- Aniline Yellow
-
Aniline Yellow 
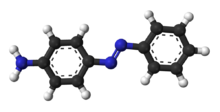 4-PhenyldiazenylanilineOther namespara-aminoazobenzene
4-PhenyldiazenylanilineOther namespara-aminoazobenzene
4-phenylazoaniline
AAB
Brasilazina oil Yellow G
Ceres Yellow
Fast spirit Yellow
Induline R
Oil Yellow AAB
Oil Yellow AN
Oil Yellow B
Oil Yellow 2G
Oil Yellow R
Organol Yellow
Organol Yellow 2A
Solvent Yellow
Somalia Yellow 2G
Stearix Brown 4R
Sudan Yellow R
Sudan Yellow RA
C.I. 11000Identifiers CAS number 60-09-3 
PubChem 6051 ChemSpider 5828 
EC number 200-453-6 KEGG C19187 
ChEBI CHEBI:233869 
ChEMBL CHEMBL83761 
Jmol-3D images Image 1 - N(=N/c1ccc(N)cc1)\c2ccccc2
Properties Molecular formula C6H5N=NC6H4NH2 Molar mass 197.24 g/mol Melting point 123-126 °C
Boiling point > 360 °C
Hazards Main hazards Highly toxic
Cancer suspect agentNFPA 704  Yellow (verify) (what is:
Yellow (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Aniline Yellow is a yellow azo dye and an aromatic amine. It is a derivate of azobenzene. It has the appearance of an orange powder. It is a carcinogen.[1]
Aniline Yellow was the first azo dye. it was first produced in 1861 by C. Mene. The second azo dye was Bismarck Brown in 1863. Aniline Yellow was commercialized in 1864 as the first commercial azo dye, a year after Aniline Black. It is manufactured from aniline.
Uses
Aniline Yellow is used in microscopy for vital staining,[2] in pyrotechnics for yellow colored smokes, in yellow pigments and inks including inks for inkjet printers. It is also used in insecticides, lacquers, varnishes, waxes, oil stains, and styrene resins. It is also an intermediate in synthesis of other dyes, e.g. chrysoidine, indulines, Solid Yellow, and Acid Yellow.
Toxic oil syndrome
Aniline Yellow was involved in the 1981 Spanish Toxic Oil Syndrome (TOS). A Madrid-based company imported denaturated rapeseed oil, dyed by aniline yellow to mark it as unsuitable for human consumption, to be used as a fuel in steel mills. However, the company distilled the oil to remove the dye, and sold it as a much more valuable olive oil for cooking. The result was a rash of pneumonia-type illnesses, with a second stage with lesions, weight loss, paralysis, and muscle wasting. The net result was over 20,000 sick and 400 dead. The chemistry of the poisonous reaction is still subject of a debate.[3]
References
Categories:- Azo dyes
- Vital stains
- Solvent dyes
- Anilines
Wikimedia Foundation. 2010.

