- Saline (medicine)
-
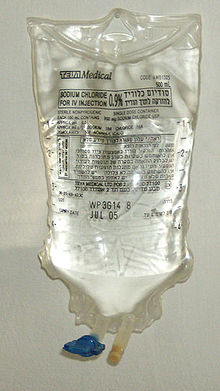 Saline solution for intravenous infusion. The white port at the base of the bag is where additives can be injected with a hypodermic needle. The port with the blue cover is where the bag is spiked with an infusion set.
Saline solution for intravenous infusion. The white port at the base of the bag is where additives can be injected with a hypodermic needle. The port with the blue cover is where the bag is spiked with an infusion set. Saline solution for irrigation. This solution is used for irrigating wounds, tissues, body cavities, and bladders. Saline solution for irrigation should not be administered intravenously. However, eye rinse drops are shared for free [?] to intravenous drug abuser in needle exhchange-programs because they contain normal saline.
Saline solution for irrigation. This solution is used for irrigating wounds, tissues, body cavities, and bladders. Saline solution for irrigation should not be administered intravenously. However, eye rinse drops are shared for free [?] to intravenous drug abuser in needle exhchange-programs because they contain normal saline.
In medicine, saline (also saline solution) is a general term referring to a sterile solution of sodium chloride (NaCl, more commonly known as salt) in water but is only sterile when it is to be placed intravenously, otherwise, a saline solution is a salt water solution. The sterile solution is typically used for intravenous infusion, rinsing contact lenses, nasal irrigation, and often used to clean a new piercing. Saline solutions are available in various formulations for different purposes. Salines are also used in cell biology, molecular biology, and biochemistry experiments.
Contents
Concentrations
Concentrations vary from low to normal to high. High concentrations are used rarely in medicine but frequently in molecular biology.
Normal
Normal saline (NS) — is the commonly-used term for a solution of 0.90% w/v of NaCl, about 300 mOsm/L or 9.0 g per liter.a Less commonly, this solution is referred to as physiological saline or isotonic saline, neither of which is technically accurate. NS is used frequently in intravenous drips (IVs) for patients who cannot take fluids orally and have developed or are in danger of developing dehydration or hypovolemia. NS is typically the first fluid used when hypovolemia is severe enough to threaten the adequacy of blood circulation, and has long been believed to be the safest fluid to give quickly in large volumes. However, it is now known that rapid infusion of NS can cause metabolic acidosis.[1]
The solution is 9 grams of sodiumchloride (NaCl) dissolved in water, to a total volume of 1000 ml. The mass of 1 millilitre of normal saline is 1.0046 grams at 22°C.[2][3] The molecular weight of sodium chloride is approximately 58.5 grams per mole, so 58.5 grams of sodium chloride equals 1 mole. Since normal saline contains 9 grams of NaCl, the concentration is 9 grams per liter divided by 58.5 grams per mole, or 0.154 moles per liter. Since NaCl dissociates into two ions – sodium and chloride – 1 molar NaCl is 2 osmolar. Thus, NS contains 154 mEq/L of Na+ and Cl−. It has a slightly higher degree of osmolarity (i.e. more solute per litre) than blood (However, if you take into account the osmotic coefficient, a correction for non-ideal solutions, then the saline solution is much closer to isotonic. Osmotic coefficient of NaCl is about 0.93; therefore 0.154 x 1000 x 2 x .93 = 290). Nonetheless, the osmolarity of normal saline is a close approximation to the osmolarity of NaCl in blood.
Usage
For medical uses, saline is often used to flush wounds and skin abrasions. Normal Saline will not burn or sting when applied.
Saline is also used in I.V. therapy, intravenously supplying extra water to a dehydrated patient or supplying the daily water and salt needs ("maintenance" needs) of a patient who is unable to take them by mouth. Because infusing a solution of low osmolality can cause problems, intravenous solutions with reduced saline concentrations typically have dextrose (glucose) added to maintain a safe osmolality while providing less sodium chloride. As the molecular weight (MW) of dextrose is greater, this has the same osmolality as normal saline despite having less sodium. Saline can also be used by addicts trying to rid their bodies of opiates and other illegal drugs, yet if not taken in the proper dose can have the opposite effect on an addicts body[citation needed]. Because the dextrose used in these preparations is dextrose monohydrate (a commercial form having MW 198 in contrast to MW 180 for glucose), 5% dextrose is equivalent to 4.5% glucose.
The amount of normal saline infused depends largely on the needs of the patient (e.g. ongoing diarrhea or heart failure) but is typically between 1.5 and 3 litres a day for an adult.
Rinse eye drops are often distributed for free by needle-exchange programmes. Containing normal saline, they are small, sterile, and safe for intravenous use.
Saline is also often used for nasal washes to relieve some of the symptoms of the common cold. This need not be sterile, as the nose cavity is not sterile either. In this case "home-made" saline may be used: this is made by dissolving approximately half a teaspoonful of table salt into a glass of clean tap water[citation needed].
Hypertonic saline
Hypertonic saline (NS) — 7% NaCl solutions are considered mucoactive agents and as such are used to hydrate thick secretions (mucous) in order to make it easier to cough up and out (expectorate). Hypertonic saline solutions are also used in critical care settings to help in haemorrhagic shock (but no other type of shock) and acute intracranial pressure.[4] Inhalation of hypertonic saline has also been shown to help in other respiratory problems, specifically bronchiolitis.[5] Hypertonic saline is currently recommended by the Cystic Fibrosis Foundation as a primary part of a Cystic Fibrosis treatment regimen.[6]
Mechanism of action
Aerosol — Nebulized hypertonic saline treatments disrupt the interaction between glycosaminoglycans and IL-8, rendering IL-8 susceptible to proteolytic degradation with subsequent decrease in neutrophil chemotaxis; all of this ultimately reducing inflammation.[4]
Other
Other concentrations commonly used include:
- Half-normal saline (0.45% NaCl), often with "D5" (5% dextrose), contains 77 mEq/L of Na and Cl and 50 g/L glucose.
- Quarter-normal saline (0.22% NaCl) has 39 mEq/L of Na and Cl and always contains 5% dextrose for osmolality reasons.
- Hypertonic saline may be used in perioperative fluid management protocols to reduce excessive intravenous fluid infusions and lessen pulmonary complications.[7] Hypertonic saline is used in treating hyponatremia and cerebral edema Rapid correction of hyopnatremia via hypertonic saline, or via any saline infusion > 40 mmol/L (Na+ having a valence of 1, 40 mmol/L = 40 mEq/L) greatly increases risk of central pontine myelinolysis (CPM), and so requires constant monitoring of patient response. Water privation combined with diuretic block does not produce as much risk of CPM as saline administration does; however, it does not correct hyponatremia as rapidly as administration of hypertonic saline does. Due to hypertonicity, administration may result in phlebitis and tissue necrosis. As such, concentrations greater than 3% NaCl should normally be administered via a central venous catheter, also known as a 'central line'. Such hypertonic saline is normally available in two strengths, the former of which is more commonly administered:
- 3% NaCl has 513 mEq/L of Na and Cl.
- 5% NaCl has 856 mEq/L of Na and Cl.
- NaCl solutions that are less commonly used are 7% (1200 mEq/L) and 23.4% (approx 4000 mEq/L), both of which are used (also via central line), often in conjunction with supplementary diuretics, in the treatment of traumatic brain injury.[8]
- Dextrose (glucose) 4% in 0.18% saline is used sometimes for maintenance replacement.
Solutions of saline with added ingredients
In medicine, common types of salines include:
- Lactated Ringer's solution
- Acetated Ringer's solution
And in cell biology, in addition to the above the following are used:
- Phosphate buffered saline (PBS) (recipes from Dulbecco = D-PBS, Galfre, Kuchler, Ausubel etc.)
- TRIS-buffered saline (TBS) (recipes from Goldsmith, Ausubel etc.)
- Hank's balanced salt solution (HBSS)
- Earle's balanced salt solution (EBSS)
- Standard saline citrate (SSC)
- HEPES-buffered saline (HBS) (recipes from Dittmar, Liu, Ausubel etc.)
- Gey's balanced salt solution (GBSS)
History
Saline was believed to have originated during the Indian Blue Cholera pandemic that swept across Europe in 1831. William Brooke O'Shaughnessy, a recent graduate of Edinburgh Medical School, proposed in an article to medical journal The Lancet to inject cholera patients with highly-oxygenated salts to treat the "universal stagnation of the venous system and rapid cessation of arterialisation of the blood" seen in severely dehydrated cholera patients.[9] He found his treatment harmless in dogs, and his proposal was soon adopted by the physician Thomas Latta in treating cholera patients to beneficial effect. In the following decades, variations and alternatives to Latta's solution were tested and used in treating cholera patients. These solutions contained a range of concentrations of sodium, chloride, potassium, carbonate, phosphate, and hydroxide. The breakthrough in achieving physiological concentrations was accomplished by Ringer in 1831, when he determined the optimal salt concentrations to maintain the contractility of frog heart muscle tissue. Normal saline is considered a descendant of the pre-Ringer solutions, as Ringer's findings were not adopted and widely used until decades later. The term "normal saline" itself appears to have little historical basis, except for Hartog Jakob Hamburger's 1882–83 in vitro studies of red cell lysis that incorrectly suggested that 0.9% was the concentration of salt in human blood (rather than 0.6%, the true concentration).[10]
See also
Notes
a^ In chemistry, a one normal of NaCl is 0.5 molar NaCl assuming complete dissociation. Physiological dissociation is approximately 1.7 ions per mole, so one normal of NaCl is 1/1.7 = 0.588 molar. This is roughly 4 times more concentrated than medical "normal saline" of 0.154 molar.
References
- ^ Prough, DS; Bidani, A (1999). "Hyperchloremic metabolic acidosis is a predictable consequence of intraoperative infusion of 0.9% saline". Anesthesiology 90 (5): 1247–1249. doi:10.1097/00000542-199905000-00003. PMID 10319767.
- ^ Fluid Density Calculator. Earthwardconsulting.com. Retrieved on 2011-02-27.
- ^ Water Density Calculator. Csgnetwork.com. Retrieved on 2011-02-27.
- ^ a b Strandvik GF (2009). "Hypertonic saline in critical care: a review of the literature and guidelines for use in hypotensive states and raised intracranial pressure". Anaesthesia 64 (9): 990–1003. doi:10.1111/j.1365-2044.2009.05986.x. PMID 19686485. http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=19686485.
- ^ Principi T, Komar L (2011). "A critical review of "a randomized trial of nebulized 3% hypertonic saline with epinephrine in the treatment of acute bronchiolitis in the emergency department."". J Popul Ther Clin Pharmacol 18 (2): e273–4. PMID 21633141.
- ^ O'Connell OJ, O'Farrell C, Harrison MJ, Eustace JA, Henry MT, Plant BJ (2011). "Nebulized hypertonic saline via positive expiratory pressure versus via jet nebulizer in patients with severe cystic fibrosis". Respir Care 56 (6): 771–5. doi:10.4187/respcare.00866. PMID 21333079. http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=21333079.
- ^ Vivian McAlister, Karen E. A. Burns, Tammy Znajda, and Brian Church. "Hypertonic Saline for Peri-operative Fluid Management" Cochrane Database of Systematic Reviews.1 (2010): CD005576 Available at: [1]
- ^ Access. Medscape. Retrieved on 2011-02-27.
- ^ O'Shaugnessy, WB (1831). "Proposal for a new method of treating the blue epidemic cholera by the injection of highly-oxygenated salts into the venous system". Lancet 17 (432): 366–71. doi:10.1016/S0140-6736(02)94163-2.
- ^ Awad, Sherif; Allison Simon P, Lobo Dileep N (2008). "The history of 0.9% saline". Clinical nutrition (Edinburgh, Scotland) 27 (2): 179–88. doi:10.1016/j.clnu.2008.01.008. PMID 18313809.
Infused substances Lactated Ringer's, Sodium bicarbonateMedicationsParenteral nutritionOtherAccess points Other equipment Specific risks Categories:- Intravenous fluids
Wikimedia Foundation. 2010.

