- Nisin
-
Nisin 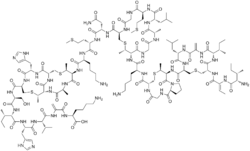

Identifiers CAS number 1414-45-5 
PubChem 16219761 ChemSpider 21106355 
UNII EN8XKG133D 
ChEMBL CHEMBL526744 
Jmol-3D images Image 1 - C[C@H](CC)[C@@H](N)C(N/C(C(N[C@@H]1C(N[C@@H](C(NC(C(N[C@@H](C(N[C@@H](C(N[C@H](C(N2[C@@H](C(NC3)=O)CCC2)=O)[C@@H](C)SC[C@H](C(N[C@H](CCCCN)C(N[C@H](C(NCC(N[C@@H](C(N[C@H](CC(C)C)C(N[C@H](CCSC)C(NCC(N[C@@H](C(N[C@H](CC(N)=O)C(N[C@H](CCSC)C(N[C@H](CCCCN)C(N[C@@H]([C@@H](C)SC[C@@H](N6)C(N[C@H](CC5=CN=CN5)C(N[C@@H](C(N[C@H](CO)C(N[C@H]([C@@H](CC)C)C(N[C@H](CC8=CN=CN8)C(N[C@H]([C@@H](C)C)C(NC(C(N[C@H](CCCCN)C(O)=O)=O)=C)=O)=O)=O)=O)=O)CS[C@H](C)[C@H]7C6=O)=O)=O)C(N[C@H](C)C(N7)=O)=O)=O)=O)=O)=O)CS4)=O)=O)=O)=O)C)=O)=O)[C@H]4C)=O)=O)NC3=O)=O)CSC1)=O)CC(C)C)=O)=C)=O)[C@H](C)CC)=O)=O)=C\C)=O
- InChI=1S/C143H246N42O45S7/c1-22-68(10)104(148)134(220)183-111(76(18)193)140(226)173-93(57-189)127(213)179-107(70(12)24-3)138(224)172-91(55-187)125(211)166-86(45-66(6)7)120(206)175-98(62-235)132(218)184-112(77(19)194)142(228)185-41-31-35-99(185)133(219)152-53-103(198)158-94(58-231)128(214)161-81(33-26-29-39-145)117(203)181-108(73(15)190)135(221)153-51-101(196)156-71(13)113(199)165-85(44-65(4)5)119(205)162-82(36-42-236-20)115(201)151-52-102(197)159-95(59-232)129(215)168-89(48-100(147)195)122(208)163-83(37-43-237-21)116(202)160-80(32-25-28-38-144)118(204)182-109(74(16)191)139(225)157-72(14)114(200)180-110(75(17)192)141(227)176-97(61-234)130(216)167-87(46-78-49-149-63-154-78)121(207)174-96(60-233)131(217)170-92(56-188)126(212)178-106(69(11)23-2)137(223)169-88(47-79-50-150-64-155-79)123(209)177-105(67(8)9)136(222)171-90(54-186)124(210)164-84(143(229)230)34-27-30-40-146/h49-50,63-77,80-99,104-112,186-194,231-235H,22-48,51-62,144-146,148H2,1-21H3,(H2,147,195)(H,149,154)(H,150,155)(H,151,201)(H,152,219)(H,153,221)(H,156,196)(H,157,225)(H,158,198)(H,159,197)(H,160,202)(H,161,214)(H,162,205)(H,163,208)(H,164,210)(H,165,199)(H,166,211)(H,167,216)(H,168,215)(H,169,223)(H,170,217)(H,171,222)(H,172,224)(H,173,226)(H,174,207)(H,175,206)(H,176,227)(H,177,209)(H,178,212)(H,179,213)(H,180,200)(H,181,203)(H,182,204)(H,183,220)(H,184,218)(H,229,230)

Key: WAMGWAJCUSJZNI-UHFFFAOYSA-N
InChI=1/C143H246N42O45S7/c1-22-68(10)104(148)134(220)183-111(76(18)193)140(226)173-93(57-189)127(213)179-107(70(12)24-3)138(224)172-91(55-187)125(211)166-86(45-66(6)7)120(206)175-98(62-235)132(218)184-112(77(19)194)142(228)185-41-31-35-99(185)133(219)152-53-103(198)158-94(58-231)128(214)161-81(33-26-29-39-145)117(203)181-108(73(15)190)135(221)153-51-101(196)156-71(13)113(199)165-85(44-65(4)5)119(205)162-82(36-42-236-20)115(201)151-52-102(197)159-95(59-232)129(215)168-89(48-100(147)195)122(208)163-83(37-43-237-21)116(202)160-80(32-25-28-38-144)118(204)182-109(74(16)191)139(225)157-72(14)114(200)180-110(75(17)192)141(227)176-97(61-234)130(216)167-87(46-78-49-149-63-154-78)121(207)174-96(60-233)131(217)170-92(56-188)126(212)178-106(69(11)23-2)137(223)169-88(47-79-50-150-64-155-79)123(209)177-105(67(8)9)136(222)171-90(54-186)124(210)164-84(143(229)230)34-27-30-40-146/h49-50,63-77,80-99,104-112,186-194,231-235H,22-48,51-62,144-146,148H2,1-21H3,(H2,147,195)(H,149,154)(H,150,155)(H,151,201)(H,152,219)(H,153,221)(H,156,196)(H,157,225)(H,158,198)(H,159,197)(H,160,202)(H,161,214)(H,162,205)(H,163,208)(H,164,210)(H,165,199)(H,166,211)(H,167,216)(H,168,215)(H,169,223)(H,170,217)(H,171,222)(H,172,224)(H,173,226)(H,174,207)(H,175,206)(H,176,227)(H,177,209)(H,178,212)(H,179,213)(H,180,200)(H,181,203)(H,182,204)(H,183,220)(H,184,218)(H,229,230)
Key: WAMGWAJCUSJZNI-UHFFFAOYAB
Properties Molecular formula C143H230N42O37S7 Molar mass 3354.07 g/mol  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Nisin is a polycyclic antibacterial peptide with 34 amino acid residues used as a food preservative. It contains the uncommon amino acids lanthionine (Lan), methyllanthionine (MeLan), didehydroalanine (Dha) and didehydroaminobutyric acid (Dhb). These unusual amino acids are introduced by posttranslational modification of the precursor peptide. In these reactions a ribosomally synthesized 57-mer is converted to the final peptide. The unsaturated amino acids originate from serine and threonine, and the enzyme-catalysed addition of cysteine residues to the didehydro amino acids result in the multiple thioether bridges.
Nisin is produced by fermentation using the bacterium Lactococcus lactis. Commercially, it is obtained from the culturing of Lactoccus lactis on natural substrates, such as milk or dextrose, and is not chemically synthesized. It is used in processed cheese, meats, beverages, etc. during production to extend shelf life by suppressing Gram-positive spoilage and pathogenic bacteria. While most bacteriocins generally inhibit only closely related species, Nisin is a rare example of a "broad-spectrum" bacteriocin effective against many Gram-positive organisms, including lactic acid bacteria (commonly associated with spoilage), Listeria monocytogenes (a known pathogen), etc. However, when coupled with the chelator EDTA, Nisin has also been known to inhibit Gram-negative bacteria, as well. Nisin is soluble in water and can be effective at levels nearing the parts per billion range. In foods, it is common to use Nisin at levels ranging from ~1-25ppm, depending on the food type and regulatory approval. Due to its naturally selective spectrum of activity, it is also employed as a selective agent in microbiological media for the isolation of gram-negative bacteria, yeast, and moulds. Subtilin and Epidermin are related to Nisin. All are members of a class of molecules known as lantibiotics.
As a food additive, nisin has E number E234.
Further reading
- K. Fukase et al., Tetrahedron Lett. 1988, 29, 7, 795. (Total synthesis)
- G. W. Buchman et al., J. Biol. Chem. 1988, 263, 31, 16260. (Biosynthesis)
External links
Antimicrobial cationic peptides Other, human Other, nonhuman Categories:- Lantibiotics
- Preservatives
- Peripheral membrane proteins
Wikimedia Foundation. 2010.
