- Atmosphere of Mercury
-
Atmosphere of Mercury[1]Species CD,[n 1] cm−2 SD,[n 2] cm−3 Hydrogen (H) ~ 3 × 109 ~ 250 Molecular hydrogen < 3 × 1015 < 1.4 × 107 Helium < 3 × 1011 ~ 6 × 103 Atomic oxygen < 3 × 1011 ~ 4 × 104 Molecular oxygen < 9 × 1014 < 2.5 × 107 Sodium ~ 2 × 1011 1.7–3.8 × 104 Potassium ~ 2 × 109 ~ 400 Calcium ~ 1.1 × 108 ~ 300 Magnesium ~ 4 × 1010 ~ 7.5 × 103 Argon ~ 1.3 × 109 < 6.6 × 106 Water < 1 × 1012 < 1.5 × 107 Other neon, silicon, sulfur, argon,
iron, carbon dioxide, etc.Mercury has a very tenuous and highly variable atmosphere (surface-bound exosphere) containing hydrogen, helium, oxygen, sodium, calcium, potassium and water vapor, with a combined pressure level of about 10−14 bar (1 nPa).[2] The exospheric species originate either from the Solar wind or from the planetary crust. Solar light pushes the atmospheric gases away from the Sun, creating a comet-like tail behind the planet.
The existence of a Hermian atmosphere had been contentious before 1974, although by that time a consensus had formed that Mercury, like the Moon, lacked any substantial atmosphere. This conclusion was confirmed in 1974 when the Mariner 10 spacecraft discovered only a tenuous exosphere. Later, in 2008, improved measurements were obtained by the MESSENGER spacecraft, which discovered magnesium in the Hermian exosphere.
Contents
History
Mercury is the most difficult planet to observe due to its proximity to the Sun. Though its phases were discovered by Zupus in 1639, the first map of its surface was not drawn until 1882. Ground-based observations led to a mistaken perception that Mercury rotates synchronously with its orbital motion, which would leave one half of the planet permanently dark.[3]
The viability of an atmosphere on Mercury had been debated before Mariner 10's 1974 flyby.[4] On the one hand proximity to the Sun would induce a high temperature in any putative atmosphere, leading to its rapid escape.[5] On the other hand the dark hemisphere would be very cold, causing many gases to freeze. To prevent the freeze, the atmosphere would either need to be relatively thick (about 5 mbar for carbon dioxide) in order to transport heat from the illuminated hemisphere, or would need to be composed of gases such as argon with low condensation temperatures.[6]
By the 1960s some evidence had accumulated indicating that Mercury might have a thin atmosphere. Polarimetric measurements by Bernard Lyot and Audouin Dollfus implied a surface pressure of 0.1–10 mbar,[7] and some spectroscopic results suggested a carbon dioxide atmosphere with a few mbars of surface pressure.[7] Additionally microwave observations showed that the temperature of Mercury's night side was higher than what had been considered possible.[6]
However all these pieces of evidence were later called into doubt. The discovery that Mercury's rotation is not synchronous explained elevated night temperatures,[6] while improved spectroscopic measurements sharply lowered the upper limit on the carbon dioxide presence to less than 0.0001 mbar.[8] The accuracy of the polarimetry was questioned as well.[7] By 1974 a consensus had formed that Mercury, like the Moon, had virtually no atmosphere. The Mariner 10 flybys confirmed that conclusion, discovering only a tenuous surface-bound exosphere (i.e. a thin shell of gases whose base coincides with the planetary surface).[4]
Composition
The Hermian exosphere consists of a variety of species originating either from the Solar wind or from the planetary crust.[9] The first constituents discovered were atomic hydrogen (H), helium (He) and atomic oxygen (O), which were observed by the ultraviolet photometer of the Mariner 10 spacecraft in 1974. The near-surface concentrations of these elements were estimated to vary from 230 cm−3 for hydrogen to 44,000 cm−3 for oxygen, with an intermediate concentration of helium.[9] In 2008 the MESSENGER probe confirmed the presence of atomic hydrogen, although its concentration appeared higher than the 1974 estimate.[10] Mercury's exospheric hydrogen and helium are believed to come from the Solar wind, while the oxygen is likely to be of crustal origin.[9]
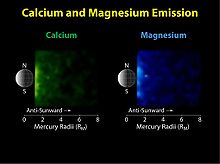
The fourth species detected in Mercury's exosphere was sodium (Na). It was discovered in 1985 by Drew Potter and Tom Morgan, who observed its Fraunhofer emission lines at 589 and 589.6 nm.[11] The average column density of this element is about 1 × 1011 cm−2. Sodium is observed to concentrate near the poles, forming bright spots.[12] Its abundance is also enhanced near the dawn terminator as compared to the dusk terminator.[13] Some research has claimed a correlation of the sodium abundance with certain surface features such as Caloris or radio bright spots;[11] however these results remain controversial. A year after the sodium discovery, Potter and Morgan reported that potassium (K) is also present in the exosphere of Mercury, though with a column density two orders of magnitude lower than that of sodium. The properties and spatial distribution of these two elements are otherwise very similar.[14] In 1998 another element, calcium (Ca), was detected with column density three orders of magnitude below that of sodium.[15] Observations by the MESSENGER probe in 2009 showed that calcium is concentrated mainly near the equator—opposite to what is observed for sodium and potassium.[16]
In 2008 the MESSENGER probe's Fast Imaging Plasma Spectrometer (FIPS) discovered several molecular and different ions in the vicinity of Mercury, including H2O+ (ionized water vapor) and H2S+ (ionized hydrogen sulfide).[17] Their abundances relative to sodium are about 0.2 and 0.7, respectively. Other ions such as H3O+ (hydroxonium), OH (hydroxyl), O2+ and Si+ are present as well.[18] During its 2009 flyby, the Ultraviolet and Visible Spectrometer (UVVS) channel of the Mercury Atmospheric and Surface Composition Spectrometer (MASCS) on board the MESSENGER spacecraft first revealed the presence of magnesium in the Hermian exosphere. The near-surface abundance of this newly detected constituent is roughly comparable to that of sodium.[16]
Properties
Mariner 10's ultraviolet observations have established an upper bound on the exospheric surface density at about 105 particles per cubic centimeter. This corresponds to a surface pressure of less than 10−14 bar (1 nPa).[19]
The temperature of the Hermian exosphere depends on species as well as geographical location. For exospheric atomic hydrogen, the temperature appears to be about 420 K, a value obtained by both Mariner 10 and MESSENGER.[10] The temperature for sodium is much higher, reaching 750–1500 K on the equator and 1,500–3,500 K at the poles.[20] Some observations show that Mercury is surrounded by a hot corona of calcium atoms with temperature between 12,000 and 20,000 K.[15]
Tail
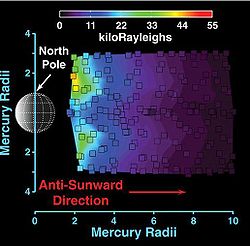
Because of Mercury's proximity to the Sun, the pressure of Solar light is much stronger than at Earth's location. Solar radiation pushes neutral atoms away from Mercury, creating a comet-like tail behind the planet.[21] The main component in the tail is sodium, which has been detected as far as 56,000 km (about 23 RM) from the planet.[21] This sodium tail expands rapidly to a diameter of about 20,000 km at a distance of 17,500 km.[22] In 2009 MESSENGER also detected calcium and magnesium in the tail, although these elements were only observed at distances less than 8 RM from the planet.[21]
See also
- Orders of magnitude (pressure)
- Magnetosphere of Mercury
Notes
- ^ Killen 2007, p. 456, Table 5
- ^ "NASA—Mercury". http://www.nasa.gov/worldbook/mercury_worldbook.html. Retrieved 2009-09-26.
- ^ Rasool 1966, p. 566
- ^ a b Williams, 1974
- ^ Rasool 1966, pp. 575–578
- ^ a b c Rasool 1966, pp. 572–575
- ^ a b c Rasool 1966, pp. 569–571
- ^ Fink 1974
- ^ a b c Killen, 2007, pp. 433–434
- ^ a b McClintock 2008, p. 93
- ^ a b Killen, 2007, pp. 434–436
- ^ Killen, 2007, pp. 438–442
- ^ Killen, 2007, pp. 442–444
- ^ Killen, 2007, pp. 449–452
- ^ a b Killen, 2007, pp. 452–453
- ^ a b McClintock 2009, p. 612–613
- ^ "MESSENGER Scientists 'Astonished' to Find Water in Mercury's Thin Atmosphere". The Planetary Society. 2008-07-03. http://www.planetary.org/news/2008/0703_MESSENGER_Scientists_Astonished_to.html. Retrieved 2010-03-28.
- ^ Zurbuchen 2008, p. 91, Table 1
- ^ Domingue, 2007, pp. 162–163
- ^ Killen, 2007, pp. 436–438
- ^ a b c McClintock 2009, p. 610–611
- ^ Killen, 2007, p. 448
References
- Domingue, Deborah L.; Koehn, Patrick L.; Killen, Rosemary M. et al. (2007). "Mercury's Atmosphere: A Surface-Bounded Exosphere". Space Science Reviews 131 (1-4): 161–186. Bibcode 2007SSRv..131..161D. doi:10.1007/s11214-007-9260-9.
- Fink, Uwe; Larson, Harold P.; Poppen, Richard F. (1974). "A new upper limit for an atmosphere of CO2, CO on Mercury". The Astrophysical Journal 187: 407–415. Bibcode 1967ApJ...149L.137B. doi:10.1086/180075.
- Killen, Rosemary; Cremonese, Gabrielle; Lammer, Helmut et al. (2007). "Processes that Promote and Deplete the Exosphere of Mercury". Space Science Reviews 132 (2-4): 433–509. Bibcode 2007SSRv..132..433K. doi:10.1007/s11214-007-9232-0.
- McClintock, William E.; Bradley, E. Todd; Vervack Jr., Ronald J. et al. (2008). "Mercury’s Exosphere: Observations During MESSENGER’s First Mercury Flyby". Science 321 (5885): 92–94. Bibcode 2008Sci...321...62M. doi:10.1126/science.1159467. PMID 18599778.
- McClintock, William E.; Vervack Jr., Ronald J.; Bradley, E. Todd et al. (2009). "MESSENGER Observations of Mercury’s Exosphere: Detection of Magnesium and Distribution of Constituents". Science 324 (5927): 610–613. Bibcode 2009Sci...324..610M. doi:10.1126/science.1172525. PMID 19407195.
- Rasool, S.I.; Gross, S.H.; McGovern, W.E. (1966). "The atmosphere of Mercury". Space Science Reviews 5 (5): 565–584. Bibcode 1966SSRv....5..565R. doi:10.1007/BF00167326.
- Williams, I.P. (1974). "Atmosphere of Mercury". Nature 249 (5454): 234. Bibcode 1974Natur.249..234W. doi:10.1038/249234a0.
- Zurbuchen, Thomas H.; Raines, Jim M.; Gloeckler, George et al. (2008). "MESSENGER Observations of the Composition of Mercury’s Ionized Exosphere and Plasma Environment". Science 321 (5885): 90–92. Bibcode 2008Sci...321...90Z. doi:10.1126/science.1159314. PMID 18599777.
Mercury Observation 
Transits Exploration Properties - Atmosphere
- Magnetic field
Geology - Beagle Rupes
- Caloris Basin
- Discovery Rupes
- Rembrandt Basin
- Resolution Rupes
Other topics Atmospheres Major 
Tenuous See also Categories:- Planetary atmospheres
- Solar system planetary atmospheres
- Mercury (planet)
Wikimedia Foundation. 2010.