- p-Toluenesulfonic acid
-
p-Toluenesulfonic acid[1] 
 4-methylbenzenesulfonic acidOther namesTosylic acid
4-methylbenzenesulfonic acidOther namesTosylic acid
tosic acid
PTSAIdentifiers CAS number 104-15-4  ,
,
6192-52-5 (monohydrate)PubChem 6101 ChemSpider 5876 
DrugBank DB03120 KEGG C06677 
ChEBI CHEBI:27849 
ChEMBL CHEMBL541253 
Jmol-3D images Image 1 - Cc1ccc(cc1)S(=O)(=O)O
Properties Molecular formula CH3C6H4SO3H Molar mass 172.20 g/mol (anhydrous)
190.22 g/mol (monohydrate)Appearance colorless (white) solid Density 1.24 g/cm3 Melting point 38 °C, 311 K (anhydrous) [2]
103-106 °C, 376-379 K (monohydrate)Boiling point 140 °C, 413 K, 284 °F (at 20 mmHg)
Solubility in water 67 g/100 mL Acidity (pKa) -2.8 (water),[3]
8.5 (acetonitrile)[4]
Structure Molecular shape tetrahedral at S Hazards MSDS External MSDS R-phrases R36/37/38 S-phrases S26 Main hazards skin irritant Related compounds Related sulfonic acids Benzenesulfonic acid
Sulfanilic acid (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references p-Toluenesulfonic acid (PTSA) or tosylic acid (TsOH) is an organic compound with the formula CH3C6H4SO3H. It is a white solid that is soluble in water, alcohols, and other polar organic solvents. The 4-CH3C6H4SO2- group is known as the Tosyl group and is often abbreviated as Ts or Tos. Most often, TsOH refers to the monohydrate, TsOH.H2O.
TsOH is a strong organic acid, about a million times stronger than benzoic acid. It is one of the few strong acids that are solid and, hence, conveniently weighed. Also, unlike some of the strong mineral acids (especially nitric acid, sulfuric acid, and perchloric acid), TsOH is non-oxidizing.
Contents
History
Safety
Preparation and handling
TsOH is prepared on an industrial scale by the sulfonation of toluene. It hydrates readily. Common impurities include benzenesulfonic acid and sulfuric acid. Impurities can be removed by recrystallization from its concentrated aqueous solution followed by azeotropic drying with toluene.[5]
Toluenesulfonic acid finds use in organic synthesis as an "organic-soluble" acid catalyst. Examples of uses:
- Acetalization of an aldehyde.[6]
- Esterification of carboxylic acids.[7]
- Transesterification of an ester.[8]
Tosylate esters
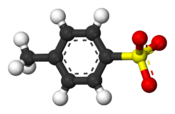 Ball-and-stick model of the tosylate anion
Ball-and-stick model of the tosylate anion
A tosylate ester has a limited shelf life in air due to its ready hydrolysis in the presence of light. The tosyl group is electron-withdrawing, which makes tosylates excellent leaving groups. The tosyl group is also a protecting group for alcohols and amines, prepared by combining the alcohol with 4-toluenesulfonyl chloride, usually in an aprotic solvent, often pyridine, the basicity of which activates the reaction.[9] Toluenesulfonate esters undergo nucleophilic attack or elimination.
Reactions
- p-Toluenesulfonic acid may be converted to phosphorus pentoxide.[10]
- When toluene sulphonic acid (o-, m-, p-,) is boiled with HCl or treated with super-heated steam, toluene is formed along with H2SO4
CH3C6H4SO3H (Toluene Sulphonic acid) + HOH (steam) → C6H5CH3 (Toluene) + H2SO4 (Sulfuric acid)
See also
References
- ^ Merck Index, 11th Edition, 9459.
- ^ Armarego, Wilfred (2003). Purification of Laboratory Chemicals. Elsevier Science. pp. 370. ISBN 0-7506-7571-3.
- ^ Guthrie, J. P. Hydrolysis of esters of oxy acids: pKa values for strong acids. Can. J. Chem. 1978, 56, 2342-2354.
- ^ Eckert, F.; Leito, I.; Kaljurand, I.; Kütt, A.; Klamt, A.; Diedenhofen, M. Prediction of Acidity in Acetonitrile Solution with COSMO-RS. J. Comput. Chem. 2009, 30, 799-810. DOI: 10.1002/jcc.21103
- ^ Perrin, D. D. and Armarego, W. L. F., Purification of Laboratory Chemicals, Pergamon Press: Oxford, 1988.
- ^ H. Griesser, H.; Öhrlein, R.; Schwab, W.; Ehrler, R.; Jäger, V. (2004), "3-Nitropropanal, 3-Nitropropanol, and 3-Nitropropanal Dimethyl Acetal", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=v77p0236; Coll. Vol. 10: 577
- ^ Furuta, K. Gao, Q.-z.; Yamamoto, H. (1998), "Chiral (Acyloxy)borane Complex-catalyzed Asymmetric Diels-Alder Reaction: (1R)-1,3,4-Trimethyl-3-cyclohexene-1-carboxaldehyde", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv9p0722; Coll. Vol. 9: 722
- ^ Imwinkelried, R.; Schiess, M.; Seebach, D. (1993), "Diisopropyl (2S,3S)-2,3-O-isopropylidenetartrate", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv8p0201; Coll. Vol. 8: 201
- ^ Nucleophilic Substitution
- ^ L. Field and J. W. McFarland (1963), "p-Toluenesulfonic Anhydride", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv4p0940; Coll. Vol. 4: 940
Categories:- Benzenesulfonic acids
- Reagents for organic chemistry
Wikimedia Foundation. 2010.
