- Dimethyl dicarbonate
-
Dimethyl dicarbonate 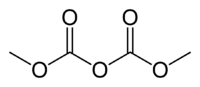
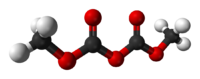 methoxycarbonyl methyl carbonateOther namesDMDC
methoxycarbonyl methyl carbonateOther namesDMDC
dicarbonic acid
dimethyl ester
dimethyl pyrocarbonate
VelcorinIdentifiers CAS number 4525-33-1 
PubChem 3086 ChemSpider 2976 
Jmol-3D images Image 1
Image 2- O=C(OC(=O)OC)OC
O=C(OC)OC(=O)OC
Properties Molecular formula C4H6O5 Molar mass 134.09 g mol−1 Appearance Colourless liquid Density 1.25 g/ml (liquid) Melting point 16-18 °C, 289-291 K, 61-64 °F
Boiling point 172 °C, 445 K, 342 °F
Viscosity 2.1 Pa·s (20 °C) Hazards R-phrases R22 R23 R34 S-phrases S7/9 S26 S36/37/39 S45 Main hazards Toxic  dicarbonate (verify) (what is:
dicarbonate (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dimethyl dicarbonate or DMDC is a colourless liquid with a sharp odour. Its primary use is as a beverage preservative and or processing aid or sterilant (INS No. 242), and acts by inhibiting the enzymes acetate kinase and L-glutamic acid decarboxylase.[1] It has also been proposed that methoxycarbonylation of the histidine part of the enzymes alcohol dehydrogenase and glyceraldehyde 3-phosphate dehydrogenase by DMDC inhibits these essential enzymes also.[2] Once it has been added to beverages, the efficacy of the chemical is provided by the following reactions:
- DMDC + H2O → 2CH3OH + 2CO2
- DMDC + EtOH → Ethyl methyl carbonate
- DMDC + NH3 → Methyl carbamate
- DMDC + Amino acid → Derived carboxymethyl
DMDC is sometimes used as a preservative in wine as a replacement to sulfur dioxide, inactivating wine spoilage yeasts such as Brettanomyces. In the U.S. the FDA approved its use in wines in 1988, with the maximum permitted level being set at 200 mg/L, and only if there were fewer than 500 yeast cells/mL at time of dosage. It is listed as approved in the EU[3] (where it is listed under E number E242) and USA[4] but is not listed in Australia and New Zealand.[5] The application of DMDC is particularly useful when wine needs to be sterilised but cannot be sterile filtered, pasteurized, or sulfured.
DMDC is used to stabilise non alcoholic beverages such as carbonated or non carbonated juice beverages, isotonic sports beverages, iced teas and flavoured waters.
DMDC is added before the filling of the beverage. It then breaks down into small amounts of methanol and carbon dioxide, which are both natural constituents of fruit and vegetable juices.
The EU Scientific Committee on food, the FDA in the United States and the JECFA of the WHO have confirmed the safe use in beverages.
References
- ^ [1][dead link]
- ^ DMDC's role in bottle stability - dimethyl dicarbonate, Wines & Vines, Oct 1990
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". http://www.food.gov.uk/safereating/chemsafe/additivesbranch/enumberlist. Retrieved 2011-10-27.
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part I". http://www.fda.gov/Food/FoodIngredientsPackaging/FoodAdditives/FoodAdditiveListings/ucm091048.htm. Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 - Labelling of ingredients". http://www.comlaw.gov.au/Details/F2011C00827. Retrieved 2011-10-27.
External links
Categories:- Oenology
- Preservatives
- Methyl esters
- Carbonate esters
- O=C(OC(=O)OC)OC
Wikimedia Foundation. 2010.
