- Mercury(II) thiocyanate
-
Mercury(II) thiocyanate 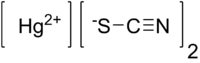 Other namesMercuric thiocyanate
Other namesMercuric thiocyanate
Mercuric sulfocyanateIdentifiers CAS number 592-85-8 Properties Molecular formula Hg(SCN)2 Molar mass 316.79 g/mol Appearance White monoclinic crystals Density 3.71 g/cm³, solid Melting point Decomposes at 165 °C
Solubility in water 0.070 g/100 mL Solubility in other solvents Soluble in dilute hydrochloric acid[1] Hazards NFPA 704  thiocyanate (verify) (what is:
thiocyanate (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Mercury(II) thiocyanate (Hg(SCN)2) is an inorganic chemical compound, the salt of Hg2+ and the thiocyanate anion. It is a stable solid at room temperature that has the appearance of white powder with chunks; it can also be grey in color, depending on purity. Mercury compounds are extremely toxic and protective equipment should be used whenever working with mercury thiocyanate. However, it is commercially available, though expensive.[2] Mercury thiocyanate is best known for its former use in pyrotechnics, as it will produce a large, winding “snake” when set on fire. This is known as the Pharaoh’s Serpent.[3] Though some people still use it for this purpose, it is generally avoided because of the production of toxic gases when this reaction occurs.
Contents
Synthesis
The first synthesis of mercury thiocyanate was probably completed in 1821 by the chemist Jöns Jacob Berzelius with evidence for the first pure sample occurring in 1866 prepared by a chemist named Hermes.[3] Because of its ionic nature, there are several ways to synthesize the compound. Mercury(II) thiocyanate is made by reacting solutions containing mercury(II) and thiocyanate ions. The low solubility product of mercury thiocyanate causes it to precipitate. It is also soluble in several solvents including benzene, hexanes, and methyl isobutyl ketone.[4] Most syntheses are achieved by precipitation. The two early syntheses achieved separately by Berzelius and Friedrich Wöhler were completed using the following reactions:
- 2 HSCN + HgO → Hg(SCN)2 + H2O (Berzelius)
- Hg(NO3)2 (aq) + 2 KSCN (aq) → Hg(SCN)2 (s) + 2KNO3 (aq) (Wohler)
Pharaoh's Serpent
Mercury thiocyanate was formerly used in pyrotechnics causing an effect known as the Pharaoh’s serpent or Pharaoh’s snake. When the compound is in the presence of a strong enough heat source, a rapid exothermic reaction is started which produces a large mass of coiling serpent-like solid. An inconspicuous flame which is often blue but can also occur in yellow/orange accompanies the combustion. The resulting solid can range from dark graphite grey to light tan in color with the inside generally much darker than the outside.[3]
This property was discovered soon after the first synthesis of mercury thiocyanate by Wohler in 1821: “winding out from itself at the same time worm-like processes, to many times its former bulk, a very light material the color of graphite…”. For some time, a firework product called “Pharaoschlangen” was available to the public in Germany, but was eventually banned when the toxic properties of the product were discovered through the death of several children mistakenly eating the resulting solid.[3]
A similar, though less extreme, effect to the Pharaoh’s Serpent can be achieved using a firework known as a black snake. These are generally benign products consisting usually of sodium bicarbonate or a mixture of linseed oil and napthalenes.
Uses and Chemical Properties
Mercury thiocyanate has a few uses in chemical synthesis. It is often involved in the synthesis of related compounds that contain mercury (II) and/or thiocyanates. These compounds include, but are not limited to, potassium tris(thiocyanato)mercurate(II) (K[Hg(SCN)3]) and caesium tris(thiocyanato)mercurate(II) (Cs[Hg(SCN)3]). The Hg(SCN)3- ion can also exist independently and is easily reacted to form the compounds above amongst others. These compounds can be examined analytically using infrared spectroscopy, Raman spectroscopy and solid state NMR.[5]
It can also be used in organic synthesis, to replace a halide on an organic compound with a thiocyanate group through a Sn2 mechanism. However, this reaction does not create one pure product as the SCN- can react on either end with the organohalide. This means that such a reaction would yield two distinct products, one with the sulfur bound to the organic compound and one with the nitrogen bound to the organic compound.[6]
It was discovered that mercury thiocyanate can improve detection limits in the determination of chloride ions in water by UV-visible spectroscopy. This technique was first suggested in 1952 and has been a common method for determination of chloride ions in laboratories worldwide ever since. An automated system was invented in 1964 and then a commercial chloroanalyzer was made available in 1974 by Technicon (Tarrytown, NY, USA). The basic mechanism involves the addition of mercury thiocyanate to a solution with unknown concentration of chloride ions and iron as a reagent. The chloride irons cause the mercury thiocyanate salt to dissociate and the thiocyanate ion to complex with Fe(III), producing Fe(SCN)2+, which absorbs visible light at 450 nm. This absorption allows for the measurement of concentration of Fe(SCN)2+, produced as a result of the reaction between chloride ion and mercury thiocyanate. From this value the concentration of chloride can then be calculated.[7]
In 1995, a new method for determining the concentration of chloride ions in aqueous solution using mercury thiocyanate was discovered. Mercury thiocyanate without iron (III) is added to a solution with an unknown concentration of chloride ions, forming a complex of the mercury thiocyanate and chloride ion that absorbs light at a wavelength of 254 nm, allowing more accurate measurements of concentration than the aforementioned technique using iron.[7]
References
- ^ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, FL: CRC Press. pp. 4–70. ISBN 0-8493-0594-2
- ^ Sigma Aldrich. Mercury Thiocyanate. http://www.sigmaaldrich.com/catalog (accessed 8 April 2010).
- ^ a b c d Davis, T. L. (1940). "Pyrotechnic Snakes". J. Chem. Educ. 17 (6): 268–270. doi:10.1021/ed017p268.
- ^ Sekine, T., Ishii, T. Studies of the Liquid-Liquid Partition systems. VIII. The Solvent Extraction of Mercury (II) Chloride, Bromide, Iodide and Thiocyanate with Some Organic Solvents. Bulletin of the Chemical Society of Japan. 1970. 43. 2422-2429.
- ^ Bowmaker, G. A., Churakov, A. V., Harris, R. K., Howard, J. A. K., Apperley, D. C. Solid-State 199Hg MAS NMR Studies of Mercury(II) Thiocyanate Complexes and Related Compounds. Crystal Structure of Hg(SeCN)2 Inorg. Chem. 1998, 37, 1734-1743.
- ^ Kitamura, T., Kobayashi, S., Taniguchi, H. Photolysis of Vinyl Halides. Reaction of Photogenerated Vinyl Cations with Cyanate and Thiocyanate Ions. J. Org. Chem. 1990. 55, 1801-1805.
- ^ a b Cirello-Egamino, J; Brindle, I D. Determination of chloride ions by reaction with mercury thiocyanate in the absence of iron(III) using a UV-photometric, flow injection method. Analyst. 1995. 120. 183-186.
External links
Mercury compounds Categories:- Pyrotechnic chemicals
- Thiocyanates
- Mercury compounds
Wikimedia Foundation. 2010.

