- Isopropylamine
-
Isopropylamine 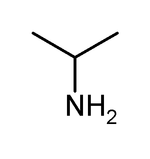
 Propan-2-amineOther names2-Propylamine
Propan-2-amineOther names2-Propylamine
2-AminopropaneIdentifiers CAS number 75-31-0 
PubChem 6363 ChemSpider 6123 
UNII P8W26T4MTD 
EC number 200-860-9 KEGG C06748 
ChEBI CHEBI:15739 
ChEMBL CHEMBL117080 
RTECS number NT8400000 Jmol-3D images Image 1
Image 2- NC(C)C
NC(C)C
Properties[2][3] Molecular formula C3H9N Molar mass 59.110 g/mol Appearance colourless liquid, hygroscopic, with odour of ammonia Density 0.722 g/cm3 (25 ºC) Melting point −95.2 ºC
Boiling point 32.4 ºC
Solubility in water miscible Acidity (pKa) 10.63[1] Refractive index (nD) 1.3742 (20 ºC) Hazards[3][4] GHS pictograms 

GHS signal word DANGER GHS hazard statements H224, H319, H335, H315 GHS precautionary statements P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P302+352, P303+361+353, P304+340, P305+351+338 EU Index 612-007-00-1 EU classification Extremely flammable (F+)
Irritant (Xi)R-phrases R12, R36/37/38 S-phrases (S2), S16, S26, S29 Flash point −51 °C (−35 ºF) open cup Autoignition
temperature402 ºC (756 ºF) Related compounds Related amines Ethylamine
Propylamine
sec-ButylamineRelated compounds Diisopropylamine  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Isopropylamine, also called 2-aminopropane, 2-propanamine, monoisopropylamine, and MIPA, is an organic compound, an amine. It is a base, as typical for amines. It is a hygroscopic colorless liquid with ammonia-like odor. Its melting point is −95.2 °C and its boiling point is 32.4 °C. It is miscible with water. It is extremely flammable, with flash point at −37 °C.
Isopropylamine can be obtained by aminating isopropyl alcohol with ammonia in presence of a nickel/copper or similar catalyst[5]:
- (CH3)2CHOH + NH3 → (CH3)2CHNH2 + H2O
The main uses of isopropylamine are in glyphosate herbicide formulations (e.g., Roundup or Vantage), a key component of atrazine (another herbicide), a regulating agent for plastics, intermediate in organic synthesis of coating materials, plastics, pesticides, rubber chemicals, pharmaceuticals and others, and as an additive in the petroleum industry.
Together with isopropyl alcohol it is used in some binary chemical weapons, as a mixture called OPA which in turn mixed with methylphosphonyl difluoride (known to the military as DF) produces sarin gas.
References
- ^ Hall, H.K., J. Am. Chem. Soc., 1957, 79, 5441.
- ^ Weast, Robert C., ed (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-478. ISBN 0-8493-0462-8..
- ^ a b Isopropylamine, International Chemical Safety Card 0908, Geneva: International Programme on Chemical Safety, April 1997, http://www.inchem.org/documents/icsc/icsc/eics0908.htm.
- ^ "Isopropylamine", Pocket Guide to Chemical Hazards, U.S. Department of Health and Human Services (NIOSH) Publication No. 2005-149, Washington, DC: Government Printing Office, 2005, ISBN 9780160727511, http://www.cdc.gov/niosh/npg/npgd0360.html.
- ^ US 4014933, Boettger, Guenther; Hubert Corr & Herwig Hoffmann et al., "Production of Amines from Alcohols", published 1977-03-29.
External links
- International Chemical Safety Card 0908
- NIOSH Pocket Guide to Chemical Hazards 0360
- Atrazine News - an Atrazine specific news site
Categories:- Amines
- NC(C)C
Wikimedia Foundation. 2010.
