- Dimethylsulfoniopropionate
-
Dimethylsulfoniopropionate 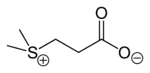 (Dimethylsulfaniumyl)propanoateOther namesdimethyl-β-propiothetin
(Dimethylsulfaniumyl)propanoateOther namesdimethyl-β-propiothetinIdentifiers CAS number 7314-30-9 PubChem 23736 ChemSpider 11207615 
Jmol-3D images Image 1
Image 2- C[S+](C)CCC(=O)[O-]
C[S+](C)C(C)C([O-])=O
Properties Molecular formula C5H10O2S Molar mass 134.1967  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dimethylsulfoniopropionate (frequently abbreviated to DMSP), is an organosulfur compound with the formula (CH3)2S+CH2CH2COO−. This zwitterionic metabolite found in marine phytoplankton, seaweeds, and some species of terrestrial and aquatic vascular plants. It functions as an osmolyte as well as several other physiological and environmental roles have also been identified.[1]
Contents
Biosynthesis
In higher plants, DMSP is biosynthesized from S-methylmethionine. Two intermediates in this conversion are dimethylsulfoniumpropylamine and dimethylsulfoniumpropionaldehyde.[2] In algae, however, the biosynthesis starts with removal of the amino group from methionine, rather than from S-methylmethionine.
Degradation
DMSP is broken down by marine microbes to form two major volatile sulfur products, each with distinct effects on the environment. Its major breakdown product is methanethiol (CH3SH), which is assimilated by bacteria into protein sulfur. Its second volatile breakdown product is dimethyl sulfide (CH3SCH3; DMS). Most DMS in seawater is cleaved from DMSP by the enzyme DMSP lyase, although many non-marine species of bacteria convert methanethiol to DMS.[citation needed]
DMS is also taken up by marine bacteria, but not as rapidly as methanethiol. Although DMS usually consists of less than 25% of the volatile breakdown products of DMSP, the high reactivity of methanethiol makes the steady-state DMS concentrations in seawater approximately 10 times those of methanethiol (~3 nM vs. ~0.3 nM). Curiously, there have never been any published correlations between the concentrations of DMS and methanethiol. This is probably due to the non-linear abiotic and microbial uptake of methanethiol in seawater, and the comparatively low reactivity of DMS. However, a significant portion of DMS in seawater is oxidized to dimethyl sulfoxide (DMSO).
Relevant to global climate, DMS is thought to play a role in the Earth's heat budget by decreasing the amount of solar radiation that reaches the Earth's surface.
DMSP has also been implicated in influencing the taste and odour characteristics of various products. For example, although DMSP is odourless and tasteless, it is accumulated at high levels in some marine herbivores or filter feeders. Increased growth rates, vigour and stress resistance among animals cultivated on such diets have been reported.[citation needed] DMS, is responsible for repellent, 'off' tastes and odours that develop in some seafood products because of the action of bacterial DMSP-lyase, which cogenerates acrylate.
See also
- CLAW hypothesis, proposing a feedback loop that operates between ocean ecosystems and the Earth's climate
- Coccolithophore, a group of marine unicellular planktonic photosynthetic algae, producer of DMSP
- Dimethyl sulfide, a breakdown product of DMSP along with methanethiol
- Dimethyl selenide, a selenium analogue of DMS produced by bacteria and phytoplankton
- Emiliania huxleyi, a coccolithophorid producing DMSP
References
- ^ DeBose, Jennifer L.; Sean C. Lema, Gabrielle A. Nevitt (2008-03-07). "Dimethylsulfoniopropionate as a foraging cue for reef fishes" (abstract). Science 319 (5868): 1356. doi:10.1126/science.1151109. PMID 18323445. http://www.sciencemag.org/cgi/content/abstract/319/5868/1356. Retrieved 2008-03-21.Vila-Costa, Maria; Rafel Simo, Hyakubun Harada, Josep M. Gasol, Doris Slezak, Ronald P. Kiene (2006-10-27). "Dimethylsulfoniopropionate uptake by marine phytoplankton" (abstract). Science 314 (5799): 652–654. doi:10.1126/science.1131043. PMID 17068265. http://www.sciencemag.org/cgi/content/abstract/314/5799/652. Retrieved 2008-03-21.
- ^ Scott D. McNeil, Michael L. Nuccio, and Andrew D. Hanson "Betaines and Related Osmoprotectants. Targets for Metabolic Engineering of Stress Resistance" Plant Physiology, August 1999, Vol. 120, pp. 945–949. doi:10.1104/pp.120.4.945
External links
Categories:- Thioethers
- Carboxylate anions
- Planktology
- C[S+](C)CCC(=O)[O-]
Wikimedia Foundation. 2010.
