- Potassium sulfate
-
Potassium sulfate 
Arcanite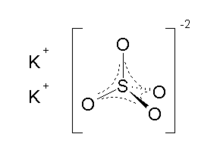 Other namesPotassium sulphate
Other namesPotassium sulphateIdentifiers CAS number 7778-80-5 
PubChem 24507 ChemSpider 22915 
UNII 1K573LC5TV 
KEGG D01726 
ChEBI CHEBI:32036 ChEMBL CHEMBL1201793 RTECS number TT5900000 Jmol-3D images Image 1 - [K+].[K+].[O-]S([O-])(=O)=O
Properties Molecular formula K2SO4 Molar mass 174.259 g/mol Appearance White solid Density 2.66 g/cm3[1] Melting point 1069 °C, 1342 K, 1956 °F
Boiling point 1689 °C, 1962 K, 3072 °F
Solubility in water 111 g/L (20 °C)
120 g/L (25 °C)
240 g/L (100 °C)Solubility slightly soluble in glycerol
insoluble in acetone, alcohol, CS2Structure Crystal structure orthorhombic Hazards MSDS External MSDS EU Index Not listed R-phrases R22 S-phrases S36 Main hazards Irritant Flash point Non-flammable LD50 6600 mg/kg Related compounds Other anions Potassium selenate
Potassium tellurateOther cations Lithium sulfate
Sodium sulfate
Rubidium sulfate
Caesium sulfateRelated compounds Potassium hydrogen sulfate
Potassium sulfite
Potassium bisulfite
Potassium persulfate sulfate (verify) (what is:
sulfate (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Potassium sulfate (K2SO4) (in British English potassium sulphate, also called sulphate of potash, arcanite, or archaically known as potash of sulfur) is a non-flammable white crystalline salt which is soluble in water. The chemical is commonly used in fertilizers, providing both potassium and sulfur.
Contents
History
Potassium sulfate (K2SO4) has been known since early in the 14th century, and it was studied by Glauber, Boyle and Tachenius. In the 17th century, it was named arcanuni or sal duplicatum, as it was a combination of an acid salt with an alkaline salt. It was also known as vitriolic tartar and Glaser's salt or sal polychrestum Glaseri after the pharmaceutical chemist Christopher Glaser who prepared it and used medicinally.[2][3]
Natural resources
The mineral form of potassium sulfate, arcanite, is relatively rare. Natural resources of potassium sulfate are minerals abundant in the Stassfurt salt. These are cocrystalisations of potassium sulfate and sulfates of magnesium calcium and sodium.
The minerals are:
- Kainite, MgSO4·KCl·H2O
- Schönite, K2SO4·MgSO4·6H2O
- Leonite, K2SO4·MgSO4·4H2O
- Langbeinite, K2SO4·2MgSO4
- Glaserite, K3Na(SO4)2
- Polyhalite, K2SO4·MgSO4·2CaSO4·2H2O
From some of the minerals like kainite, the potassium sulfate can be separated, because the corresponding salt is less soluble in water.
Kieserite, MgSO4·2H2O, can be combined with a solution of potassium chloride to produce potassium sulfate.
Manufacture
The process for manufacturing potassium sulfate is similar to that used for the manufacture of sodium sulfate.
Potassium sulfate can be synthesised by reaction of potassium chloride with sulfuric acid according to the Leblanc process. Potassium sulfate is produced according to the following reaction:
- 2 KCl + H2SO4 → 2 HCl + K2SO4
The Hargreaves process uses sulfur dioxide, oxygen and water and potassium chloride as the starting materials to produce potassium sulfate. Hydrochloric acid evaporates off. SO2 is produced through the burning of sulfur.
Properties
The anhydrous crystals form a double six-sided pyramid, but are in fact classified as rhombic. They are transparent, very hard and have a bitter, salty taste. The salt is soluble in water, but insoluble in solutions of potassium hydroxide (sp. gr. 1.35), or in absolute ethanol. It melts at 1067 °C.[4]
Uses
The principal use of potassium sulfate is as a fertilizer. K2SO4 does not contain chloride, which can be harmful to some crops. Potassium sulfate is preferred for these crops, which include tobacco and some fruits and vegetables. Crops that are less sensitive may still require potassium sulfate for optimal growth if the soil accumulates chloride from irrigation water.[5]
The crude salt is also used occasionally in the manufacture of glass. Potassium sulfate is also used as a flash reducer in artillery propellant charges. It reduces muzzle flash, flareback and blast overpressure.
Potassium hydrogen sulfate
Potassium hydrogen sulfate or bisulfate, KHSO4, is readily produced by mixing K2SO4 with an equivalent number of moles of sulfuric acid. It forms rhombic pyramids, which melt at 197 °C. It dissolves in three parts of water at 0 °C. The solution behaves much as if its two congeners, K2SO4 and H2SO4, were present side by side of each other uncombined; an excess of ethanol the precipitates normal sulfate (with little bisulfate) with excess acid remaining.
The behavior of the fused dry salt is similar when heated to several hundred degrees; it acts on silicates, titanates, etc., the same way as sulfuric acid that is heated beyond its natural boiling point does. Hence it is frequently used in analytical chemistry as a disintegrating agent. For information about other salts that contain sulfate, see sulfate.
References
- ^ Patnaik, Pradyot (2002). Handbook of Inorganic Chemicals. McGraw-Hill. ISBN 0070494398.
- ^ De Milt, Clara (1942). "Christopher Glaser". Journal of Chemical Education 19 (2): 53. doi:10.1021/ed019p53.
- ^ Klooster, van (1959). "Three centuries of Rochelle salt". Journal of Chemical Education 36 (7): 314. doi:10.1021/ed036p314.
- ^ Windholtz, M(Ed.)& Budavari, S (Ed). (1983). The Merck Index, Rahay: Merck & Co.
- ^ Fertilizer manual (3rd ed. ed.). Dordrecht: Kluwer academic publ.. 1998. pp. 615. ISBN 0-7923-5032-4.
See also
- Arcanum duplicatum, the term for this substance in alchemy
Potassium compounds KBr · KBrO3 · KCN · KCNO · KCl · KClO3 · KClO4 · KF · KH · KHCO2 · KHCO3 · KHF2 · KHSO3 · KHSO4 · KH2AsO4 · KI · KIO3 · KIO4 · KMnO4 · KN3 · KNO2 · KNO3 · KOCN · KOH · KO2 · KPF6 · KSCN · K2CO3 · K2CrO4 · K2Cr2O7 · K2FeO4 · K2MnO4 · K2O · K2O2 · K2PtCl4 · K2PtCl6 · K2S · K2SO3 · K2SO4 · K2SO5 · K2S2O5 · K2S2O7 · K2S2O8 · K2SiO3 · K3[Fe(CN)6] · K3[Fe(C2O4)3] · K4[Fe(CN)6] · K3PO4 · Categories:
- Potassium compounds
- Sulfates
- Fertilizers
Wikimedia Foundation. 2010.
Look at other dictionaries:
potassium sulfate — n a white crystalline compound K2SO4 which has been used medicinally as a cathartic … Medical dictionary
potassium sulfate — n. a white, crystalline solid, K2SO4, used in fertilizers, medicine, etc … English World dictionary
potassium sulfate — kalio sulfatas statusas T sritis chemija formulė K₂SO₄ atitikmenys: angl. normal potassium sulfate; potassium sulfate; salt of Lemery rus. калий сернокислый; калия сульфат ryšiai: sinonimas – dikalio tetraoksosulfatas … Chemijos terminų aiškinamasis žodynas
potassium sulfate — noun : either of two crystalline sulfates of potassium: a. : the normal salt K2SO4 occurring naturally especially in complex sulfates (as langbeinite, polyhalite) and used chiefly as a fertilizer and in the manufacture of alums and other… … Useful english dictionary
potassium sulfate — noun Date: 1869 a white crystalline compound K2SO4 used especially as a fertilizer … New Collegiate Dictionary
potassium sulfate — Chem. a crystalline, water soluble solid, K2SO4, used chiefly in the manufacture of fertilizers, alums, and mineral water, and as a reagent in analytical chemistry. [1880 85] * * * … Universalium
potassium sulfate — potas′sium sul′fate n. chem. a crystalline, water soluble solid, K2SO4, used chiefly in the manufacture of fertilizers, alums, and mineral water and as a reagent in analytical chemistry • Etymology: 1880–85 … From formal English to slang
normal potassium sulfate — kalio sulfatas statusas T sritis chemija formulė K₂SO₄ atitikmenys: angl. normal potassium sulfate; potassium sulfate; salt of Lemery rus. калий сернокислый; калия сульфат ryšiai: sinonimas – dikalio tetraoksosulfatas … Chemijos terminų aiškinamasis žodynas
aluminium-potassium sulfate — aliuminio kalio alūnas statusas T sritis chemija formulė KAl(SO₄)₂·12H₂O atitikmenys: angl. aluminium potassium alum; aluminium potassium sulfate rus. алюминиево калиевые квасцы ryšiai: sinonimas – aliuminio kalio bis(tetraoksosulfatas)… … Chemijos terminų aiškinamasis žodynas
aluminum potassium sulfate — potassium alum … Medical dictionary
