- Copper indium gallium selenide solar cells
-
Copper indium gallium selenide (CuIn1-xGaxSe2 or CIGS) is a direct bandgap semiconductor useful for the manufacture of solar cells. Because the material strongly absorbs sunlight, a much thinner film is required than of other semiconductor materials. The CIGS absorber is deposited on a glass backing, along with electrodes to collect current.
CIGS's absorption coefficient is higher than any other semiconductor used for solar modules. Devices made with CIGS belong to the thin-film category of photovoltaics (PV). The market for thin-film PV grew at a 60% annual rate from 2002 to 2007 and is still growing rapidly (Figure 1).[1] Therefore, a strong incentive exists to develop and improve deposition methods for these films that will allow lower cost and increased throughput.
Contents
Properties
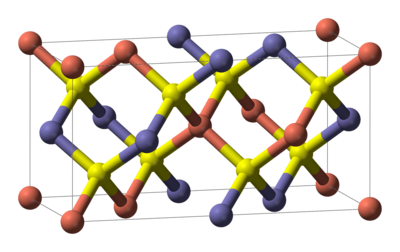
CIGS is a I-III-VI2 compound semiconductor material composed of copper, indium, gallium, and selenium. The material is a solid solution of copper indium selenide (often abbreviated "CIS") and copper gallium selenide, with a chemical formula of CuInxGa(1-x)Se2, where the value of x can vary from 1 (pure copper indium selenide) to 0 (pure copper gallium selenide). It is a tetrahedrally bonded semiconductor, with the chalcopyrite crystal structure. The bandgap varies continuously with x from about 1.0 eV (for copper indium selenide) to about 1.7 eV (for copper gallium selenide).[2]
CIGS has an exceptionally high absorption coefficient of more than 105/cm for 1.5 eV and higher energy photons.[3] CIGS solar cells with efficiencies greater than 20% have been claimed by both the National Renewable Energy Laboratory (NREL) and the Zentrum für Sonnenenergie und Wasserstoff Forschung (ZSW), which is the record to date for any thin film solar cell.[4][5]
CIGS photovoltaic cells
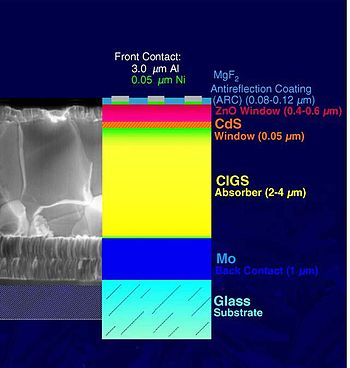
The most common device structure for CIGS solar cells is shown in Figure 2. Glass is commonly used as a substrate, however, many companies are also looking at lighter and more flexible substrates such as polyimide or metal foils.[6] A molybdenum layer is deposited (commonly by sputtering) which serves as the back contact and to reflect most unabsorbed light back into the absorber. Following Mo deposition a p-type CIGS absorber layer is grown by one of several unique methods. A thin n-type buffer layer is added on top of the absorber. The buffer is typically CdS deposited via chemical bath deposition. The buffer is overlaid with a thin, intrinsic ZnO layer which is capped by a thicker, Al doped ZnO layer. Despite increasing the series resistance, the intrinsic ZnO layer is beneficial to cell performance. The precise mechanism for the improvement is still being debated.[7] The Al doped ZnO serves as a transparent conducting oxide to collect and move electrons out of the cell while absorbing as little light as possible.
The materials based on CuInSe2 that are of interest for photovoltaic applications include several elements from groups I, III and VI in the periodic table. These semiconductors are especially attractive for thin film solar cell application because of their high optical absorption coefficients and versatile optical and electrical characteristics which can in principle be manipulated and tuned for a specific need in a given device.[8]
Conversion efficiency
CIGS is mainly used in the form of polycrystalline thin films. The best efficiency achieved as of December 2005 was 19.5%.[9] A team at the National Renewable Energy Laboratory achieved 19.9% new world record efficiency[10] by modifying the CIGS surface and making it look like CIS.[9]
These efficiencies are different from module conversion efficiencies. Two of the leading manufacturers of CIGS thin-film PV have hit new record highs in module conversion efficiencies. The U.S. National Renewable Energy Laboratory has confirmed 13.8% efficiency of a large-area (meter-square) production panel, and 13% total-area (and 14.2% aperture-area) efficiency with some production modules.[9]
Higher efficiencies (around 30%) can be obtained by using optics to concentrate the incident light. The use of gallium increases the optical band gap of the CIGS layer as compared to pure CIS, thus increasing the open-circuit voltage.[9][11] In another point of view, gallium is added to replace as much indium as possible due to gallium’s relative availability to indium.
Deposition
CIGS films can be manufactured by several different methods:
- The most common vacuum-based process co-evaporates or co-sputters copper, gallium, and indium, then anneals the resulting film with a selenide vapor to form the final CIGS structure. An alternative is to directly co-evaporate copper, gallium, indium and selenium onto a heated substrate.
- A non-vacuum-based alternative process deposits nanoparticles of the precursor materials on the substrate and then sinters them in situ. Electroplating is another low cost alternative to apply the CIGS layer.
With record CIGS efficiency at just below 20% for several years,[9] new trends of CIGS research has been focused on lower-cost deposition methods as an alternative to expensive vacuum processes. This new research progressed quickly and efficiencies of 10%–15% have been achieved by many teams.[9]
CIGS and silicon
Unlike the silicon cells based on a homojunction, the structure of CIGS cells is a more complex heterojunction system. CIGS solar cells are not as efficient as crystalline silicon solar cells, for which the record efficiency lies at 24.7%.[12] Though some argue that CIGS will be substantially cheaper due to much lower material costs and potentially lower fabrication costs, in the second quarter of 2011, Veeco, Inc., a company with expertise in thin film deposition technology, closed a major CIGS development project due in part to the continued declining costs of the competing silicon materials and technology.[13]
A direct bandgap material, CIGS has very strong light absorption and only 1–2 micrometers of CIGS is enough to absorb most of the sunlight. A much greater thickness of crystalline silicon is required for the same absorption.
The active layer (CIGS) can be deposited in a polycrystalline form directly onto molybdenum coated glass sheets or steel bands. This uses less energy than growing large crystals, which is a necessary step in the manufacture of crystalline silicon solar cells. Also unlike crystalline silicon, these substrates can be flexible.[14]
CIGS and other thin films
CIGS belongs in the category of thin film solar cells (TFSC).[15] The semiconductors used as absorber layer in thin-film photovoltaics exhibit direct bandgaps allowing the cells to be a few micrometers thin; hence, the term thin-film solar cells is used. Other materials in this group of TFSC include CdTe and amorphous Si.[16] Their record efficiencies are slightly lower than that of CIGS for lab-scale top performance cells. The 19.9% efficiency is by far the highest compared with those achieved by other thin film technologies such as Cadmium Telluride (CdTe) or amorphous silicon (a-Si).[10] As for CIS, and CGS solar cells, the world record total area efficiencies are 15.0% and 9.5%[17] respectively. Another advantage of CIGS compared to CdTe is smaller amount of toxic material cadmium are present in CIGS cells.
Structure of a CIGS thin-film solar cell
The basic structure of a Cu(In,Ga)Se2 thin-film solar cell is depicted in the image to the right. The most common substrate is soda-lime glass of 1–3 mm thickness. This is coated on one side with molybdenum (Mo) that serves as metal back contact. The heterojunction is formed between the semiconductors CIGS and ZnO, separated by a thin layer of CdS and a layer of intrinsic ZnO. The CIGS is doped p-type from intrinsic defects, while the ZnO is doped n-type to a much larger extent through the incorporation of aluminum (Al). This asymmetric doping causes the space-charge region to extend much further into the CIGS than into the ZnO. Matched to this are the layer thicknesses and the bandgaps of the materials: the wide CIGS layer serves as absorber with a bandgap between 1.02 eV (CuInSe2) and 1.65 eV (CuGaSe2). Absorption is minimized in the upper layers, called window, by the choice of larger bandgaps: Eg,ZnO=3.2 eV and Eg,CdS=2.4 eV. The doped ZnO also serves as front contact for current collection. Laboratory scale devices, typically 0.5 cm2 large, are provided with a Ni/Al-grid deposited onto the front side to contact the ZnO.[18]
Production of modules involves the depositon layer being cut into a series of parallel connected strips. A further transparent protective cover is applied to the module. This sandwich construction is then sealed against the ingress of moisture.[19] Some method of physical support is required to prevent fracture of this fragile structure.[18]
General properties of high performance CIGS absorbers
All high performance CIGS absorbers in solar cells have several similarities independent of the growth technique used. First, they are polycrystalline α-phase which has the chalcopyrite crystal structure shown in Figure 3. The second property is an overall Cu deficiency.[20] Cu deficiency increases the majority carrier (hole) concentration by increasing the number of Cu vacancies. These vacancies act as electron acceptors. Also, when CIGS films are In rich (Cu deficient) the surface layer of the film forms an ordered defect compound (ODC) with a stoichiometry of Cu(In,Ga)3Se5. The ODC is n-type, forming a p-n homojunction in the film at the interface between the α phase and the ODC. Recombination velocity at the CIGS/CdS interface is decreased by presence of the homojunction. The drop in interface recombination attributable to ODC formation is demonstrated by experiments which have shown recombination in the bulk of the film is the main loss mechanism in Cu deficient films, while in Cu rich films the main loss is at the CIGS/CdS interface.[7][20]
Sodium (Na) incorporation is also necessary for optimal performance. Ideal Na concentration is considered to be approximately 0.1 at%. Na is commonly supplied by the soda-lime glass used as the substrate, but in processes that do not use this substrate the Na must be deliberately added. Beneficial effects of Na include increases in p-type conductivity, texture, and average grain size. Furthermore, Na incorporation allows for performance to be maintained over larger stoichiometric deviations.[3] Simulations have predicted that Na on an In site creates a shallow acceptor level and that Na serves to remove In on Cu defects (donors), but reasons for these benefits are still being debated. Na is also credited with catalyzing oxygen absorption. Oxygen passivates Se vacancies that act as compensating donors and recombination centers.
Alloying CIS (CuInSe2) with CGS (CuGaSe2) increases in the bandgap. To reach the ideal bandgap for a single junction solar cell, 1.5 eV, a Ga/(In+Ga) ratio of roughly 0.7 would be optimal. However, at ratios above ~0.3 device performance drops off. Industry currently targets the 0.3 Ga/(In+Ga) ratio, resulting in bandgaps between 1.1 and 1.2 eV. The decreasing performance has been postulated to be a result of CGS not forming the ODC, which is necessary for a good interface with CdS.[20]
The highest efficiency devices show a high degree of texturing, or preferred crystallographic orientation. Until recently record efficiency devices displayed a (112) texture, but now a (204) surface orientation is observed in the best quality devices.[3] A smooth absorber surface is preferred to maximize the ratio of the illuminated area to the area of the interface. The area of the interface increases with roughness while illuminated area remains constant, decreasing open circuit voltage (VOC). Studies have also linked an increase in defect density to decreased VOC. recombination in CIGS has been suggested to be dominated by non-radiative processes. Theoretically, recombination can be controlled by engineering of the film, as opposed to being intrinsic to the material.[21]
Precursor deposition and post processing
Perhaps the most common method used to create CIGS films for commercial use is deposition of precursor materials – always including Cu, In, and Ga, and sometimes including Se – onto a substrate and processing these films at high temperatures under a proper atmosphere. The following sections outline the various techniques for precursor deposition processing, including sputtering of metallic layers at low temperatures, printing of inks containing nanoparticles, electrodeposition, and a technique inspired by wafer-bonding.
General selenization concerns
The Se supply and selenization environment is extremely important in determining the properties and quality of the film produced from precursor layers. When Se is supplied in the gas phase (for example as H2Se or elemental Se) at high temperatures the Se will become incorporated into the film by absorption and subsequent diffusion. During this step, called chalcogenization, complex interactions occur to form a chalcogenide. These interactions include formation of Cu-In-Ga intermetallic alloys, formation of intermediate metal-selenide binary compounds, and phase separation of various stoichiometric CIGS compounds. Because of the variety and complexity of the reactions taking place, the properties of the CIGS film are difficult to control.[3]
Differences exist between films formed using different Se sources. Using H2Se yields the fastest Se incorporation into the absorber; 50 at% Se can be achieved in CIGS films at temperatures as low as 400 °C. By comparison, elemental Se only achieves full incorporation with reaction temperatures of 500 °C and above. Below 500 °C films formed from elemental Se were not only Se deficient, but also had multiple phases including metal selenides and various alloys. Use of H2Se also provides the best compositional uniformity and the largest grain sizes. However, H2Se is highly toxic and is classified as hazardous to the environment.
Sputtering of metallic layers followed by selenization
In this method of forming CIGS absorbers, a metal film of Cu, In, and Ga is sputtered at or near room temperature and reacted in a Se atmosphere at high temperature. This process has higher throughput than coevaporation and compositional uniformity can be more easily achieved.
Sputtering a stacked multilayer of metal – for example a Cu/In/Ga/Cu/In/Ga... structure – produces a smoother surface and better crystallinity in the absorber, when compared to a simple bilayer (Cu-Ga alloy/In) or trilayer (Cu/In/Ga) sputtering. These attributes result in higher efficiency devices, but forming the multilayer is a more complicated deposition process and is likely not worth the cost of extra equipment or the added process complexity.[20] Additionally, the reaction rates of Cu/Ga and Cu/In layers with Se are different. If the reaction temperature is not high enough, or not held long enough, CIS and CGS form as separate phases. The same considerations outlined in the previous section apply to Se incorporation.
Companies currently using similar processes include Showa Shell, Avancis(formerly Shell Solar), Miasolé, Honda Soltec, and Energy Photovoltaics (EPV).[22] Showa Shell sputters a Cu-Ga alloy layer and an In layer, followed by selenization in H2Se and sulfurization in H2S. The sulfurization step appears to passivate the surface in a way similar to CdS in most other cells. Thus, the buffer layer used is Cd-free which eliminates the worries related to the toxicity and environmental impact of Cd. Showa Shell has reported a maximum module efficiency of 13.6% with an average of 11.3% for 3600 cm2 substrates.[6] Shell Solar uses the same technique as Showa Shell to create the absorber; however, they use a CdS layer deposited by chemical vapor deposition. Modules sold by Shell Solar have a specification of 9.4% module efficiency.
Miasole has had great success in procuring venture capital funds for its process and scale up. However, little is known about their sputtering/selenization process beyond their stated efficiency of 9 to 10% for modules.
EPV uses a hybrid between coevaporation and sputtering in which In and Ga are evaporated in a Se atmosphere. This is followed by Cu sputtering and a selenization step. Finally, In and Ga are again evaporated in the presence of Se. Based on Hall measurements, these films have a low carrier concentration and high mobility compared to other devices. EPV films have also been shown to have a low defect concentration.
Chalcogenization of particulate precursor layers
In this method, metal or metal-oxide nanoparticles are used as the precursors for CIGS growth. These nanoparticles are generally suspended in a water based solution and then applied to large areas by various methods, with printing the most common. The film is then dehydrated and, if the precursors are metal-oxides, reduced in a H2/N2 atmosphere. Following dehydration, the remaining porous film is sintered and selenized at temperatures greater than 400 °C.[20][21][23]
Nanosolar and International Solar Electric Technology (ISET) are attempting to scale up this process.[6] ISET uses oxide particles while Nanosolar is extremely secretive about their ink. The ink composition is unknown but there is some implication that Se is also incorporated into Nanosolar's ink. The advantages of this process include uniformity over large areas, non-vacuum or low-vacuum equipment, and adaptability to roll-to-roll manufacturing. When compared to laminar metal precursor layers, the selenization of sintered nanoparticles is more rapid. The increased rate is a result of the greater surface area associated with porosity. Decreasing high temperature selenization reduces the thermal budget. Unfortunately, the drawback of porosity is a tendency towards rougher absorber surfaces. Use of particulate precursors allows for printing on a large variety of substrates with high materials utilization, around 90% or more. A disadvantage is that little research and development exists in this area of deposition. In Nanosolar's manufacturing the printed rolls are cut into cells and must be binned and integrated in a fashion similar to how Si devices are made today. The binning process is different from the monolithic integration that many CIGS companies are using. Monolithic integration is far more adaptable to inline production.
Nanosolar has reported a cell (not module) efficiency of 14%, however this has not been verified by any national laboratory testing, nor are they allowing any onsite inspections of their facilities to verify this and other claims made in the past. In independent testing[21] ISET's absorber had the 2nd lowest efficiency at 8.6%. However, all the modules that beat ISET's module were coevaporated, a process which has manufacturing disadvantages and higher costs. ISET's sample suffered most from low VOC and low fill factor, indicative of a rough surface and/or a high number of defects aiding recombination. Related to these issues, the film had poor transport properties including a low Hall mobility and short carrier lifetime.
Electrodeposition followed by selenization
Precursors can also be deposited by electrodeposition. Two different methodologies exist: deposition of elemental layered structures, and simultaneous deposition of all elements (including Se). Both methods require thermal treatment in a Se atmosphere to make device quality films. Because electrodeposition requires conductive electrodes, metal foils are a logical substrate. Electrodeposition of elemental layers is similar to the sputtering of elemental layers. Currently no company is scaling up this process.
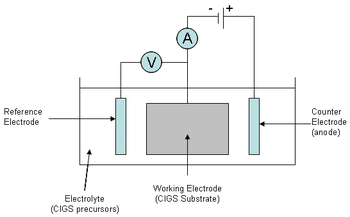
Simultaneous deposition is performed using a working electrode (cathode), a counter electrode (anode), and a reference electrode as in Figure 4. A metal foil substrate is used as the working electrode in industrial processes. An inert material is used for the counter electrode, and the reference electrode exists to measure and control the potential difference between the anode and cathode. The reference electrode allows the process to be performed potentiostatically, meaning the potential of the substrate can be controlled.[20]
Electrodeposition of all elements simultaneously is a difficult processing problem for a variety of reasons. First, the standard reduction potentials of the elements are not the same, causing preferential deposition of a single element. This problem is commonly alleviated by adding different counter ions into solution for each ion to be deposited (Cu2+, Se4+, In3+, and Ga3+), thus changing the reduction potential for that ion. Second, the Cu-Se system has a complicated behavior and the composition of the film depends on the Se4+/Cu2+ ion flux ratio which can vary over the film surface. Because of this behavior the deposition conditions, specifically precursor concentrations and deposition potential, need to be optimized. Even with optimization, reproducibility is low over large areas due to composition variations and potential drops along the substrate.
The resulting films have small grains, are Cu-rich, and generally contain Cu2-xSex phases along with impurities from the solution. Annealing is required to improve crystallinity. In order to achieve efficiencies higher than 7%, a stoichiometry correction is also required. The correction is done via high temperature physical vapor deposition which is not practical in industry.
Having solved the optimization issues, Solopower is currently producing cells with >13.7% conversion efficiency as per NREL. SoloPower is currently attempting to scale up the process, but few details have been released. SoloPower is relying on the advantages of roll-to-roll manufacturing and flexible metal foil substrates.
Precursor combination by wafer-bonding inspired technique
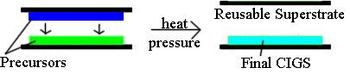
In this process, two different precursor films are deposited separately on a substrate and a superstrate. The films are pressed together and heated to release the film from the superstrate leaving a CIGS absorber on the substrate. This technique allows the superstrate to be reused (Figure 5). Heliovolt patented this procedure and has named it the FASST process. Therefore, Heliovolt is the only company currently scaling up the technique. In principle, the precursors can be deposited at low temperature using low cost deposition techniques, lowering the final cost of the module. However, the first one or two generations of product will still use higher temperature PVD methods and not achieve full cost cutting potential. Flexible substrates could eventually be used in this process.
Typical film characteristics are not known outside of the company as no research has been conducted by independently funded laboratories. However, Heliovolt has claimed a top cell efficiency of 12.2%.
Coevaporation
Coevaporation, or codeposition, is the most prevalent CIGS fabrication technique in the laboratory and an important method in industry. The Boeing coevaporation process deposits bilayers of CIGS with different stoichiometries onto a heated substrate and allows them to intermix. The National Renewable Energy Laboratory (NREL) has developed another process which involves three deposition steps and produced the current CIGS efficiency record holder at 19.9%. The first step in NREL's method is codeposition of In, Ga, and Se. This is followed by Cu and Se deposited at a higher temperature to allow for diffusion and intermixing of the elements. In the final stage In, Ga, and Se are again deposited to make the overall composition Cu deficient.[20]
Würth Solar has been producing CIGS cells using an inline coevaporation system since 2005 with module efficiencies between 11% and 12% by the end of that year. They have subsequently opened another production facility and continued to improve efficiency and yield. Other companies scaling up coevaporation processes include Global Solar and Ascent Solar.[22] Global Solar also uses an inline three stage deposition process. In all of the steps Se is supplied in excess in the vapor phase. In and Ga are first evaporated followed by Cu and then by In and Ga to make the film Cu deficient. These films performed quite favorably in relation not only to other manufacturers but also to absorbers grown at NREL and the Institute for Energy Conversion (IEC).[21] However, fully fabricated modules of Global Solar’s films did not perform as well. The property in which the module most obviously under-performed was a low VOC, which is characteristic of high defect density and high recombination velocities. Interestingly, Global Solar’s absorber layer outperformed the NREL absorber in carrier lifetime and hall mobility. However, as completed cells the NREL sample performed better. This is evidence of a poor CIGS/CdS interface, possibly due to the lack of an ODC surface layer on the Global Solar film.
As most of the CIGS research at national laboratories and universities covers coevaporation, companies using this technique stand to gain the most from the scientific community. However, they also face significant disadvantages including uniformity issues over large areas and the related difficulty of coevaporating elements in an inline system. Another disadvantage is high growth temperatures which raise the thermal budget and cost. Additionally, coevaporation is plagued by low material utilization (deposition on chamber walls instead of the substrate, especially for selenium) and expensive vacuum equipment.[6][23] A way to enhance the utilisation of selenium is the usage of a thermal or plasma-enhanced selenium-cracking process,[24][25] which can additionally be coupled with an ion beam source for ion beam assisted deposition.[26]
Chemical vapor deposition
Chemical vapor deposition (CVD) has been implemented in multiple ways for the deposition of CIGS. Processes include atmosphere pressure metal organic CVD (AP-MOCVD), plasma-enhanced CVD (PECVD), low-pressure MOCVD (LP-MOCVD), and aerosol assisted MOCVD (AA-MOCVD). Current work is focused on changing the typical dual-source precursors to single-source precursors.[20] Multiple source precursors must be homogeneously mixed and the flow rates of the precursors have to be kept at the proper stoichiometry. Single-source precursor methods do not suffer from these drawbacks and should enable better control of film composition compared to multiple source precursors.
CVD is not yet being used by any companies for CIGS synthesis. Currently, CVD produced films have low efficiency and a low VOC, partially a result of a high defect concentration. Additionally, the surfaces of the films are generally quite rough which serves to further decrease the VOC. However, the requisite Cu deficiency has been achieved using AA-MOCVD along with a (112) crystal orientation.
However, if the film quality produced by CVD can be improved, any company using this technique could benefit from knowledge gained in other industries using large area CVD deposition, such as glass coating manufacturers. CVD deposition temperatures are lower than those used for other processes such as co-evaporation and selenization of metallic precursors. Therefore, CVD has a lower thermal budget, reducing the cost. Potential manufacturing problems include difficulties converting CVD to an inline process as well as the expense of handling volatile precursors.
Breakthroughs and Commercial
Despite CIGS having the advantage over CdTe where both heavy metal Cadmium usage as well as rare-earth Telluride availability issues, the development of the CIGS lags behind CdTe commercially. In the laboratory, 18.7% efficiency on a flexible medium was achieved with CIGS cells.,[27] though it remains to be seen how well mass production values can be achieved with any CIGS cell. Commercial production of flexible CIGS cells has begun, in Berlin, Germany, commercial production after initial production runs has begun on an annual 35MW capable facility.[28]
See also
References
- ^ Thin-Film wins PV market share: Three New Plants in Germany Total Almost 50 MW. Sustainableenergyworld.eu (2009-03-14). Retrieved on 2011-09-13.
- ^ Tinoco, T.; Rincón, C.; Quintero, M.; Pérez, G. Sánchez (1991). "Phase Diagram and Optical Energy Gaps for CuInyGa1−ySe2 Alloys". Physica Status Solidi (a) 124 (2): 427. Bibcode 1991PSSAR.124..427T. doi:10.1002/pssa.2211240206.
- ^ a b c d B. J. Stanbery (2002). Critical Reviews in Solid State and Materials Science 27: 73.
- ^ Repins, I.; Contreras, Miguel A.; Egaas, Brian; Dehart, Clay; Scharf, John; Perkins, Craig L.; To, Bobby; Noufi, Rommel (2008). "19.9%-efficient ZnO/CdS/CuInGaSe2 solar cell with 81.2% fill factor". Progress in Photovoltaics: Research and applications 16 (3): 235. doi:10.1002/pip.822.
- ^ ZSW: Press Releases. Zsw-bw.de. Retrieved on 2011-09-13.
- ^ a b c d Dhere, Neelkanth G. (2007). "Toward GW/year of CIGS production within the next decade". Solar Energy Materials and Solar Cells 91 (15–16): 1376. doi:10.1016/j.solmat.2007.04.003.
- ^ a b Ihlal, A; Bouabid, K; Soubane, D; Nya, M; Aittalebali, O; Amira, Y; Outzourhit, A; Nouet, G (2007). "Comparative study of sputtered and electrodeposited CI(S,Se) and CIGSe thin films". Thin Solid Films 515 (15): 5852. Bibcode 2007TSF...515.5852I. doi:10.1016/j.tsf.2006.12.136.
- ^ "Thin film CuInSe2/Cd(Zn)S Heterojunction Solar Cell : Characterization and Modeling", Murat Nezir Eron, PhD. Thesis, Drexel University, 1984, Philadelphia
- ^ a b c d e f "The staus and future of the photovoltaics industry". David E. Carlson Chief Scientist BP Solar 14 March 2010. http://www.aps.org/units/gera/meetings/march10/upload/CarlsonAPS3-14-10.pdf. Retrieved 10 February 2011.
- ^ a b "Characterization of 19.9%-Efficient CIGS Absorbers". National Renewable Energy Laboratory May 2008. http://www.nrel.gov/docs/fy08osti/42539.pdf. Retrieved 10 February 2011.
- ^ "Solar cell efficiency tables Ver.33". National Institute of Advanced Industrial Science and Technology (AIST). http://159.226.64.60/fckeditor/UserFiles/File/tyndc/reference/19909584825552.pdf. Retrieved 10 February 2011.
- ^ Green, M.A.; Jianhua Zhao; Wang, A.; Wenham, S.R. (1999). "Very high efficiency silicon solar cells-science and technology". IEEE Transactions on Electron Devices 46 (10): 1940. Bibcode 1999ITED...46.1940G. doi:10.1109/16.791982.
- ^ "Second Quarter 2011 Earning Call Press Release". WSJ.com. http://online.wsj.com/article/PR-CO-20110728-915286.html?mod=crnews. Retrieved 28 July 2011.
- ^ "First sales for 'world's cheapest solar cells'". Chemistry world February 2008. http://www.rsc.org/chemistryworld/Issues/2008/February/FirstSalesForWorldsCheapestSolarCells.asp. Retrieved 6 April 2011.
- ^ "Process for making solar cells". United States Patent Application 20090223551. http://www.freepatentsonline.com/y2009/0223551.html. Retrieved 10 February 2011.
- ^ "Solar Power Lightens Up with Thin-Film Technology". Scientific American 25 April 2008. http://www.scientificamerican.com/article.cfm?id=solar-power-lightens-up-with-thin-film-cells. Retrieved 10 February 2011.
- ^ Young, D. L.; Keane, James; Duda, Anna; Abushama, Jehad A. M.; Perkins, Craig L.; Romero, Manuel; Noufi, Rommel (2003). "Improved performance in ZnO/CdS/CuGaSe2 thin-film solar cells". Progress in Photovoltaics: Research and Applications 11 (8): 535. doi:10.1002/pip.516.
- ^ a b "Polycrystalline Thin Film Solar Cell Technologies". National Renewable Energy Laboratory Colorado U.S.A.. http://www.nrel.gov/docs/fy09osti/44622.pdf. Retrieved 10 February 2011.
- ^ "Degradation of ZnO Window Layer for CIGS by Damp-Heat Exposure". National Renewable Energy Laboratory August 2008. http://www.nrel.gov/docs/fy08osti/42792.pdf. Retrieved 10 February 2011.
- ^ a b c d e f g h Kemell, Marianna; Ritala, Mikko; Leskelä, Markku (2005). "Thin Film Deposition Methods for CuInSe2 Solar Cells". Critical Reviews in Solid State and Materials Sciences 30: 1. doi:10.1080/10408430590918341.
- ^ a b c d Repins, I. L.; Stanbery, B. J.; Young, D. L.; Li, S. S.; Metzger, W. K.; Perkins, C. L.; Shafarman, W. N.; Beck, M. E. et al. (2006). "Comparison of device performance and measured transport parameters in widely-varying Cu(In,Ga) (Se,S) solar cells". Progress in Photovoltaics: Research and Applications 14: 25. doi:10.1002/pip.654.
- ^ a b H. S. Ulal, and B. von Roedern (2008). "Critical. Issues for Commercialization of Thin-Film. PV Technologies". Solid State Technology 51 (2): 52–54.
- ^ a b K. Derbyshire (2008). Solid State Technology 51: 32.
- ^ Ishizuka, S. et al. (2009). "Large grain CIGS thin film growth using a Se-radical beam source". Solar Energy Materials & Solar Cells 93 (6–7): 792. doi:10.1016/j.solmat.2008.09.043.
- ^ Kawamura, M. et al. (2009). "CIGS thin-film solar cells grown with cracked selenium". Journal of Crystal Growth 311 (3): 753. Bibcode 2009JCrGr.311..753K. doi:10.1016/j.jcrysgro.2008.09.091.
- ^ Solarion AG (Hrsg.): World record: 13.4 % conversion efficiency in solar cells on plastic film. (press release)
- ^ http://www.sciencedaily.com/releases/2011/09/110921131729.htm
- ^ http://www.semiconductor-today.com/news_items/2011/OCT/GSE_041011.html
External links
- Copper Indium Diselenide Publications, Presentations, and News Database of the National Renewable Energy Laboratory.
- World's Largest CIGS Solar Array Operational In Arizona.
- Michael Kanellos Silicon vs. CIGS: With solar energy, the issue is material October 2, 2006 CNET News.com
- CIGS to emerge as the major thin-film photovoltaic technology by 2020
Photovoltaics Concepts - Photoelectric effect
- Photovoltaics
- History of photovoltaics
- Timeline of solar cells
- Solar insolation
- Solar constant
- Solar cell efficiency
- Third generation photovoltaic cell
- Solar cell research
- Quantum efficiency of a solar cell
- Cadmium telluride
- Thermophotovoltaic
- Polycrystalline silicon photovoltaics
- Thermodynamic efficiency limit
- Sun-free photovoltaics
- Polarizing organic photovoltaics

Photovoltaic system Solar cells- Solar cell
- Solar panel
- Thin film solar cell
- Polymer solar cell
- Nanocrystal solar cell
- Organic solar cell
- Quantum dot solar cell
- Hybrid solar cell
- Plasmonic solar cell
- Carbon nanotubes in photovoltaics
- Dye-sensitized solar cell
- Cadmium telluride photovoltaics
- Copper indium gallium selenide solar cells
- Multijunction photovoltaic cell
- Printed solar panel
System components- Solar charge controller
- Solar inverter
- Solar micro-inverter
- Solar cable
- Solar combiner box
- Photovoltaic mounting system
- Maximum power point tracker
- Solar tracker
- Solar shingles
- Solar mirror
System concepts- Perturb and observe method
- Incremental conductance method
- Constant voltage method
- Fill factor
- Concentrated photovoltaics
- Photovoltaic thermal hybrid solar collector
- Space-based solar power
- Watt-peak
Applications Appliances- Solar-powered refrigerator
- Solar air conditioning
- Solar lamp
- Solar charger
- Solar backpack
- Solar tree
- Solar-powered pump
- Solar-powered watch
- Solar Tuki
- Photovoltaic keyboard
- Solar road stud
- Solar cell phone charger
- Solar notebook
- Solar powered calculator
- Solar powered fountain
- Solar powered radio
- Solar powered flashlight
- Solar fan
- Solar street light
- Solar traffic light
Land transport- Solar vehicle
- Solar car
- Solar roadway
- Solar golf cart
- The Quiet Achiever
- Sunmobile
Air transport- Electric aircraft
- Mauro Solar Riser
- Solar panels on spacecraft
- Solar-Powered Aircraft Developments Solar One
- Gossamer Penguin
- Qinetiq Zephyr
- Solar Challenger
Water transportSolar vehicle racing- Solar car racing
- List of solar car teams
- Solar challenge
- Solar Cup
- Blue Sky Solar Racing
- Frisian Solar Challenge
- UC Solar Team
- Solar Splash
- South African Solar Challenge
- Tour de Sol
- World Solar Challenge
- Hunt-Winston School Solar Car Challenge
- North American Solar Challenge
- Victorian Model Solar Vehicle Challenge
Generation systems - Solar Energy Generating Systems
- Stand-alone photovoltaic power system
- Grid-connected photovoltaic power system
- Rooftop photovoltaic power station
- Topaz Solar Farm
- Solar Ark
- Solar Umbrella house
- Erlasee Solar Park
- Guadarranque solar power plant
- Pocking Solar Park
- Copper Mountain Solar Facility
- Wyandot Solar Facility
- Köthen Solar Park
- Building-integrated photovoltaics
- Moura Photovoltaic Power Station
- Nevada Solar One
- Beneixama photovoltaic power plant
- Gottelborn Solar Park
- Darro Solar Park
- Olmedilla Photovoltaic Park
- Blythe Photovoltaic Power Plant
- Strasskirchen Solar Park
- Puertollano Photovoltaic Park
- Alamosa photovoltaic power plant
By countryList of countries by photovoltaics productionPV companies Categories:- Solar cells
- Photovoltaics
Wikimedia Foundation. 2010.