- Tannic acid
-
Tannic acid 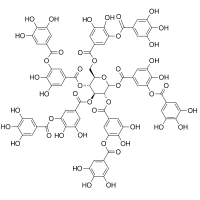 2,3-dihydroxy-5-({[(2R,3R,4S,5R,6R)-3,4,5,6-tetrakis({3,4-dihydroxy-5-[(3,4,5-trihydroxyphenyl)carbonyloxy]phenyl}carbonyloxy)oxan-2-yl]methoxy}carbonyl)phenyl 3,4,5-trihydroxybenzoateOther namesAcidum tannicum
2,3-dihydroxy-5-({[(2R,3R,4S,5R,6R)-3,4,5,6-tetrakis({3,4-dihydroxy-5-[(3,4,5-trihydroxyphenyl)carbonyloxy]phenyl}carbonyloxy)oxan-2-yl]methoxy}carbonyl)phenyl 3,4,5-trihydroxybenzoateOther namesAcidum tannicum
Gallotannic acid
Digallic acid
Gallotannin
Tannimum
Quercitannin
Oak bark tannin
Quercotannic acid
Querci-tannic acid
Querco-tannic acidIdentifiers CAS number 1401-55-4 
PubChem 16129878 KEGG C13452 
ChEMBL CHEMBL506247 
Beilstein Reference 8186386 Jmol-3D images Image 1 - Oc1cc(cc(O)c1O)C(=O)Oc1cc(cc(O)c1O)C(=O)OC[C@H]1O[C@H](OC(=O)c2cc(O)c(O)c(OC(=O)c3cc(O)c(O)c(O)c3)c2)[C@H](OC(=O)c2cc(O)c(O)c(OC(=O)c3cc(O)c(O)c(O)c3)c2)[C@@H](OC(=O)c2cc(O)c(O)c(OC(=O)c3cc(O)c(O)c(O)c3)c2)[C@@H]1OC(=O)c1cc(O)c(O)c(OC(=O)c2cc(O)c(O)c(O)c2)c1
Properties Molecular formula C76H52O46 Molar mass 1701.2 g mol−1 Exact mass 1700.172974 Melting point decomposes above 200°C
Solubility in water 2850 g/L Acidity (pKa) ca. 10  acid (verify) (what is:
acid (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Tannic acid is a specific commercial form of tannin, a type of polyphenol. Its weak acidity (pKa around 6) is due to the numerous phenol groups in the structure. The chemical formula for commercial tannic acid is often given as C76H52O46, which corresponds with decagalloyl glucose, but in fact it is a mixture of polygalloyl glucoses or polygalloyl quinic acid esters with the number of galloyl moieties per molecule ranging from 2 up to 12 depending on the plant source used to extract the tannic acid. Commercial tannic acid is usually extracted from any of the following plant part: Tara pods (Caesalpinia spinosa), gallnuts from Rhus semialata or Quercus infectoria or Sicilian Sumac leaves (Rhus coriaria).
According to the definitions provided in external references such as international pharmacopoeia, Food Chemical Codex and FAO-WHO tannic acid monograph only tannins sourced from the above mentioned plants can be considered as tannic acid. Sometimes extracts from chestnut or oak wood are also described as tannic acid but this is an incorrect use of the term. It is a yellow to light brown amorphous powder which is highly soluble in water; one gram dissolves in 0.35 mL of water.
While 'tannic acid' is a specific type of 'tannin' (plant polyphenol), the two terms are sometimes (incorrectly) used interchangeably. The long standing misuse of the terms, and its inclusion in scholarly articles have compounded the confusion. This is particularly widespread in relation to green tea and black tea. Although tea contains tannin and various types of polyphenols, "contrary to widespread belief, tea does not contain tannic acid." [1]
Tannic acid is not an appropriate standard for any type of tannin analysis because of its poorly defined composition.[2]
Contents
Quercitannic and gallotannic acids
Quercitannic acid is one of the two forms of tannic acid[3] found in oak bark and leaves.[4] The other form is called gallotannic acid and is found in oak galls.
The quercitannic acid molecule is also present in quercitron, a yellow dye obtained from the bark of the Eastern black oak (Quercus velutina), a forest tree indigenous in North America. It is described as as a yellowish brown amorphous substance.[5]
In 1838, Jöns Jacob Berzelius wrote that quercitannate is used to dissolve morphine.[6]
In 1865 in the fifth volume of "A dictionary of chemistry", Henry Watts wrote :
It exhibits with ferric salts the same reactions as gallotannic acid. It differs however from the latter in not being convertible into gallic acid, and not yielding pyrogallic acid by dry distillation. It is precipitated by sulfuric acid in red flocks. (Stenhouse, Ann. Ch. Pharm. xlv. 16.)
According to Rochleder (ibid lxiii. 202), the tannic acid of black tea is the same as that of oak-bark.[7]In 1880, Etti gave for it the molecular formula C17H16O9. He described it as an unstable substance, having a tendency to give off water to form anhydrides (called phlobaphenes), one of which is called oak-red (C34H30O17). For him, it was not a glycoside.[8][9]
In Allen’s "Commercial Organic Analysis", published in 1912, the formula given was C19H16O10.[10]
Other authors gave other molecular formulas like C28H26O15, while another formula found is C28H24O11.[11]
According to Lowe, two forms of the principle exist - one soluble in water, of the formula C28H28O14, and the other scarcely soluble, C28H24O12. Both are changed by the loss of water into oak red, C28H22O11.[12]
Quercitannic acid was for a time a standard used to assess the phenolic content in spices, given as quercitannic acid equivalent.[13]
Uses
Tannins are a basic ingredient in the chemical staining of wood, and are already present in woods like oak, walnut, and mahogany. Tannic acid can be applied to woods low in tannin so chemical stains that require tannin content will react. The presence of tannins in the bark of redwood (Sequoia) is a strong natural defense against wildfire, decomposition and infestation by certain insects such as termites. It is found in the seeds, bark, cones, and heartwood.
Tannic acid is a common mordant used in the dyeing process for cellulose fibers such as cotton, often combined with alum and/or iron. The tannin mordant should be done first as metal mordants combine well with the fiber-tannin complex. However this use has lost considerable interest.
Similarly tannic acid can also be used as an aftertreatment to improve wash fastness properties of acid dyed polyamide. It is also an alternative for fluorcarbon aftertreatments to impart anti-staining properties to polyamide yarn or carpets. However, due to economic considerations currently the only widespread use as textile auxiliary is the use as an agent to improve chlorine fastness (i.e. resistance against dye bleaching due to cleaning with hypochlorite solutions in high-end polyamide 6,6-based carpets and swimwear.
A particular and unique nich textile application of tannic acid is the activation of flock, which is basically an anti-static aftertreatment.
Tannic acid is used in the conservation of ferrous (iron based) metal objects to passivate and inhibit corrosion. Tannic acid reacts with the corrosion products to form a more stable compound, thus preventing further corrosion from taking place. After treatment the tannic acid residue is generally left on the object so that if moisture reaches the surface the tannic acid will be rehydrated and prevent or slow any corrosion. Tannic acid treatment for conservation is very effective and widely used but it does have a significant visual effect on the object, turning the corrosion products black and any exposed metal dark blue. It should also be used with care on objects with copper alloy components as the tannic acid can have a slight etching effect on these metals.
Tannic acid is also found in commercially available iron/steel corrosion treatments such as Hammerite Kurust.
Use in food
Use of tannic acid in food applications is far more widespread and significant amounts are used as process aids in beer clarification, aroma compound in soft drinks and juices. Equally important are applications in the wine industry, where it finds use as a natural clarifying agent, colour stabilizer and taste enhancer.
In many parts of the world such uses are permitted. In the United States, tannic acid is generally recognized as safe by the Food and Drug Administration.
According to EU directive 89/107/EEC tannic acid cannot be considered as a food additive and consequently does not hold an E number. Under directive 89/107/EEC tannic acid can be referred to as a food ingredient. The E-number E181 is sometimes incorrectly used to refer to tannic acid; this in fact refers to the INS number assigned to tannic acid under the FAO-WHO Codex Alimentarius system.
Uses as a medication
Tannic acid was once used as a treatment for many toxic substances, such as strychnine, mushroom, and ptomaine poisonings in the late 19th and early 20th centuries.[14]
The introduction of tannic acid treatment of severe burn injuries in the 1920s significantly reduced mortality rates[15] During World War I, tannic acid dressings were prescribed to treat "burns, whether caused by incendiary bombs, mustard gas or lewisite... Where tannic acid is not available, strong, lukewarm tea is a good substitute."[16] After the war this use was abandoned due to the development of more modern treatment regimens.
Today tannic acid is still used in pharmaceutical applications to produce albumine tannate which is used as an anti-diarrhea agent. Tannic acid is also used to produce tannate salts of certain anti-histamins and anti-tussives to impart increased stability or slow release properties to the API (active pharmaceutical ingredient).
There are many unverified and/or dubious claims about tannic acid, such as that soaking feet in tannic acid (or strong tea) can treat or prevent blisters; foot odor; and rough, dry feet.[17][18] Other claims about tannic acid seem to either add to, or have been created by the confusion between tannic acid and tannin. One example is that "A popular home remedy to stop the bleeding after wisdom tooth extraction is applying tea bags in the back of the jaws and biting down, given that the tannic acid [sic] in tea helps to clot blood."
Tannic acid glycerite is used for local application.[19]
Trivia
Skin color in Crocodilia (crocodiles and alligators) is very dependent on water quality. Algae-laden waters produce greener skin, while tannic acid from overhanging trees often produce darker skin.
References
- ^ Hamilton-Miller JM (1 November 1995). "Antimicrobial properties of tea (Camellia sinensis L.)". Antimicrob. Agents Chemother. 39 (11): 2375–7. PMC 162950. PMID 8585711. http://aac.asm.org/cgi/pmidlookup?view=long&pmid=8585711.
- ^ Tannin Chemistry pg 11
- ^ L'Energie Homo-Hydrogne By Patricia Le Roux (French)
- ^ Quercus on www.henriettesherbal.com
- ^ Quercitannic on everything2.com
- ^ Traité de chimie, Volume 2. Jöns Jakob Berzelius (friherre) and Olof Gustaf Öngren, A. Wahlen et Cie., 1838
- ^ A dictionary of chemistry, Volume 5, by Henry Watts, 1865
- ^ Über die Gerbsäure der Eichenrinde. C. Etti, Monatshefte für Chemie, Volume 1, Number 1, 262-278, 1880 doi:10.1007/BF01517069 (German)
- ^ Zur Geschichte der Eichenrindegerbsäuren. C. Etti, Monatshefte für Chemie, Volume 4, Number 1, 512-530, 1883 doi:10.1007/BF01517990 (German)
- ^ The nature of tea infusions. Henry L. Smith, The Lancet, Volume 181, Issue 4673, 22 March 1913, Page 846, doi:10.1016/S0140-6736(01)03766-7
- ^ What are the historic and contemporary ethnobotanical uses of native Rhode Island wetlands plants? by Courtney Reckford, 1997
- ^ A manual of organic materia medica and pharmacognosy - An introduction to the study of the vegetable Kingdom and the vegetable and animal drugs (Sayre's Materia Medica) fourth edition , by Lucius E Sayre
- ^ Government standards for spices. Journal of the American Pharmaceutical Association, Volume 12, Issue 12, pages 1091–1094,December 1923, doi:10.1002/jps.3080121212
- ^ Sturmer, J.W., and . "Pharmaceutical Toxicity." The Pharmaceutical Era. Vol 21. New York: D.O. Haynes & Co., 1899,pg 511-13.
- ^ S.B.A. Halkens et al., "The use of tannic acid in the local treatment of burn wounds: intriguing old and new perspectives.", Wounds, 2001, 13(4), p 144-158.
- ^ "Medicine: War Wounds", TIME, 18 September 1939, http://www.time.com/time/magazine/article/0,9171,762600-2,00.html
- ^ Buck. "Medicine Man". Backpacker August 2003: 63.
- ^ Cox, L., & Cox, J. (2009). Ecobeauty: Scrubs, rubs, masks, and bath bombs for you and your friends. Berkeley [Calif.]: Ten Speed Press, pg 219.
- ^ Tanic acid glycerite on www.henriettesherbal.com
General references
The Merck Index, 9th edition, Merck & Co., Rahway, New Jersey, 1976.
External links
- "Medicine: Tea & the Atom" at TIME.com
- "Tannin chemistry" (PDF, 1.45 MB)
- "tea does not tan leather"
- "Dental Medicine. A Manual Of Dental Materia Medica And Therapeutics", by Ferdinand J. S. Gorgas
Aglycones Examples Burkinabin A | B | C | Galloyl glucose | Digalloyl glucose | Trigalloyl glucose: 1,2,6-Trigalloyl glucose / 1,3,6,-Trigalloyl glucose (Corilagin) | Tetragalloyl glucose: 1,2,3,6-tetragalloylglucose | Pentagalloyl glucose: 1,2,3,4,6-pentagalloyl-glucose and 6-digalloyl-1,2,3-trigalloyl-glucose | Hexagalloyl glucose | Heptagalloyl glucose | Octagalloyl glucose | Nonagalloyl glucose | Decagalloyl glucose | 1,3,4-Tri-O-galloylquinic acid | 3,4,5-Tri-O-galloylquinic acid | 3,5-di-O-galloyl-shikimic acid | 3,4,5-tri-O-galloylshikimic acid | 1,2,6-trigalloyl alloside | 1,3,6-trigalloyl alloside | 1,2,3,6-tetragalloyl allosideCategories:- Gallotannins
- Astringent flavors
Wikimedia Foundation. 2010.

