- Cresol
-
Cresols are organic compounds which are methylphenols. They are a widely occurring natural and manufactured group of aromatic organic compounds which are categorized as phenols (sometimes called phenolics). Depending on the temperature, cresols can be solid or liquid because they have melting points not far from room temperature. Like other types of phenols, they are slowly oxidized by long exposure to air and the impurities often give cresols a yellowish to brownish red tint. Cresols have an odor characteristic to that of other simple phenols, reminiscent to some of a "coal tar" smell.
Contents
Chemical structure
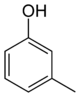
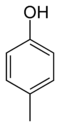
In its chemical structure, a cresol molecule has a methyl group substituted onto the benzene ring of a phenol molecule. There are three forms of cresols that are only slightly different in their chemical structure and physical properties: ortho-cresol (o-cresol), meta-cresol (m-cresol), and para-cresol (p-cresol). These forms occur separately or as a mixture. The word tricresol can be used as a synonym for cresol where it means a mixture of o-, m- and p-cresols.
Isomers of Cresol Skeletal formula 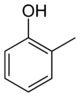
Ball-and-stick model 
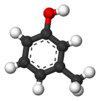
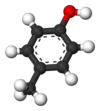
General Common name o-cresol m-cresol p-cresol Systematic name 2-methylphenol 3-methylphenol 4-methylphenol Other names ortho-cresol meta-cresol para-cresol Molecular formula C7H8O SMILES Oc1c(C)cccc1 Oc1cc(C)ccc1 Oc1ccc(C)cc1 Molar mass 108.14 g/mol Appearance at room
temperature and pressurecolorless crystals thicker liquid greasy-looking solid CAS number [95-48-7] [108-39-4] [106-44-5] mixture of cresols (tricresol): [1319-77-3] Properties Density and phase 1.05 g/cm3, solid 1.03 g/cm3, liquid 1.02 g/cm3, liquid Solubility in pure water
at 20-25°C2.5 g/100 ml 2.4 g/100 ml 1.9 g/100 ml soluble in strongly alkaline water Melting point 29.8 °C (303.0 K) 11.8 °C (285.0 K) 35.5 °C (309.7 K) Boiling point 191.0 °C (464.2 K) 202.0 °C (475.2 K) 201.9 °C (475.1 K) Acidity (pKa) 10.26 10.09 10.26 Viscosity solid at 25 °C ? cP at 25 °C solid at 25 °C Structure Dipole moment 1.35 D 1.61 D 1.58 D Hazards MSDS External MSDS ? ? Main hazards flammable, ingestion and inhalation toxicity hazard Flash point 81°C c.c. 86 °C 86 °C c.c. R/S statement R24/25-R34 ((S1/2)-)S36/37/S39-S45 RTECS number ? ? ? Supplementary data page Structure & properties n, εr, etc. Thermodynamic data Phase behaviour
Solid, liquid, gasSpectral data UV, IR, NMR, MS Related compounds Related phenols phenol, xylenols Related compounds bromo cresol Except where noted otherwise, data are given for
materials in their standard state (at 25°C, 100 kPa)
Infobox disclaimer and referencesApplications
Cresols are used to dissolve other chemicals, as disinfectants and deodorizers, and to make specific chemicals that kill insect pests.
Cresol solutions are used as household cleaners and disinfectants, perhaps most famously under the trade name Lysol. Cresol solutions can also be found in photographic developers. In the past, cresol solutions have been used as antiseptics in surgery, but they have been largely displaced in this role by less toxic compounds. Lysol was also advertised as a disinfecting vaginal douche in mid-twentieth century America.[1]
Cresols are found in many foods and in wood and tobacco smoke, crude oil, coal tar, and in brown mixtures such as creosote, cresolene and cresylic acids, which are wood preservatives. Small organisms in soil and water produce cresols when they break down materials in the environment. Cresols are also a chemical component found in Sharpie Markers.
Xylenols are dimethylphenols, or they can be thought of as methylcresols.
p-cresol is one of the very few compounds to attract the orchid bee Euglossa cyanura, and has been used to capture and study the species.[2]
Some types of insulin solution contain metacresol. "Each milliliter of Humalog injection contains ... 3.15mg Metacresol" Also, metacresol hydrophobic structure has been shown to interact with plastic and tubing used in drug administration products such as PVC IV bags and catheters. More research is being done to as how this interaction affects specific drug pharmacological profile at some research institutions in Colorado.
In chemistry, cresols can be used to synthesise toluene. When cresol (o-, m-, p-) is distilled with zinc dust, toluene is obtained.
Commercial examples
p-cresol derivatives include:
- Bupranolol, non-selective beta blocker
- Indo-1, popular calcium indicator
- Butylated hydroxytoluene, common antioxidant
o-cresol derivatives include include Fluo-4, a green-fluorescent calcium indicator and the herbicides:
- MCPA (4-Chloro-2-methylphenoxy)acetic acid
- Mecoprop (RS)-2-(4-chloro-2-methylphenoxy)propanoic acid
- MCPB 4-(4-chloro-2-methylphenoxy)butanoic acid
- the amine Atomoxetine, (3 R)-N-methyl-3-(2-methylphenoxy)-3-phenylpropan-1-amine
- the diol Mephenesin 3-(2-methylphenoxy)propane-1,2-diol
m-cresol derivatives include the following:
- Tolimidone, 5-(3-methylphenoxy)pyrimidin-2(1H)-one
- Bevantolol, (RS)-[2-(3,4-dimethoxyphenyl)ethyl][2-hydroxy-3-(3-methylphenoxy)propyl]amine
Health effects
Most exposures to cresols are at very low levels that are not harmful. When cresols are breathed, ingested, or applied to the skin at very high levels, they can be very harmful. Effects observed in people include irritation and burning of skin, eyes, mouth, and throat; abdominal pain and vomiting; heart damage; anemia; liver and kidney damage; facial paralysis; coma; and death.
Breathing high levels of cresols for a short time results in irritation of the nose and throat. Aside from these effects, very little is known about the effects of breathing cresols, for example, at lower levels over longer times.
Ingesting high levels results in kidney problems, mouth and throat burns, abdominal pain, vomiting, and effects on the blood and nervous system.
Skin contact with high levels of cresols can burn the skin and damage the kidneys, liver, blood, brain, and lungs.
Short-term and long-term studies with animals have shown similar effects from exposure to cresols. No human or animal studies have shown harmful effects from cresols on reproduction.
It is not known what the effects are from long-term ingestion or skin contact with low levels of cresols.
References for Table of Properties
Footnotes
- ^ "Lysol Douche Advertisements"
- ^ Williams & Whitten, 1983: 387
References
- Williams, Norris H. & Whitten, W. Mark (1983): Orchid floral fragrances and male euglossine bees: methods and advances in the last sesquidecade. Biol. Bull. 164: 355-395.
Categories:- Antiseptics
- Hazardous air pollutants
- Cresols
Wikimedia Foundation. 2010.
