- Methyl carbamate
-
Methyl carbamate 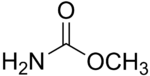
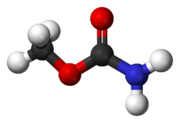 Methyl carbamate
Methyl carbamateIdentifiers CAS number 598-55-0 
PubChem 11722 ChemSpider 11229 
KEGG C19445 
ChEMBL CHEMBL1085707 
Jmol-3D images Image 1 - O=C(OC)N
Properties Molecular formula C2H5NO2 Molar mass 75 g/mol Melting point 52 °C, 325 K, 126 °F
Boiling point 177 °C, 450 K, 351 °F
 carbamate (verify) (what is:
carbamate (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Methyl carbamate (also called methylurethane, or urethylane) is an organic compound and the simplest ester of the hypothetical carbamic acid (NH2COOH). Its sum formula is C2H5NO2.
Methyl carbamate is formed by the reaction of ammonia with methyl chloroformate or methyl carbonate. According to US patent number 2834799 urea can be reacted with methanol to form it using boron trifluoride as a reagent. Unlike its close relative ethyl carbamate it is not mutagenic in salmonella (it tested negative in the Ames test), but it is mutagenic in Drosophila.[1] Experimental evidence does show that it is a carcinogen in rat, and not carcinogenic in mice. The compound is "known to the state of California to cause cancer" per Proposition 65.[2]
The compound was detected in wines preserved with dimethyl dicarbonate.[3]
Methyl carbamate is used by the textile industry to manufacture resins to be applied on polyester/cotton blend fabrics as durable-press finishes.[4]
N-Methyl carbamates are widely used as insecticides.[5]
See also
References
- ^ P. Foureman, J.M. Mason, R. Valencia and S. Zimmering, Environ. Mol. Mutagen., 1994, 23 (1), 51 - 63.
- ^ OEHHA
- ^ Inchem.org
- ^ National Toxocology Program
- ^ National Pesticide Information Center at Oregon State University
External links
Categories:- Methyl esters
- Carbamates
Wikimedia Foundation. 2010.
