- Cerebrospinal fluid
-
Cerebrospinal fluid (CSF), Liquor cerebrospinalis, is a clear, colorless, salty bodily fluid, that occupies the subarachnoid space and the ventricular system around and inside the brain and spinal cord. In essence, the brain "floats" in it.
The CSF occupies the space between the arachnoid mater (the middle layer of the brain cover, meninges), and the pia mater (the layer of the meninges closest to the brain). It constitutes the content of all intra-cerebral (inside the brain, cerebrum) ventricles, cisterns, and sulci (singular sulcus), as well as the central canal of the spinal cord.
It acts as a "cushion" or buffer for the cortex, providing a basic mechanical and immunological protection to the brain inside the skull.
It is produced in the choroid plexus.
Contents
Circulation
CSF is produced in the brain by modified ependymal cells in the choroid plexus (approx. 50-70%), and the remainder is formed around blood vessels and along ventricular walls. It circulates from the lateral ventricles to the foramen of Monro (Interventricular foramen), third ventricle, aqueduct of Sylvius (Cerebral aqueduct), fourth ventricle, foramen of Magendie (Median aperture) and foramina of Luschka (Lateral apertures); subarachnoid space over brain and spinal cord. CSF is reabsorbed into venous sinus blood via arachnoid granulations.
It had been thought that CSF returns to the vascular system by entering the dural venous sinuses via the arachnoid granulations (or villi). However, some[1] have suggested that CSF flow along the cranial nerves and spinal nerve roots allow it into the lymphatic channels; this flow may play a substantial role in CSF reabsorbtion, in particular in the neonate, in which arachnoid granulations are sparsely distributed. The flow of CSF to the nasal submucosal lymphatic channels through the cribriform plate seems to be especially important.[2]
Amount and constitution
Reference ranges in CSF Substance Lower limit Upper limit Unit Glucose 50[3] 80[3] mg/dL Protein 15[3] 40[4]–45[3] mg/dL RBCs n/a[3] 0[3] / negative cells/µL or
cells/mm3WBCs 0[3] 3[3] cells/µL
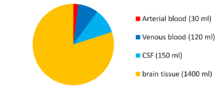
cells/mm3The CSF is produced at a rate of 500 ml/day. Since the brain can contain only 135 to 150 ml, large amounts are drained primarily into the blood through arachnoid granulations in the superior sagittal sinus. Thus the CSF turns over about 3.7 times a day. This continuous flow into the venous system dilutes the concentration of larger, lipoinsoluble molecules penetrating the brain and CSF.[5]
The CSF contains approximately 0.3% plasma proteins, or approximately 15 to 40 mg/dL, depending on sampling site.[4] CSF pressure ranges from 80 to 100 mmH2O (780–980 Pa or 4.4–7.3 mmHg) in newborns, and < 200 mmH20 (1.94 kPa) in normal children and adults, with most variations due to coughing or internal compression of jugular veins in the neck.
There are quantitative differences in the distributions of a number of proteins in the CSF. In general, globular proteins and albumin are in lower concentration in ventricular CSF compared to lumbar or cisternal fluid.[6]
Functions
CSF serves four primary purposes:
- Buoyancy: The actual mass of the human brain is about 1400 grams; however, the net weight of the brain suspended in the CSF is equivalent to a mass of 25 grams.[7] The brain therefore exists in neutral buoyancy, which allows the brain to maintain its density without being impaired by its own weight, which would cut off blood supply and kill neurons in the lower sections without CSF.[8]
- Protection: CSF protects the brain tissue from injury when jolted or hit. In certain situations such as auto accidents or sports injuries, the CSF cannot protect the brain from forced contact with the skull case, causing hemorrhaging, brain damage, and sometimes death.[8]
- Chemical stability: CSF flows throughout the inner ventricular system in the brain and is absorbed back into the bloodstream, rinsing the metabolic waste from the central nervous system through the blood-brain barrier. This allows for homeostatic regulation of the distribution of neuroendocrine factors, to which slight changes can cause problems or damage to the nervous system. For example, high glycine concentration disrupts temperature and blood pressure control, and high CSF pH causes dizziness and syncope.[8]
- Prevention of brain ischemia: The prevention of brain ischemia is made by decreasing the amount of CSF in the limited space inside the skull. This decreases total intracranial pressure and facilitates blood perfusion.
Pathology and laboratory diagnosis
When CSF pressure is elevated, cerebral blood flow may be constricted. When disorders of CSF flow occur, they may therefore affect not only CSF movement but also craniospinal compliance and the intracranial blood flow, with subsequent neuronal and glial vulnerabilities. The venous system is also important in this equation. Infants and patients shunted as small children may have particularly unexpected relationships between pressure and ventricular size, possibly due in part to venous pressure dynamics. This may have significant treatment implications, but the underlying pathophysiology needs to be further explored.
CSF connections with the lymphatic system have been demonstrated in several mammalian systems. Preliminary data suggest that these CSF-lymph connections form around the time that the CSF secretory capacity of the choroid plexus is developing (in utero). There may be some relationship between CSF disorders, including hydrocephalus and impaired CSF lymphatic transport.
CSF can be tested for the diagnosis of a variety of neurological diseases.[9] It is usually obtained by a procedure called lumbar puncture. Removal of CSF during lumbar puncture can cause a severe headache after the fluid is removed, because the brain hangs on the vessels and nerve roots, and traction on them stimulates pain fibers. The pain can be relieved by intrathecal injection of sterile isotonic saline. Lumbar puncture is performed in an attempt to count the cells in the fluid and to detect the levels of protein and glucose. These parameters alone may be extremely beneficial in the diagnosis of subarachnoid hemorrhage and central nervous system infections (such as meningitis). Moreover, a CSF culture examination may yield the microorganism that has caused the infection. By using more sophisticated methods, such as the detection of the oligoclonal bands, an ongoing inflammatory condition (for example, multiple sclerosis) can be recognized. A beta-2 transferrin assay is highly specific and sensitive for the detection for, e.g., CSF leakage.
Cause Appearance Polymorphonuclear cell Lymphocyte Protein Glucose Pyogenic bacterial meningitis Yellowish, turbid Markedly increased Slightly increased or Normal Markedly increased Decreased Viral meningitis Clear fluid Slightly increased or Normal Markedly increased Slightly increased or Normal Normal Tuberculous meningitis Yellowish and viscous Slightly increased or Normal Markedly increased Increased Decreased Fungal meningitis Yellowish and viscous Slightly increased or Normal Markedly increased Slightly increased or Normal Normal or decreased Lumbar puncture
Lumbar puncture can also be performed to measure the intracranial pressure, which might be increased in certain types of hydrocephalus. However a lumbar puncture should never be performed if increased intracranial pressure is suspected because it could lead to brain herniation and ultimately death.
Baricity
This fluid has an importance in anesthesiology. Baricity refers to the density of a substance compared to the density of human cerebral spinal fluid. Baricity is used in anesthesia to determine the manner in which a particular drug will spread in the intrathecal space.
Alzheimer's disease
A 2010 study showed analysis of CSF for three protein biomarkers can indicate the presence of Alzheimer's disease. The three biomarkers are CSF amyloid beta 1-42, total CSF tau protein and P-Tau181P. In the study, the biomarker test showed good sensitivity, identifying 90% of persons with Alzheimer's disease, but poor specificity, as 36% of control subjects were positive for the biomarkers. The researchers suggested the low specificity may be explained by developing but not yet symptomatic disease in controls.[10][11]
See also
- Meningitis
- CSF rhinorrhea
- Lumbar puncture
References
- ^ Zakharov A, Papaiconomou C, Djenic J, Midha R, Johnston M (2003). "Lymphatic CSF absorption pathways in neonatal sheep revealed by sub arachnoid injection of Microfil". Neuropathol. Appl. Neurobiol. 29 (6): 563–73. doi:10.1046/j.0305-1846.2003.00508.x. PMID 14636163.
- ^ Johnston M (2003). "The importance of lymphatics in cerebrospinal fluid transport". Lymphat. Res. Biol. 1 (1): 41–4. doi:10.1089/15396850360495682. PMID 15624320.
- ^ a b c d e f g h Normal Reference Range Table from The University of Texas Southwestern Medical Center at Dallas. Used in Interactive Case Study Companion to Pathologic basis of disease.
- ^ a b Felgenhauer K (1974). "Protein size and CSF composition". Klin. Wochenschr. 52 (24): 1158–64. doi:10.1007/BF01466734. PMID 4456012.
- ^ Saunders NR, Habgood MD, Dziegielewska KM (1999). "Barrier mechanisms in the brain, I. Adult brain". Clin. Exp. Pharmacol. Physiol. 26 (1): 11–9. doi:10.1046/j.1440-1681.1999.02986.x. PMID 10027064.
- ^ Merril CR, Goldman D, Sedman SA, Ebert MH (March 1981). "Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins". Science 211 (4489): 1437–8. doi:10.1126/science.6162199. PMID 6162199.
- ^ Noback, Charles; Norman L. Strominger, Robert J. Demarest, David A. Ruggiero (2005). The Human Nervous System. Humana Press. p. 93. ISBN 978-1588290403.
- ^ a b c Saladin, Kenneth (2007). Anatomy and Physiology: The Unity of Form and Function. McGraw Hill. p. 520. ISBN 978-0-07-287506-5.,
- ^ Seehusen DA, Reeves MM, Fomin DA (September 2003). "CSF analysis". Am Fam Physician 68 (6): 1103–8. PMID 14524396. http://www.aafp.org/afp/20030915/1103.html.
- ^ De Meyer, Geert et al. (August 2010). "Diagnosis-Independent Alzheimer Disease Biomarker Signature in Cognitively Normal Elderly People". Archive of Neurology 67 (8): 949–56. doi:10.1001/archneurol.2010.179. PMC 2963067. PMID 20697045. http://archneur.ama-assn.org/cgi/content/short/67/8/949. Retrieved 2010-09-08.
- ^ Herskovits, A. Zara; Growdon, John H. (August 2010). "Sharpen That Needle (editorial)". Archive of Neurology 67 (8): 918–20. doi:10.1001/archneurol.2010.151. PMID 20697041. http://archneur.ama-assn.org/cgi/content/extract/67/8/918. Retrieved 2010-09-08.
Anatomy: meninges of the brain and medulla spinalis (TA A14.1.01, GA 9.749/9.872) Layers Arachnoid granulation · Arachnoid trabeculae
Subarachnoid cisterns: Cisterna magna · Pontine cistern · Interpeduncular cistern · Chiasmatic · Lateral cerebral fossa · Of great cerebral vein · Of lamina terminalisTela chorioidea (Tela chorioidea of third ventricle, Tela chorioidea of fourth ventricle) · Choroid plexusCombinedSpaces External Links
- Circulation of Cerebrospinal Fluid (CSF) - Interactive Tool
Categories:- Body fluids
- Central nervous system
- Neurology
Wikimedia Foundation. 2010.