- Disulfur dichloride
-
Disulfur dichloride 
 Disulfur dichlorideSystematic nameDichlorodisulfaneOther namesBis[chloridosulfur](S–S)
Disulfur dichlorideSystematic nameDichlorodisulfaneOther namesBis[chloridosulfur](S–S)
Dimeric sulfenic chloride
Sulfur monochlorideIdentifiers CAS number 10025-67-9  , 85408-26-0 (isobutenate)
, 85408-26-0 (isobutenate)PubChem 24807  , 174464 (isobutenate)
, 174464 (isobutenate) 
ChemSpider 23192  , 152167 (isobutenate)
, 152167 (isobutenate) 
EC number 233-036-2 UN number 3390 MeSH Sulfur+monochloride RTECS number WS4300000 Jmol-3D images Image 1 - ClSSCl
Properties Molecular formula S2Cl2 Molar mass 135.04 g/mol Appearance yellow liquid Density 1.688 g/cm3 Melting point -80 °C, 193 K, -112 °F
Boiling point 137.1 °C, 410 K, 279 °F
Solubility in water decomp with loss of HCl Solubility soluble in ethanol, benzene, ether, chloroform, CCl4 [1] Refractive index (nD) 1.658 Structure Coordination
geometrygauche Dipole moment 1.60 D [1] Hazards MSDS ICSC 0958 EU Index 016-012-00-4 EU classification Toxic (T)
Harmful (Xn)
Corrosive (C)
Dangerous for the environment (N)R-phrases R14, R20, R25, R29, R35, R50 S-phrases (S1/2), S26, S36/37/39, S45, S61 NFPA 704 Flash point 118.5 °C Autoignition
temperature234 °C Related compounds Related sulfur chlorides Sulfur dichloride
Thionyl chloride
Sulfuryl chlorideRelated compounds Disulfur difluoride
Disulfur dibromide (verify) (what is:
(verify) (what is:  /
/ ?)
?)
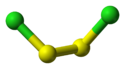
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Disulfur dichloride is the chemical compound with the formula S2Cl2 [2][3][4] [5] . Some alternative names for this compound are sulfur monochloride (the name implied by its empirical formula, SCl), disulphur dichloride (British English Spelling) and sulphur monochloride (British English Spelling). S2Cl2 has the structure implied by the formula Cl-S-S-Cl, wherein the angle between the Cla-S-S and S-S-Clb planes is 90°. This structure is referred to as gauche, and is akin to that for H2O2. A different isomer of S2Cl2 is S=SCl2; this isomer forms transiently when S2Cl2 is exposed to UV-radiation (see thiosulfoxides).
Synthesis and basic properties
Disulfur dichloride is an orange/yellow liquid that smokes in air due to reaction with water:
-
- 2 S2Cl2 + 2 H2O → SO2 + 4 HCl + 3/8 S8
It is synthesized by partial chlorination of elemental sulfur:
-
- S8 + 4 Cl2 → 4 S2Cl2 ΔH = −58.2 kJ/mol
Excess chlorine gives sulfur dichloride:
-
- S2Cl2 + Cl2 → 2 SCl2 ΔH = −40.6 kJ/mol
Both reactions are reversible, and upon standing, SCl2 releases chlorine to revert to the disulfur dichloride. It has the ability to dissolve sulfur, which reflects in part the formation of polysulfanes:
-
- S2Cl2 + n S → S2+nCl2
S2Cl2 also arises from the chlorination of CS2 as in the synthesis of thiophosgene.
Applications
S2Cl2 has been used to introduce C-S bonds. In the presence of AlCl3, S2Cl2 reacts with benzene to give diphenyl sulfide:
-
- S2Cl2 + 2 C6H6 → (C6H5)2S + 2 HCl + 1/8 S8
Anilines react with S2Cl2 in the presence of NaOH via the so-called Herz reaction to give ortho-aminothiophenolates. These species are precursors to thioindigo dyes. It is also used to prepare the sulfur mustard "gas" by reaction with ethylene:
-
- S2Cl2 + 2 C2H4 → (ClC2H4)2S + 1/8 S8
- S2Cl2 + 2 C2H4 → (ClC2H4)2S + 1/8 S8
Other uses include manufacturing sulfur dyes, insecticides, synthetic rubbers. Also used in cold vulcanization of rubbers, as polymerization catalyst for vegetable oils and for hardening soft woods.
References
- ^ a b Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0070494398
- ^ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ^ Hartman, W. W.; Smith, L. A.; Dickey, J. B. (1934), "Diphenylsulfide", Org. Synth. 14: 36, http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv2p0242; Coll. Vol. 2: 242
- ^ R. J. Cremlyn “An Introduction to Organosulfur Chemistry” John Wiley and Sons: Chichester (1996). ISBN 0-471-95512-4
- ^ Garcia-Valverde M., Torroba T. (2006). "Heterocyclic chemistry of sulfur chlorides - Fast ways to complex heterocycles". European J. Org. Chem. 4 (4): 849–861. doi:10.1002/ejoc.200500786.
Categories:- Sulfur chlorides
Wikimedia Foundation. 2010.

