- Cyanine
-
Cyanine is a non-systematic name of a synthetic dye family belonging to polymethine group. Cyanines have many uses as fluorescent dyes, particularly in biomedical imaging. Depending on the structure, they cover the spectrum from IR to UV.
Cyanines were originally used, and still are, to increase the sensitivity range of photographic emulsions, i.e., to increase the range of wavelengths which will form an image on the film. Cyanines are also used in CD-R and DVD-R media. The ones used are mostly green or light blue in color, and are chemically unstable. This makes unstabilized cyanine discs unsuitable for archival CD and DVD use, as they can fade and become unreadable in a few years, however, recent cyanine discs contain stabilizers that slow down the deterioration significantly. These discs are often rated with an archival life of 75 years or more. The other dyes used in CD-Rs are phthalocyanine and azo.
Cyanines were first synthesized over a century ago, and there are a large number reported in the literature.
Contents
Structure
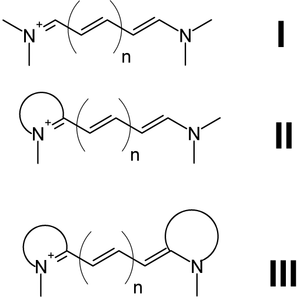
There are three types of cyanines:
- Streptocyanines or open chain cyanines:
- R2N+=CH[CH=CH]n-NR2 (I)
- Hemicyanines:
- Aryl=N+=CH[CH=CH]n-NR2 (II)
- Closed chain cyanines:[1]
- Aryl=N+=CH[CH=CH]n-N=Aryl (III)
where two nitrogens are joined by a polymethine chain.[2] Both nitrogens are each independently part of a heteroaromatic moiety, such as pyrrole, imidazole, thiazole, pyridine, quinoline, indole, benzothiazole, etc.
Cy3 and Cy5
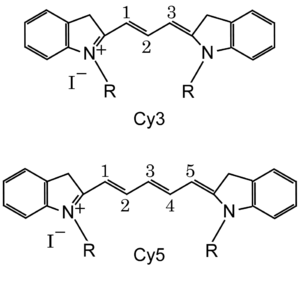
Cy3 and Cy5 are reactive water-soluble fluorescent dyes of the cyanine dye family. Cy3 dyes are red (~550 nm excitation, ~570 nm emission and therefore appear red), while Cy5 is fluorescent in the far red region (~650/670 nm) but absorbs in the orange region (~649 nm).[3] They are usually synthesized with reactive groups on either one or both of the nitrogen side chains so that they can be chemically linked to either nucleic acids or protein molecules. Labeling is done for visualization and quantification purposes. They are used in a wide variety of biological applications including comparative genomic hybridization and in gene chips, which are used in transcriptomics. They are also used to label proteins and nucleic acid for various studies including proteomics and RNA localization.[4]
Nomenclature and Structure
Standard chemical names specify exactly the chemical structure of the molecule. The Cy3 and Cy5 nomenclature was first proposed by Ernst, et al.[2] in 1989, and is non-standard, since it gives no hint of their chemical structures. In the original paper the number designated the count of the methines (as shown), and the side chains were unspecified. Thus various structures are designated Cy3 and Cy5 in the literature.
The R groups do not have to be identical. In the dyes as used they are short aliphatic chains one or both of which ends in a highly reactive moieties such as N-hydroxysuccinimide or maleimide.
Spectral characteristics
Dye Absorbance Max Emission Max Quantum yield in PBS buffer Molecular weight (Da) Cy3 550 nm 570 nm 0.04[5] 766 Cy5 649 nm 670 nm 0.28 792
The scanners actually use different laser emission wavelengths (typically 532 nm and 635 nm) and filter wavelengths (550-600 nm and 655-695 nm) to avoid background contamination. They are thus able to easily distinguish between two samples when one sample has been labeled with Cy3 and the other labeled with Cy5. They are also able to quantify the amount of labeling in either sample.Cy dye alternatives
Alexa Fluor dyes, Atto Dyes, Dylight, IRIS Dyes, Seta dyes, SeTau dyes, SRfluor dyes and Square dyes dyes can be used interchangeably with Cy dyes in most biochemical applications.
Cy5 ozone susceptibility
In 2003, researchers at Inpharmatics and Agilent reported in Analytical Chemistry that microarrays which used Cy5 were susceptible to intermittent data quality decrease caused by environmental ozone. Exposures to ozone levels above 5-10 ppb for 10–30 seconds were reported to decrease the reproducibility of Cy5 microarrays. Much higher levels of ozone (>100 ppb) were required to observe an effect in Cy3.[6]
Applications
Nucleic acid labeling
In microarray experiments DNA or RNA is labeled with either Cy3 or Cy5 that has been synthesized to carry an N-hydroxysuccinimidyl ester (NHS-ester) reactive group. Since, NHS-esters react readily only with aliphatic amine groups, which nucleic acids lack, nucleotides have to be modified with aminoallyl groups. This is done through incorporating aminoallyl-modified nucleotides during synthesis reactions. A good ratio is a label every 60 bases such that the labels are not too close to each other, thus resulting in quenching effects.
Protein labeling
For protein labeling, Cy3 and Cy5 dyes sometimes bear maleimide reactive groups instead. The maleimide functionality allows conjugation of the fluorescent dye to the sulfhydryl group of cysteine residues. Cysteines can be added and removed from the protein domain of interest via PCR mutagenesis.
Cy5, is sensitive to the electronic environment it resides in. Changes in the conformation of the protein it is attached to will produce an enhancement or quenching of the emission. The rate of this change can be measured to determine enzyme kinetic parameters. The dyes can be used for similar purposes in FRET experiments.
Cy3 and Cy5 are used in proteomics experiments so that samples from two sources can be mixed and run together thorough the separation process.[7] This eliminates variations due to differing experimental conditions that are inevitable if the samples were run separately. These variations make it extremely difficult, if not impossible, to use computers to automate the acquisition of the data after the separation is complete. Using these dyes makes the automation trivial.
Spectrophotometric quantification of dye incorporation
The amount of each sample as well as the amount of incorporated dyes has to be quantified following nucleic acid or protein labeling and purification of the labeling reaction. To determine the dye incorporation rate, the absorbance reading at the wavelength reported for maximum absorbance of the fluorescence dye is used. The corresponding extinction coefficient of the dye is used in the Lambert-Beer Law to determine the dye concentration (c = A / (e * d)). Comparing these values with the DNA, RNA or protein concentration gives a dye incorporation rate (Frequency of incorporation (FOI)).
FOI = C(dye) / C(nucleic acid)
Example: Frequency of Incorporation (FOI) of Cy3 per 1000 bases: FOI(Cy3) = 58.5 * A550/A260
In multiple biological applications only minimal sample amounts can be afforded for these measurements (e.g. array CGH). Therefore, specialized photometers like the NanoPhotometer™[8] offer the possibility to determine sample concentrations as well as dye incorporation with submicroliter volumes (starting with 0.3 µl). In addition, due to the reduction of the optical pathlength samples are diluted automatically in comparison to standard cuvette measurements. The respective virtual dilution factors are considered by the software of the instrument. Because the measurements are processed with undiluted samples, the reproducibility of the results is very high. And if desired, samples can be retrieved after the measurement for further processing.
See also
- Merocyanine
- N-Hydroxysuccinimide
- Maleimide
- Indocyanine green
- J-aggregate
References
- ^ Johannes, H.H.: Cyanine: Direkte Funktionalisierung, Oligomerisierung, linear und nichtlinear optische Eigenschaften, Dissertation TU Braunschweig, 2000
- ^ a b Ernst LA, Gupta RK, Mujumdar RB, Waggoner AS (January 1989). "Cyanine dye labeling reagents for sulfhydryl groups". Cytometry 10 (1): 3–10. doi:10.1002/cyto.990100103. PMID 2917472.
- ^ Jackson ImmunoResearch. "Cyanine Dyes (Cy2, Cy3, and Cy5)". http://www.jacksonimmuno.com/technical/f-cy3-5.asp. Retrieved 2008-10-31.
- ^ Blower MD, Feric E, Weis K, Heald R (December 2007). "Genome-wide analysis demonstrates conserved localization of messenger RNAs to mitotic microtubules". The Journal of cell biology 179 (7): 1365–73. doi:10.1083/jcb.200705163. PMC 2373496. PMID 18166649. http://www.jcb.org/cgi/pmidlookup?view=long&pmid=18166649.
- ^ R.B. Mujumdar, L.A. Ernst, S.R. Mujumdar, C.J. Lewis, A.S. Waggoner, Cyanine dye labeling reagents: sulfoindocyanine succinimidyl esters. Bioconj Chem 4, 105-111, 1993.
- ^ Fare TL, Coffey EM, Hongyue D, et al. Effects of Atmospheric Ozone on Microarray Data Quality. Analytical Chemistry. 2003;75:4672-4675. [1]
- ^ Unlü M, Morgan ME, Minden JS (October 1997). "Difference gel electrophoresis: a single gel method for detecting changes in protein extracts". Electrophoresis 18 (11): 2071–7. doi:10.1002/elps.1150181133. PMID 9420172.
- ^ Kartha, R. Spectrophotometric Quantification of Nano- and Standard-Volume Samples, (2008, October 7), American Biotechnology Laboratory, http://www.iscpubs.com/Media/PublishingTitles/b0608kar.pdf
Categories:- Cyanine dyes
- Quaternary ammonium compounds
Wikimedia Foundation. 2010.