- Ouabain
-
Ouabain 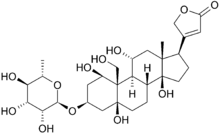
Systematic (IUPAC) name 1β,3β,5β,11α,14,19-Hexahydroxycard-20(22)-enolide 3-(6-deoxy-α-L-mannopyranoside)
OR
4-[(1R,3S,5S,8R,9S,10R,11R,13R,14S,17R)-1,5,11,14-tetrahydroxy-10-
(hydroxymethyl)-13-methyl-3-((2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-
pyran-2-yloxy)hexadecahydro-1H-cyclopenta[a]phenanthren-17-yl]furan-2(5H)-oneClinical data Trade names Strodival AHFS/Drugs.com International Drug Names Pregnancy cat. ? Legal status ? Identifiers CAS number 630-60-4 
ATC code C01AC01 PubChem CID 439501 DrugBank DB01092 ChemSpider 388599 
UNII 5ACL011P69 
KEGG D00112 
ChEBI CHEBI:472805 
ChEMBL CHEMBL447086 
Chemical data Formula C29H44O12 Mol. mass 584.652 SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Ouabain (
 /ˈwɑːbɑːɪn/,[1] /wɑːˈbɑːɪn/ or /ˈwɑːbeɪn/; from Somali waabaayo, "arrow poison" through French ouabaïo) which is also named g-strophanthin, is a poisonous cardiac glycoside.
/ˈwɑːbɑːɪn/,[1] /wɑːˈbɑːɪn/ or /ˈwɑːbeɪn/; from Somali waabaayo, "arrow poison" through French ouabaïo) which is also named g-strophanthin, is a poisonous cardiac glycoside.Contents
Sources
Ouabain (g-strophanthin) is found in the ripe seeds of African plants Strophanthus gratus and the bark of Acokanthera ouabaio.
Function
It is well understood that the classical mechanism of action of ouabain involves its binding to and inhibition of the plasma membrane Na+/K+-ATPase (sodium pump) especially at the higher concentrations attainable in vitro or with intravenous dosage. Inhibition of the sodium pump and the secondary effect on the handling of calcium ions by sodium calcium exchanger (NCX) is widely believed to underlie the original beneficial effect as an inotropic agent following intravenous use of ouabain; digoxin is a structurally related and more lipophilic cardiac glycoside that largely replaced ouabain for therapy because of its superior bioavailability per os. Digoxin continues to be used therapeutically for many of the same indications in which ouabain was used (including atrial fibrillation and congestive heart failure).
In addition to the classical mechanism of action of ouabain at high concentrations, low (nanomolar and subnanomolar) concentrations of ouabain, as they were found endogenously (see below) or after oral intake, stimulate the Na-K-ATPase. e.g.,[2] This stimulatory effect does not appear to be mimicked by digoxin.[3] Furthermore ouabain acts as a signal transducer. [4]
Endogenous ouabain and ouabain mimics
In 1991, a potent sodium pump inhibitor was isolated from the human circulation and identified as ouabain. Several additional observations led to the view that the circulating ouabain in humans was an endogenous hormone.[5] A reinterpretation of a portion of the analytical data led to the proposal that endogenous ouabain may have been the 11 epimer, i.e., an isomer of plant ouabain.[6] However, this possibility has been excluded by the synthesis of the 11 epimer and the demonstration that it has different chromatographic behavior from ouabain. There is compelling analytical evidence that ouabain is synthesized in the adrenal gland.[7] A ouabain-like factor may also be made in the hypothalamus[8] and possibly the heart where it may be stimulated by oxygen deficiency.[9] In the latter instances, analytical data are needed to support the identity of the secreted materials. Further, secretion is not equivalent to biosynthesis; most all tissues sequester ouabain from the circulation. When such tissues are removed from the circulation they secrete ouabain but this phenomenon, by itself, is not proof of biosynthesis.
The mode of action and significance of physiological levels of endogenous ouabain are under active investigation. In human plasma from healthy individuals, the circulating levels are normally distributed in the population and range typically from 30 - 380 pM. Significantly higher levels of endogenous ouabain that may approach or even exceed 1 nM have been observed in many patients with congestive heart failure, essential hypertension, renal failure and some cancers. It has been suggested that physiological concentrations of ouabain promote cell growth and in some manner stimulate the Na+/K+-ATPase activity while the higher levels achieved during intravenous therapy or in pathophysiological disorders may inhibit the Na+/K+-ATPase.[1]
In the years following the discovery of ouabain in the human circulation, it was found in the adrenal glands of cows[10] and dogs.[11]
Uses
Ouabain isolated from plants is widely used by scientists in in vitro studies to specifically block the sodium pump (Na-K-ATPase). In many non rodent species, low concentrations of this substance (i.e., in the subnanomolar range) may stimulate the Na-K-ATPase. The mechanism of the stimulatory effect is not understood and remains controversial. Further, the issue in rodents is more complicated because there are different isoforms of the Na-K-ATPase - some of which are very sensitive to ouabain while others are not.
The parenteral absorption seems to be better than indicated by the textbooks.[12]
In France and Germany, intravenous ouabain has a long history in the treatment of heart failure, and some continue to advocate its use intravenously and per os (orally) in angina pectoris and myocardial infarction. The positive properties of ouabain regarding the prophylaxis and treatment of these two indications are documented by a clutch of studies.[12] One of them is available in full text modus at pubmed.[13]
Extracts containing ouabain have long been used by Somali tribesmen and other groups to poison hunting arrows. A sufficiently concentrated ouabain dart can bring down a Hippopotamus probably as the result of respiratory and/or cardiac arrest. The Galápagos tortoise, however, is immune to the effects of ouabain darts.[citation needed]
See also
References
- ^ "ouabain" in the World English Dictionary
- ^ Gao, J. et al. (2002): Isoform-specific stimulation of cardiac Na/K pumps by nanomolar concentrations of glycosides. J Gen Physiol 119:297-312. PMID 11929882 PDF
- ^ Balzan S. et al (2007): Erythrocyte sodium pump stimulation by ouabain and an endogenous ouabain-like factor. Cell Biochem Funct. 25(3):297-303.PMID 17191274
- ^ Aperia A. (2007): New roles for an old enzyme: Na,K-ATPase emerges as an interesting drug target. Review. J Intern Med. 261(1):44-52. PMID 17222167
- ^ Hamlyn JM et al.: Identification and characterization of an ouabain-like compound from human plasma. Proceedings of the National Academy of Sciences of the United States of America 88 (14): 6259-63, 1991. PMID 1648735
- ^ Hamlyn JM et al.: 11-hydroxylation in the biosynthesis of endogenous ouabain: multiple implications. Ann N Y Acad Sci 986: 685-93, 2003 PMID 12763919
- ^ Laredo J et al.: Ouabain is secreted by bovine adrenocortical cells. Endocrinology 135: 794-7, 1994 PMID 8033829
- ^ Murrell JR: Endogenous ouabain: upregulation of steroidogenic genes in hypertensive hypothalamus but not adrenal. Circulation 112(9): 1301-8, 2005 PMID 16116051
- ^ D´Urso et al. 2004: Production of ouabain-like factor in normal and ischemic rat heart. J Cardiovasc Pharmacol 43(5):657-62, 2004. PMID 15071352
- ^ Schneider, Ralf; Victor Wray, Manfred Nimtz, Wolf Dieter Lehmannpar , Ulrike Kirch, Roberto Antolovic, and Wilhelm Schoner (January 9, 1998). "Bovine Adrenals Contain, in Addition to Ouabain, a Second Inhibitor of the Sodium Pump". The Journal of Biological Chemistry 273 (2): 784–92. doi:10.1074/jbc.273.2.784. PMID 9422732. http://www.jbc.org/cgi/content/full/273/2/784.
- ^ Boulanger, B. R.; M. P. Lilly, J. M. Hamlyn, J. Laredo, D. Shurtleff and D. S. Gann (March 1993). "Ouabain is secreted by the adrenal gland in awake dogs". The American Journal of Physiology: Endocrinology and Metabolism 264 (3): E413–E419. PMID 8460688. http://ajpendo.physiology.org/cgi/reprint/264/3/E413.
- ^ a b PMID 20946265, see http://onlinelibrary.wiley.com/doi/10.1111/j.1742-1241.2010.02395.x/abstract
- ^ Sharma B et al.: Clinical, electrocardiographic, and haemodynamic effects of digitalis (ouabain) in angina pectoris. Br Heart J. 34: 631-7, 1972. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pubmed&pubmedid=4402698
External links
- Ouabainomics by Hamlyn
- "THE USE OF CIRCULATORY STIMULANTS IN THE CARE OF THE SICK" from Canadian Medical Association Journal (1922) refers to use of strophanthin in heart failure
Cardiac glycosides (C01A) Bufadienolides Proscillaridin • ScillirosideCardenolides Ouabain (g-Strophanthin) • k-Strophanthin • CymarinCategories:- Cardenolides
- Alcohols
Wikimedia Foundation. 2010.

