- Dipotassium phosphate
-
Dipotassium phosphate 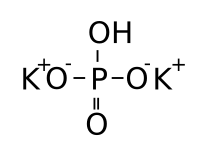 Potassium hydrogen phosphateOther namesPotassium monohydrogen phosphate
Potassium hydrogen phosphateOther namesPotassium monohydrogen phosphate
Phosphoric acid dipotassium salt
Potassium phosphate dibasicIdentifiers CAS number 7758-11-4 
PubChem 24450 ChemSpider 22858 
UNII CI71S98N1Z 
Jmol-3D images Image 1 - [K+].[K+].[O-]P([O-])(=O)O
Properties Molecular formula K2HPO4 Molar mass 174.2 g/mol Appearance white powder
deliquescentOdor odorless Density 2.44 g/cm3 Melting point >465 °C decomp.
Solubility in water 149.25 g/100 mL (20 °C) Solubility slightly soluble in alcohol Acidity (pKa) 12.4 Basicity (pKb) 6.8 Hazards EU Index Not listed NFPA 704 Flash point Non-flammable Related compounds Other cations Disodium phosphate
Diammonium phosphateRelated compounds Monopotassium phosphate
Tripotassium phosphate (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dipotassium phosphate (K2HPO4) - also phosphoric acid, dipotassium salt; dipotassium hydrogen orthophosphate; potassium phosphate, dibasic - is a highly water-soluble salt which is often used as a fertilizer, food additive and buffering agent. It is a common source of phosphorus and potassium.
The pH of a dipotassium phosphate solution is almost neutral. It is formed by the stoichiometric neutralization of phosphoric acid with potassium hydroxide:
- H3PO4 + 2 KOH → K2HPO4 + 2 H2O
It reacts with phosphoric acid to generate monopotassium phosphate:
K2HPO4 + H3PO4 → 2 KH2PO4
Safety
As a food additive, Dipotassium phosphate is on the FDA's Generally recognized as safe, or GRAS, list of substances[1]. It is used in non-dairy creamers to prevent coagulation.[2]
References
- ^ "Database of Select Committee on GRAS Substances (SCOGS) Reviews". http://www.cfsan.fda.gov/~dms/opascogd.html. Retrieved 2008-03-22. (listed as potassium phosphate, dibasic)
- ^ "dipotassium phosphate". http://everything2.com/e2node/dipotassium%2520phosphate. Retrieved 2009-01-06. (uses: Food Industry)
Categories:- Potassium compounds
- Phosphates
- Food ingredient stubs
- Inorganic compound stubs
Wikimedia Foundation. 2010.


