- Methoxsalen
-
Methoxsalen 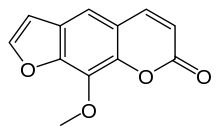
Systematic (IUPAC) name 9-methoxy-7H-furo[3,2-g]chromen-7-one Clinical data AHFS/Drugs.com Consumer Drug Information Pregnancy cat. ? Legal status ? Pharmacokinetic data Half-life ~2 hours Identifiers CAS number 298-81-7 
ATC code D05AD02 D05BA02 PubChem CID 4114 DrugBank APRD00157 ChemSpider 3971 
UNII U4VJ29L7BQ 
KEGG D00139 
ChEBI CHEBI:18358 
ChEMBL CHEMBL416 
Chemical data Formula C12H8O4 Mol. mass 216.19 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Methoxsalen (also called xanthotoxin, marketed under the trade names Oxsoralen, Deltasoralen, Meladinine) is a drug used to treat psoriasis, eczema, vitiligo, and some cutaneous Lymphomas in conjunction with exposing the skin to sunlight. Methoxsalen is extracted from Ammi majus, a plant of the family Apiaceae. Methoxsalen modifies the way skin cells receive the UVA radiation, allegedly clearing up the disease. The dosage comes in 10 mg tablets, which are taken in the amount of 30 mg 75 minutes before a PUVA (psoralen + UVA) light treatment. The substance is also present in other Apiaceae as well as Rutaceae, for example bergamot oil which is used in many perfumes and aromatherapy oils.
Chemically, methoxsalen belongs to a class of organic natural molecules known as furanocoumarins. They consist of coumarin annulated with furan.
Contents
Biosynthesis
The biosynthetic pathway is a combination of the shikimate pathway, which produces umbelliferone, and the mevalonate pathway.
Synthesis of umbelliferone
Umbelliferone is a phenylpropanoid and as such is synthesized from L-phenylalanine, which in turn is produced via the shikimate pathway. Phenylalanine is lysated into cinnamic acid, followed by hydroxylation by cinnamate 4-hydroxylase to yield 4-coumaric acid. The 4-coumaric acid is again hydroxylated by cinnamate/coumarate 2-hydroxylase to yield 2,4-dihydroxy-cinnamic acid followed by a bond rotation of the unsaturated bond adjacent to the carboxylic acid group. Finally an intramolecular attack from the hydroxyl group of C2' to the carboxylic acid group closes the ring and forms the lactone umbelliferone.
Synthesis of methoxsalen
The biosynthetic route then continues with the activation of dimethylallyl pyrophosphate (DMAPP), produced via the mevalonate pathway, to form a carbo-cation via the cleavage of the diphosphates. Once activated, the enzyme umbelliferone 6-prenyltransferase catalyzes a C-alkylation between DMAPP and umbelliferone at the activated position ortho to the phenol, yielding demethylsuberosin. This is then followed by a hydroxylation catalyzed by the enzyme marmesin synthase to yield marmesin. Another hydroxylation is catalyzed by psoralen synthase to yield psoralen. A third hydroxylation by the enzyme psoralen 8-monooxygenase yields xanthotoxol which is followed by a methylation via the enzyme xanthotoxol O-methyltransferase and S-adenosyl methionine to yield methoxsalen.[1]
Risks and side effects
Patients with high blood pressure or a history of liver problems are at risk for inflammation and irreparable damage to both liver and skin. The eyes must be protected from UVA radiation. Side effects include nausea, headaches, dizziness, and in rare cases insomnia.
Cultural aspects
Author John Howard Griffin used the chemical to darken his skin in order to investigate racial segregation in the south. He wrote the non-fiction book Black Like Me about his experiences.[2]
Some cologne/perfume contains bergamot oil as one of the ingredients (for the scent). People wearing the concoction on places where the skin receives UV radiation was radiated by the sun noticed that their skin turned brownish at those spots (due to the phototoxic effects of methoxsalen present in the bergamot oil[3]). Most modern formulations of perfumes containing bergamot are de-methoxsalenised.
References
- ^ Dewick, P. M. (2009). Medicinal Natural Products: A Biosynthetic Approach (3rd ed.). John Wiley & Sons. pp. 161, 164–165.
- ^ Dead Like Me on snopes.com
- ^ "Bergamot". oilsandplants.com. http://www.oilsandplants.com/bergamot.htm. Retrieved 2010-12-29.
Antipsoriatics (D05) Topical TarsTrioxysalen • MethoxsalenOtherSystemic PsoralensTrioxysalen • Methoxsalen • BergaptenAglycones glycosides Aesculin | Skimmin (7-O-β-D-glucopyranosylumbelliferone) | ScopolinFuran derivatives Furanocoumarins (Angelicin | Apterin | Bergamottin | Bergapten | Imperatorin | Marmesin | Methoxsalen | Psoralen | Trioxsalen) | FuranochromonesMonoterpene coumarin ether Synthetic or drugs Categories:- Photosensitizing agents
- IARC Group 1 carcinogens
- Furanocoumarins
- Phenol ethers
Wikimedia Foundation. 2010.