- Diborane
-
Diborane 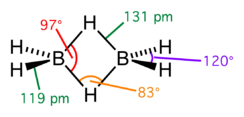
 Diborane(6)
Diborane(6)Identifiers CAS number 19287-45-7 
ChemSpider 17215804 
EC number 242-940-6 ChEBI CHEBI:33590 
RTECS number HQ9275000 Jmol-3D images Image 1 - [BH2]1[H][BH2][H]1
Properties Molecular formula H6B2 Molar mass 27.67 g mol−1 Exact mass 28.065561124 g mol-1 Appearance Colorless gas Density 1.216 g dm-3 Melting point −165 °C, 108 K, -265 °F
Boiling point -92 °C, 181 K, -134 °F
Structure Coordination
geometryTetrahedral (for boron) Molecular shape see text Dipole moment 0 D Hazards MSDS External MSDS NFPA 704 Autoignition
temperature38 °C Related compounds Related boron compounds Decaborane
BF3Supplementary data page Structure and
propertiesn, εr, etc. Thermodynamic
dataPhase behaviour
Solid, liquid, gasSpectral data UV, IR, NMR, MS  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Diborane is the chemical compound consisting of boron and hydrogen with the formula B2H6. It is a colorless gas at room temperature with a repulsively sweet odor. Diborane mixes well with air, easily forming explosive mixtures. Diborane will ignite spontaneously in moist air at room temperature. Synonyms include boroethane, boron hydride, and diboron hexahydride.
Diborane is a key boron compound with a variety of applications. The compound is classified as "endothermic", meaning that its heat of formation, ΔH°f is positive (36 kJ/mol). Despite its thermodynamic instability, diborane is kinetically robust and exhibits an extensive chemistry, much of it entailing loss of hydrogen.
Contents
Structure and bonding
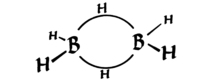 Bonding diagram of diborane (B2H6) showing with curved lines a pair of three-center two-electron bonds, each of which consists of a pair of electrons bonding three atoms, two boron atoms and a hydrogen atom in the middle.
Bonding diagram of diborane (B2H6) showing with curved lines a pair of three-center two-electron bonds, each of which consists of a pair of electrons bonding three atoms, two boron atoms and a hydrogen atom in the middle.
Diborane adopts a D2h structure containing four terminal and two bridging hydrogen atoms. The model determined by molecular orbital theory indicates that the bonds between boron and the terminal hydrogen atoms are conventional 2-center, 2-electron covalent bonds. The bonding between the boron atoms and the bridging hydrogen atoms is, however, different from that in molecules such as hydrocarbons. Having used two electrons in bonding to the terminal hydrogen atoms, each boron has one valence electron remaining for additional bonding. The bridging hydrogen atoms provide one electron each. Thus the B2H2 ring is held together by four electrons, an example of 3-center 2-electron bonding. This type of bond is sometimes called a 'banana bond'. The lengths of the B-Hbridge bonds and the B-Hterminal bonds are 1.33 and 1.19 Å respectively, and this difference in the lengths of these bonds reflects the difference in their strengths, the B-Hbridge bonds being relatively weaker. The structure is isoelectronic with C2H62+, which would arise from the diprotonation of the planar molecule ethene.[1] Diborane is one of many compounds with such unusual bonding.[2]
Of the other elements in Group 13, gallium is known to form a similar compound, digallane, Ga2H6. Aluminium forms a polymeric hydride, (AlH3)n, although unstable Al2H6 has been isolated in solid hydrogen and is isostructural with diborane.[3] No hydrides of indium and thallium have yet been found.[4]
Production and synthesis
Diborane is so central and has been studied so often that many syntheses exist. Most preparations entail reactions of hydride donors with boron halides or alkoxides. The industrial synthesis involves the reduction of BF3 by sodium hydride:
- 2 BF3 + 6 NaH → B2H6 + 6 NaF
Two laboratory methods start from boron trichloride with lithium aluminium hydride or from boron trifluoride ether solution with sodium borohydride. Both methods yield in up to 30% of diborane:
- 4 BCl3 + 3 LiAlH4 → 2 B2H6 + 3 LiAlCl4
- 4 BF3 + 3 NaBH4 → 2 B2H6 + 3 NaBF4
Older methods entail the direct reaction of borohydride salts with a non-oxidizing acid, such as phosphoric acid or dilute sulfuric acid.
- 2 BH4− + 2 H+ → 2 H2 + B2H6
Similarly, oxidation of borohydride salts has been demonstrated and remains convenient for small scale preparations. For example, using iodine as oxidizer:
- 2 NaBH4 + I2 → 2 NaI + B2H6 + H2
Reactions
Diborane is a highly reactive and versatile reagent that has a large number of applications.[5] Its dominating reaction pattern involves formation of adducts with Lewis bases. Often such initial adducts proceed rapidly to give other products. It reacts with ammonia to form ammonia borane or the diammoniate of diborane, DADB, depending on the conditions used. Diborane also reacts readily with alkynes to form substituted alkene products which will readily undergo further addition reactions.
Diborane reacts with water to form hydrogen and boric acid.
The compound forms complexes with Lewis bases. Notable are the complexes with THF and dimethyl sulfide, both liquid compounds that are popular reducing agents in organic chemistry. In these 1:1 complexes, boron assumes a tetrahedral geometry, being bound to three hydrides and the Lewis base (THF or Me2S). The THF adduct is usually prepared as a 1:5 solution in THF. The latter is indefinitely stable when stored under nitrogen at room temperature.
Reagent in organic synthesis
Diborane is the central organic synthesis reagent for hydroboration, whereby alkenes add across the B-H bonds to give trialkylboranes:
- (THF)BH3 + 3 CH2=CHR → B(CH2CH2R)3 + THF
This reaction is regioselective, and the product trialkylboranes can be converted to useful organic derivatives. With bulky alkenes one can prepare species such as [HBR2]2, which are also useful reagents in more specialized applications.
Diborane is used as a reducing agent roughly complementary to the reactivity of lithium aluminium hydride. The compound readily reduces carboxylic acids to the corresponding alcohols, whereas ketones react only sluggishly.
History
Diborane was first synthesised in the 19th century by hydrolysis of metal borides, but it was never analysed. From 1912 to 1936, the major pioneer in the chemistry of boron hydrides, Alfred Stock, undertook his research that led to the methods for the synthesis and handling of the highly reactive, volatile, and often toxic boron hydrides. He proposed the first ethane-like structure of diborane.[6] Electron diffraction measurements by S. H. Bauer initially appeared to support his proposed structure.[7][8]
Because of a personal communication with L. Pauling (who supported the ethane-like structure), H. I. Schlessinger did not specifically discuss 3-center-2-electron bonding in his then classic review in the early 1940s.[9] The review does, however, discuss the C2v structure in some depth, "It is to be recognized that this formulation easily accounts for many of the chemical properties of diborane..."
In 1943 an undergraduate student at Balliol College, Oxford, H. Christopher Longuet-Higgins, published the currently accepted structure together with R. P. Bell.[10] This structure had already been described in 1921.[11][12][13] The years following the Longuet-Higgins/Bell proposal witnessed a colorful discussion about the correct structure. The debate ended with the electron diffraction measurement in 1951 by K. Hedberg and V. Schomaker, with the confirmation of the structure shown in the schemes on this page.[14]
William Nunn Lipscomb, Jr. further confirmed the molecular structure of boranes using X-ray crystallography in the 1950s, and developed theories to explain its bonding. Later, he applied the same methods to related problems, including the structure of carboranes on which he directed the research of future Nobel Prize winner Roald Hoffmann. Lipscomb himself received the Nobel Prize in Chemistry in 1976 for his efforts.
Other uses
Diborane is used in rocket propellants, as a rubber vulcaniser, as a catalyst for hydrocarbon polymerisation, as a flame-speed accelerator, and as a doping agent for the production of semiconductors. It is also an intermediate in the production of highly pure boron for semiconductor production. It is also used to coat the walls of tokamaks to reduce the amount of heavy metal impurities in the plasma.
Safety
The toxic effects of diborane are primarily due to its irritant properties. Short-term exposure to diborane can cause a sensation of tightness of the chest, shortness of breath, cough, and wheezing. These signs and symptoms can occur immediately or be delayed for up to 24 hours. Skin and eye irritation can also occur. Studies in animals have shown that diborane causes the same type of effects observed in humans.[citation needed]
People exposed for a long time to low amounts of diborane have experienced respiratory irritation, seizures, fatigue, drowsiness, confusion, and occasional transient tremors.
References
- ^ G. Rasul, G. K. S. Prakash, G. A. Olah (2005). "Comparative ab Initio Study of the Structures and Stabilities of the Ethane Dication C2H62+ and Its Silicon Analogues Si2H62+ and CSiH62+". J. Phys. Chem. A 109 (5): 798–801. doi:10.1021/jp0404652. PMID 16838949.
- ^ Laslo P (2000). "A Diborane Story". Angewandte Chemie International Edition 39 (12): 2071–2072. doi:10.1002/1521-3773(20000616)39:12<2071::AID-ANIE2071>3.0.CO;2-C. PMID 10941018. abstract
- ^ Andrews, Lester; Wang, Xuefeng (2003). "The Infrared Spectrum of Al2H6 in Solid Hydrogen". Science 299 (5615): 2049–2052. doi:10.1126/science.1082456. PMID 12663923.
- ^ Downs, Anthony J.; Colin R. Pulham (1994). "The hydrides of aluminium, gallium, indium and thallium: A re-evaluation". Chemical Society Reviews (Cambridge: Royal Society of Chemistry) 23 (3): 175–184. doi:10.1039/cs9942300175.
- ^ Mikhailov BM (1962). "The Chemistry of Diborane". Russian Chemical Review 31 (31): 207–224. doi:10.1070/RC1962v031n04ABEH001281.
- ^ Stock A. (1933). The Hydrides of Boron and Silicon. New York: Cornell University Press.
- ^ Bauer S.H. (1937). "The Structure of Diborane". Journal of the American Chemical Society 59 (6): 1096. doi:10.1021/ja01285a041.
- ^ Bauer S.H. (1942). "Structures and Physical Properties of the Hydrides of Boron and of their Derivatives". Chemical Reviews 31: 43–75. doi:10.1021/cr60098a002.
- ^ Schlesinger H.I., Burg A.B. (1942). "Recent Developments in the Chemistry of the Boron Hydrides". Chemical Reviews 31: 1–41. doi:10.1039/JR9430000250.
- ^ Longuet-Higgins, H. C.; Bell, R. P. (1943). "64. The structure of the boron hydrides". Journal of the Chemical Society (Resumed): 250. doi:10.1039/JR9430000250.
- ^ Dilthey W. (1921). "Über die Konstitution des Wassers". Zeitschriffte fuer Angewandte Chemie 34 (95): 596. doi:10.1002/ange.19210349509.
- ^ Nekrassov BV (1940). J Gen Chem USSR 10: 1021.
- ^ Nekrassov BV (1940). J Gen Chem USSR 10: 1056.
- ^ Hedberg K, Schomaker V (1951). "A Reinvestigation of the Structures of Diborane and Ethane by Electron Diffraction". Journal of the American Chemical Society 73 (4): 1482–1487. doi:10.1021/ja01148a022.
Further reading
H. C. Brown "Organic Synthesis via Boranes" John Wiley, New York, 1975. ISBN 0-471-11280-1.
External links
- International Chemical Safety Card 0432
- Computational Chemistry Wiki
- National Pollutant Inventory - Boron and compounds
- NIOSH Pocket Guide to Chemical Hazards
- U.S. EPA Acute Exposure Guideline Levels
Boron compounds Categories:- Boranes
- Rocket fuels
- Reducing agents
Wikimedia Foundation. 2010.

