- Sodium diethyldithiocarbamate
-
Sodium diethyldithiocarbamate 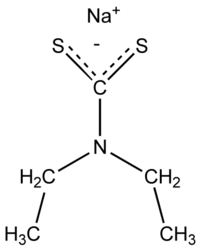 sodium (diethylcarbamothioyl)sulfanide
sodium (diethylcarbamothioyl)sulfanideIdentifiers CAS number 148-18-5 
ChemSpider 8642 
UNII A5304YEB5E 
ChEMBL CHEMBL107217 
Jmol-3D images Image 1 - [Na+].[S-]C(=S)N(CC)CC
Properties Molecular formula C5H10NS2Na Molar mass 171.259 g/mol (anhydrous) Appearance White, slightly brown, or slightly pink crystalline solid Density 1.1 g/cm3 Melting point 95 °C
Solubility in water Soluble Hazards Main hazards Harmful  diethyldithiocarbamate (verify) (what is:
diethyldithiocarbamate (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Sodium diethyldithiocarbamate is the organosulfur compound with the formula NaS2CN(C2H5)2.
Contents
Preparation
This salt is obtained by treating carbon disulfide with diethylamine in the presence of sodium hydroxide:
- CS2 + HN(C2H5)2 + NaOH → NaS2CN(C2H5)2 + H2O
Other dithiocarbamates can be prepared similarly from secondary amines and carbon disulfide. They are used as chelating agents for transition metal ions and as precursors to herbicides and vulcanization reagents.
Oxidation to thiuram disulfide
Oxidation of sodium diethyldithiocarbamate gives the disulfide, also called a thiuram disulfide (Et = ethyl):
- 2 NaS2CNEt2 + I2 → Et2NC(S)S-SC(S)NEt2 + 2 NaI
This disulfide is marketed as an anti-alcoholism drug under the labels Antabuse and Disulfiram. Chlorination of the above-mentioned thiuram disulfide affords the thiocarbamoyl chloride.[1]
Ligand bonding
The diethyldithiocarbamate ion chelates to many "softer" metals via the two sulfur atoms. Other more complicated bonding modes are known including binding as unidentate ligand and a bridging ligand using one or both sulfur atoms.[2]
Spin trapping of nitric oxide radicals
Complexes of Dithiocarbamates with iron provide one of the very few methods to study the formation of nitric oxide (NO) radicals in biological materials. Although the lifetime of NO in tissues is too short to allow detection of this radical itself, NO readily binds to iron-dithiocarbamate complexes. The resulting mono-nitrosyl-iron complex (MNIC) is stable, and may be detected with Electron Paramagnetic Resonance (EPR) spectroscopy.[3][4][5]
In cancer
The effect of diethyldithiocarbamate of chelating zinc inhibits metalloproteinases, which in turn prevents the degradation of extracellular matrix, which is an initial step in cancer metastasis and angiogenesis. [6]
Antioxidant
Diethyldithiocarbamate inhibits superoxide dismutase, which can both have antioxidant and oxidant effects on cells, depending on the time of administration.[6]
References
- ^ Goshorn, R. H.; Levis, Jr., W. W. ;Jaul, E.; Ritter, E. J. (1963), "Diethylthiocarbamyl Chloride", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv4p0307; Coll. Vol. 4: 307
- ^ Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (1999), Advanced Inorganic Chemistry (6th ed.), New York: Wiley-Interscience, ISBN 0-471-19957-5
- ^ Henry Y.; Guissani A.; Ducastel B. (eds); "Nitric oxide research from chemistry to biology: EPR spectroscopy of nitrosylated compounds." Landes, Austin 1997.
- ^ Vanin A.F.; Huisman A.; van Faassen E.E.; "Iron dithiocarbamates as spin trap for nitric oxide: Pitfalls and successes." Methods in Enzymology vol 359 (2002) 27 - 42.
- ^ van Faassen E.E.; Vanin A.F. (eds); "Radicals for life: The various forms of nitric oxide." Elsevier, Amsterdam 2007.
- ^ a b diethyldithiocarbamate National Cancer Institute - Drug Dictionary
Further reading
- Cvek B, Dvorak Z (2007). "Targeting of nuclear factor-kappaB and proteasome by dithiocarbamate complexes with metals". Curr. Pharm. Des. 13 (30): 3155–67. doi:10.2174/138161207782110390. PMID 17979756. http://www.bentham-direct.org/pages/content.php?CPD/2007/00000013/00000030/0010B.SGM.
Categories:- Sodium compounds
- Chelating agents
- Dithiocarbamates
Wikimedia Foundation. 2010.
