- Dichlorophenolindophenol
-
DCPIP 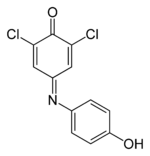
 2,6-dichlorophenol-indophenolOther names2,5-cyclohexadien-1-one;
2,6-dichlorophenol-indophenolOther names2,5-cyclohexadien-1-one;
dichloroindophenol;
2,6-Dichloro-4-((4-hydroxyphenyl)imino) -2,5-cyclohexadien-1-one
2,6-dichloro-4-[(4-hydroxyphenyl)imino] -2,6-dichloroindophenol;Identifiers CAS number 956-48-9 
ChemSpider 10661857 
KEGG C00102 
ChEBI CHEBI:945 
ChEMBL CHEMBL500871 
Jmol-3D images Image 1 - Cl\C2=CC(=N/c1ccc(O)cc1)/C=C(/Cl)C2=O
Properties Molecular formula C12H7NCl2O2 Molar mass 268.1 g mol−1  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
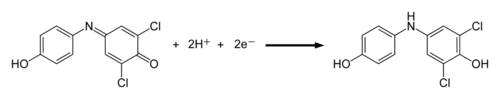
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references 2,6-dichlorophenolindophenol, ( DCPIP ) is a blue chemical compound used as a redox dye. Oxidized DCPIP is blue, reduced DCPIP is colorless.
The rate of photosynthesis can be measured by the rate at which this dye changes color (is reduced) when exposed to light in a photosynthetic system. This reaction is reversible, as colorless DCPIP can be reoxidized to blue. It is often used in measurements of the electron transport chain in plants because of its higher affinity for electrons than ferredoxin. For example, it is possible to compare rates of photosynthesis with DCPIP. Due to the reductions that occur during the light reactions, DCPIP can be substituted for NADPH, the final electron carrier in the light reactions. The light reactions will reduce the DCPIP and turn it colorless. As DCPIP is reduced and becomes colorless, the following increase in light transmittance can be measured by a spectrophotometer.
DCPIP
DCPIP is a redox dye commonly used as a monitor of the light reactions in photosynthesis because it is an electron acceptor that is blue when oxidized and colorless when reduced. It is part of the Hill reagents family. DCPIP is commonly used as a substitute for NADP+. The dye changes color when it is reduced, due to its chemical structure. The rate of photosynthesis light-dependent reaction can be measured with this property of DCPIP, because one of the stages of the light reaction is an electron transport chain that normally ends with the reduction of NADP+. When DCPIP is present, it also gets reduced by the light reaction. The amount of DCPIP reduced can be found by measuring the solution's light transmittance with a spectrophotometer.
DCPIP can also be used as an indicator for Vitamin C.[1] If vitamin C, which is a good reducing agent, is present, the blue dye, which turns pink in acid conditions, is reduced to a colorless compound by ascorbic acid.
- DCPIP (blue) + H+ ——→ DCPIPH (pink)
- DCPIPH (pink) + VitC ——→ DCPIPH2 (colorless)
- C6H8O6 + C12H7NCl2O2 ——→ C6H6O6 + C12H9NCl2O2
In a titration, when all the ascorbic acid in the solution has been used up, there will not be any electrons available to reduce the DCPIPH and the solution will remain pink due to the DCPIPH. The end point is a pink color that persists for 10 seconds or more. Pharmacological experiments suggest that DCPIP may serve as a pro-oxidant chemotherapeutic targeting human cancer cells in an animal model of human melanoma; DCPIP-induced cancer cell death occurs by depletion of intracellular glutathione and upregulation of oxidative stress.[2]
See also
References
- ^ VanderJagt DJ, Garry PJ, Hunt WC (June 1986). "Ascorbate in plasma as measured by liquid chromatography and by dichlorophenolindophenol colorimetry". Clin. Chem. 32 (6): 1004–6. PMID 3708799. http://www.clinchem.org/cgi/pmidlookup?view=long&pmid=3708799.
- ^ Cabello CM, Bair WB, Bause AS, Wondrak GT (August 2009). "Antimelanoma activity of the redox dye DCPIP (2,6-dichlorophenolindophenol) is antagonized by NQO1". Biochem. Pharmacol. 78 (4): 344–54. doi:10.1016/j.bcp.2009.04.016. PMID 19394313. http://linkinghub.elsevier.com/retrieve/pii/S0006-2952(09)00301-3.
- Denby, Derek (May 1996). Chemistry Review.
Categories:- Indophenol dyes
- Redox indicators
Wikimedia Foundation. 2010.