes); and a three dimensional structure extends in all three directions (x, y, and z axes).[6] This is depicted in Figure 1.
Contents
History
Even though coordination polymers have experienced a research boom in the past decade or so, research in this field has been around for a long time. Alfred Werner, sometimes called the father of coordination chemistry, won the Nobel Prize for Chemistry in 1913 "for his work on the linkage of atoms in molecules, by which he has thrown new light on earlier investigations and opened up new fields of research especially in inorganic chemistry."[7] This work laid a lot of the groundwork for the future study of coordination polymers. Terms ubiquitous in the field, such as coordination number, were coined by Werner. He also determined that even neutral, stable molecules in their own right (such as ammonia or water) can behave like ligands, and enter into coordination bonds with metals.
In the past few decades, research in this field has boomed, partially because of the potential applications for metal-organic frameworks (MOF's) in areas such as hydrogen storage.
Synthesis and Propagation
Much of the mechanism for growth coordination polymers is based on self-assembly, involving crystallization of a metal salt with a ligand. This can be achieved by known crystallization and crystal engineering techniques. The mechanisms of Molecular self-assembly processes on a molecular level are currently being investigated, however it is accepted that coordination generally emerges due to the interaction of lone pairs of electrons on the ligand with the regions of low electron density on the cationic metal ion.[4] Crystals of coordination polymers are nucleated when an aggregate of coordinated ligand/metal complexes reaches a size where lowered energy of the aggregate as a whole is more significant than the added surface energy. This means that the crystal will grow more easily in terms of energy as opposed to de-aggregation. This phenomenon is known as nucleation. The nucleus is a localized, thermodynamic site for crystal growth to occur. More molecules (metals and ligands) can self-assemble onto the growing crystal in a periodic array.
For the most part, it is not possible right now to draw any direct correlations between particular structural aspects and applications.
The synthesis methods utilized to produce coordination polymers are generally the same methods used to grow any crystal. These generally include solvent layering (slow diffusion), slow evaporation, and slow cooling. (Because the main method of characterization of coordination polymers is X-ray crystallography, growing a crystal of sufficient size and quality is important.)
Intermolecular Forces and Bonding
The forces that led to spontaneous arrangement of molecules (metal-ligand complexes in this case) are thought to be van der Waals forces, pi-pi interactions, hydrogen bonding, and stabilization of pi bonds by polarized bonds in addition to the coordination bond formed between the metal and the ligand. These intermolecular forces tend to be weak, with a long equilibrium distance (bond length) compared to covalent bonds. The pi-pi interactions between benzene rings, for example, have energy roughly 5-10 kJ/mol and optimum spacing 3.4-3.8 Ångstroms between parallel faces of the rings.
Coordination
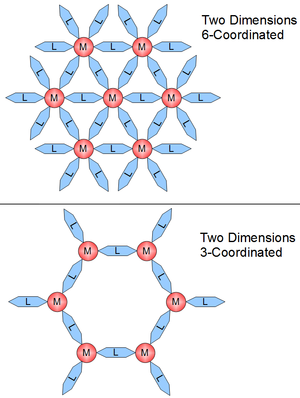
The crystal structure and dimensionality of the coordination polymer is determined by the functionality of the linker and the coordination geometry of the metal center. Dimensionality is generally driven by the metal center which can have the ability to bond to as 16 functional sites on linkers. This number of possible bonds is the coordination number. Coordination numbers are more often between 2 and 10, and the bonding sites are separated by distinct angles. Coordination numbers are shown in planer geometry in Figure 2. In Figure 1 the 1D structure is 2-coordinated, the planer is 4-coordinated, and the 3D is 6-coordinated. Figure 3 shows structures hinting at the complexity that is possible by changing coordination geometry.
Metal Centers
 Figure 3. Shown here are three coordination polymers of different dimensionality. All three were made using the same ligand (4,5-dihydroxybenzene-1,3-disulfonate (L)), but different metal cations. All of the metals come from Group 2 on the periodic table (alkaline earth metals) and in this case, dimensionality increases with cation size and polarizability. A. [Ca(L)(H2O)4]•H2O B. [Sr(L)(H2O)4]•H2O C.[Ba(L)(H2O)]•H2O[8] In each case, the metal is represented in green.
Figure 3. Shown here are three coordination polymers of different dimensionality. All three were made using the same ligand (4,5-dihydroxybenzene-1,3-disulfonate (L)), but different metal cations. All of the metals come from Group 2 on the periodic table (alkaline earth metals) and in this case, dimensionality increases with cation size and polarizability. A. [Ca(L)(H2O)4]•H2O B. [Sr(L)(H2O)4]•H2O C.[Ba(L)(H2O)]•H2O[8] In each case, the metal is represented in green.
Metal Centers, also called nodes or hubs, bond to a specific number of linkers at well defined angles. The number of linkers bound to a node is known as the coordination number, which, along with the angles they are held at, determines the dimensionality of the structure. The coordination number and coordination geometry of a metal center is determined by the nonuniform distribution of electron density around it, and in general the coordination number increases with cation size. Several models, most notably hybridization model and molecular orbital theory, use the Schrödinger equation to predict and explain coordination geometry, however this is difficult in part because of the complex effect of environment on electron density distribution.[9]
Transition Metals
Transition metals are commonly used as nodes. Partially filled d orbitals, either in the atom or ion, can hybridize differently depending on environment. This electronic structure causes some of them to exhibit multiple coordination geometries, particularly copper and gold ions which as neutral atoms have full d-orbitals in their outer shells.
Lanthanides
Lanthanides are large atoms with coordination numbers varying from 7 to 10 due to filling of f orbitals. They are strongly affected by their environment and difficult to predict, making them challenging to use as nodes. This challenge may be worth it however, because the bonds formed by multiplexing(attaching to multiple linkers) have characteristic luminescent wavelengths.
Alkali/Alkaline Earth Metals
Alkali metals and alkaline earth metals exist as stable cations. Alkali metals readily form cations with stable valence shells, giving them different coordination behavior than lanthanides and transition metals. They are strongly effected by the counter ion from the salt used in synthesis, which is difficult to avoid. The coordination polymers shown in Figure 3 are all group two metals. In this case, the dimensionality of the of these structures increases as the radius of the metal increases down the group (from calcium to strontium to barium).[10]
Ligands
In most coordination polymers, a ligand (atom or group of atoms) will formally donate a lone pair of electrons to a metal cation and form a coordination complex via a Lewis acid/ base relationship (lewis acids and bases). Coordination polymers are formed when a ligand has the ability to form multiple coordination bonds and act as a bridge between multiple metal centers. Ligands that can form one coordination bond are referred to as monodentate, but those which form multiple coordination bonds, which could lead to coordination polymers are called polydentate. Polydentate ligands are particularly important because it is through ligands that connect multiple metal centers together that an infinite array is formed. Polydentate ligands can also form multiple bonds to the same metal (which is called chelation). Monodentate ligands are also referred to as terminal because they do not offer a place for the network to continue. Often, coordination polymers will consist of a combination of poly- and monodentate, bridging, chelating, and terminal ligands.
Chemical Composition
Almost any type of atom with a lone pair of electrons can be incorporated into a ligand. Ligands that are commonly found in coordination polymers include polypyridines, phenanthrolines, hydroxyquinolines and polycarboxylates. Oxygen and nitrogen atoms are commonly encountered as binding sites, but other atoms, such as sulfur[11] and phosphorus,[12][13] have been observed.
 Figure 4. A flexible ligand in both a gauche and anti conformation.
Figure 4. A flexible ligand in both a gauche and anti conformation.
Ligands and metal cations tend to follow hard soft acid base theory (HSAB) trends. This means that larger, more polarizable soft metals will coordinate more readily with larger more polarizable soft ligands, and small, non-polarizable, hard metals coordinate to small, non-polarizable, hard ligands.
Structural Orientation
Ligands can be flexible or rigid. A rigid ligand is one that has no freedom to rotate around bonds or reorient within a structure. Flexible ligands can bend, rotate around bonds, and reorient themselves. This creates more variety in the structure. There are examples of coordination polymers that include two configurations of the same ligand within one structure,[14] as well as two separate structures where the only difference between them is ligand orientation. An example of a flexible ligand is shown in Figure 4.
Other Factors
 Figure6. The addition and removal of guest molecules can have a large effect on the resulting structure of a coordination polymer. A few examples are (top) change of a linear 1D chain to a zigzag pattern, (middle) staggered 2D sheets to stacked, and (bottom) 3D cubes become more widely spaced.
Figure6. The addition and removal of guest molecules can have a large effect on the resulting structure of a coordination polymer. A few examples are (top) change of a linear 1D chain to a zigzag pattern, (middle) staggered 2D sheets to stacked, and (bottom) 3D cubes become more widely spaced.
Counter Ion
Besides metal and ligand choice, there are many other factors that affect the structure of the coordination polymer. For example, most metal centers are positively charged ions which exist as salts. The counter ion in the salt can effect the overall structure. For example, silver salts such as AgNO3, AgBF4, AgClO4, AgPF6, AgAsF6 and AgSbF are all crystallized with the same ligand, the structures vary in terms of the coordination environment of the metal, as well as the dimensionality of the entire coordination polymer.[15]
Crystallization Environment
Additionally, variations in the crystallization environment can also change the structure. Changes in pH,[16] exposure to light, or changes in temperature[17] can all change the resulting structure. Influences on the structure based on changes in crystallization environment are determined on a case by case basis.
Guest Molecules
The structure of coordination polymers oftentimes incorporates empty space in the form of pores or channels. This empty space is thermodynamically unfavorable. In order to stabilize the structure and prevent collapse, the pores or channels are often occupied by guest molecules. Guest molecules do not form bonds with the surrounding lattice, but sometimes interact via intermolecular forces, such as hydrogen bonding or pi stacking. Most often, the guest molecule will be the solvent that the coordination polymer was crystallized in, but can really be anything (other salts present, atmospheric gases such as oxygen, nitrogen, carbon dioxide, etc.) The presence of the guest molecule can sometimes influence the structure by supporting a pore or channel, where otherwise none would exist. Figure 5 shows a schematic of a few different ways a structure can change upon the addition or evacuation of a guest molecule.
Applications
Molecular Storage
Thanks to coordination polymerization in three dimensions, pores within the structure can be made. The size and shapes of the pore can be controlled by the linker size and the connecting ligands’ length and functional groups. To modify the pore size in order to achieve effective adsorption, nonvolatile guests (host-guest chemistry) are intercalated in the porous coordination polymer space to decrease the pore size. Active surface guests can also be used contribute to adsorption. For example, the large-pore MOF-177, 11.8 Å in diameter, can be doped by C60 molecules (6.83 Å in diameter) or polymers with a highly conjugated system in order to increase the surface area for H2 adsorption. Flexible porous coordination polymers are also convenient for molecular storage, since their pore sizes can be easily altered by physical changes. And example of this might be seen in a polymer that contains gas molecules in its normal state, but upon compression the polymer collapses and releases the stored molecules. Depending on the crystal structure of the polymer, it is possible that the structure be flexible enough that collapsing the pores is reversible and the polymer can be reused to uptake the gas molecules again.[18] The Metal-organic framework page has a detailed section dealing with H2 gas storage.
Luminescence
Luminescence arises from electronic transitions from an excited state, caused by photoexcitation, to the ground state, resulting in the emission of light. Most luminescent compounds require organic chromophoric ligands, which absorb light and then pass the excitation energy to the metal ion, which is referred to as a ligand-to-metal charge-transfer process (LMCT). Coordination polymers are potentially the most versatile luminescent species due to their emission properties being coupled with guest exchange. Luminescent supramolecular architectures have recently attracted much interest because of their potential applications in optoelectronic devices or as fluorescent sensors and probes. Coordination polymers are often more stable (thermo- and solvent-resistant) than purely organic species. For ligands that fluoresce without the presence of the metal linker (not due to LMCT), the intense photoluminescence emission of these materials tend to be magnitudes of order higher than that or the free ligand alone. These materials can be used for designing potential candidates for light emitting diode (LED) devices. The dramatic increase in fluorescence is caused by the increase in rigidity and asymmetry of the ligand when coordinated to the metal center.[19]
Electrical Conductivity
Coordination polymers can have short inorganic and conjugated organic bridges in their structures, which provide pathways for electrical conduction. Some one-dimensional coordination polymers built as shown in Fig. X exhibit conductivities in a range of 1x10−6 to 2x10−1 S/cm. The conductivity is due to the interaction between the metal d-orbital and the pi* level of the bridging ligand. In some cases coordination polymers can have semi-conductor behavior. Three-dimensional structures consisting of sheets of silver-containing polymers demonstrate semi-conductivity when the metal centers are aligned, and conduction decreases as the silver atoms go from parallel to perpendicular.[19]
Magnetism
Interest in coordination polymers is continued by the application of magnetism. Antiferromagnetism, ferrimagnetism, and ferromagnetism are cooperative phenomena of the magnetic spins within a solid. They require an interaction or coupling between the spins of the paramagnetic centers. Since intramolecular interactions (such as bonding) are much more efficient than intermolecular interactions (i.e. through space), three-dimensional coordination polymers are of interest because they connect their magnetic centers through direct coordinative links, and have the possibility for displaying high critical temperatures. In order to allow efficient magnetic, metal ions should be bridged by small ligands allowing for short metal-metal contacts (such as oxo, cyano, and azido bridges).[19]
Sensor Capability
Coordination polymers can also show color changes upon the change of solvent molecules incorporated into the structure. An example of this would be the two Co coordination polymers of the [Re6S8(CN)6]4- cluster that contains water ligands that coordinate to the cobalt atoms. This originally orange solution turns either purple or green with the replacement of water with tetrahydrofuran, and blue upon the addition of diethyl ether. The polymer can thus act as a solvent sensor that physically changes color in the presence of certain solvents. The color changes are attributed to the incoming solvent displacing the water ligands on the cobalt atoms, resulting in a change of their geometry from octahedral to tetrahedral.[19]
References
- ^ Carlucci, Lucia; Gianfranco Ciani, Davide M. Proserpio (1999). "Coordination polymers and supramolecular architectures". http://dcssi.istm.cnr.it/1995-1999/coordination_polymers.htm. Retrieved 26 March 2010.
- ^ IUPAC Current Projects. May 09, 2010. Retrieved on May 09, 2010.
- ^ CP and MOF Project. May 09, 2010. Retrieved on May 09, 2010.
- ^ a b Fromm, K. (2008). "Coordination polymer networks with s-block metal ions". Coord. Chem. Rev. 252: 856–885. doi:10.1016/j.ccr.2007.10.032.
- ^ You, X. (2006). "Synthesis, structures and properties of alkaline earth metal benzene-1,4-dioxylacetates with three-dimensional hybrid networks". Coord. Chem. Rev. (Elsevier) 359: 3257–3263.
- ^ Chen, X; Ye, B., Tong, M. (2005). "Metal-organic molecular architectures with 2,2′-bipyridyl-like and carboxylate ligands". Coord. Chem. Rev (Elsevier) 249: 545–565. doi:10.1016/j.ccr.2004.07.006.
- ^ "Nobel Prize in Chemistry 1913: Presentation Speech". Amsterdam: Elsevier Publishing Company. 1996. http://www.angelfire.com/ms3/my-page/www/press-werner.html. Retrieved 29 March 2010.
- ^ Cote, A; Shimizu, G. (2003). "Coordination Solids via Assembly of Adaptable Components : Systematic Structural Variation in Alkaline Earth Organosulfonate Networks". Chem. Eur. J. (Wiley) 9: 5361–5370. doi:10.1002/chem.200305102.
- ^ Bernstein, Jeremy; Paul M. Fishbane, Stephen G. Gasiorowicz (April 3, 2000). Modern Physics. Prentice-Hall. pp. 624. ISBN 978-0139553110.
- ^ alkaline earth
- ^ Yan, S.; et. al. (2001). "Silver(I) coordination polymers of cyclic sulfur ligand, 2,2′,3,3′-tetrahydro-4,4′-dithia-1,1′-binaphthylidene". Inorg. Chim. Acta 322: 133–137. doi:10.1016/S0020-1693(01)00556-4.
- ^ Hung-Low, F; Gary, J.B., Klausmeyer, K. (2009). "Effect of anion and ligand ratio in self-assembled silver(I) complexes of 4-(diphenylphosphinomethyl)pyridine and their derivatives with bipyridine ligands". Inorg. Chim. Acta. 20: 426–436.
- ^ Leone, G; Ricci, G., Sommazzi, A., Massi, F., Ricci, M., Boglia, A. (2009). "Well-defined transition metal complexes with phosphorus and nitrogen ligands for 1,3-dienes polymerization". Coord. Chem. Rev. 254: 661–676. doi:10.1016/j.ccr.2009.09.023.
- ^ Keller, S.; Knaust, J. (2002). "A Mixed-Ligand Coordination Polymer from the in Situ, Cu(I)-Mediated Isomerization of Bis(4-pyridyl)ethylene". Inorg. (ACS) 41: 5650–5652. doi:10.1021/ic025836c.
- ^ Rizzato, Silvia; Carlucci, Ciani, Proserpio (2002). "New polymeric networks from the self-assembly of silver(I) salts and the flexible ligand 1,3-bis(4-pyridyl)propane (bpp). A systematic investigation of the effects of the counter ions and a survey of the coordination polymers based on bpp". CrystEngComm (RCS) 4 (22): 121–129. doi:10.1039/b201288j.
- ^ Zhou, Z.; et. al. (2009). "pH- and mol-ratio dependent formation of zinc(II) coordination polymers with iminodiacetic acid: Synthesis, spectroscopic, crystal structure and thermal studies". J. Of Sol. Stat. Chem. 182: 2698–2706. doi:10.1016/j.jssc.2009.06.042.
- ^ Ng, S.; et. al., (2005). "Supramolecular Isomerism in Cadmium Hydroxide Phases. Temperature-Dependent Synthesis and Structure of Photoluminescent Coordination Polymers of α- and β-Cd2(OH)2(2,4-pyda)". Cryst. Grow. & Des. 5: 837–839. doi:10.1021/cg049610r.
- ^ Bureekaew, Sareeya; et. al., (2007). "Chemistry and Application of Flexible Porous Coordination Polymers". Sci. Technol. Adv. Mater. 9: 2–4. doi:10.1088/1468-6996/9/1/014108.
- ^ a b c d Stuart R. Batten (2008). Coordination Polymers: Design, Analysis and Application. RSC Publishing. pp. 297–307, 396–407.
Categories:- Polymers
Wikimedia Foundation. 2010.