- Copper(II) tetrafluoroborate
-
Copper(II) tetrafluoroborate[1] 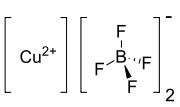 Copper(II) tetrafluoroborate
Copper(II) tetrafluoroborateIdentifiers CAS number 14735-84-3 ChemSpider 21241480 
Jmol-3D images Image 1 - [Cu+2].F[B-](F)(F)F.F[B-](F)(F)F.O
Properties Molecular formula Cu(BF4)2 Molar mass 237.155 g/mol Appearance solid Solubility in water soluble in water Related compounds Other anions Copper(II) chloride
Copper(II) oxide
Copper(II) triflateOther cations Sodium tetrafluoroborate
Lithium tetrafluoroborate
Silver tetrafluoroborate (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Copper(II) tetrafluoroborate is the copper salt of tetrafluoroboric acid which involves copper in its +2 oxidation state bonded to two tetrafluoroborate anions (BF4−). Tetrafluoroborate adopts a tetrahedral shape, similar to that of methane. The central boron atom has a formal charge of −1, because of its four covalent bonds. Its oxidation state is +3.[2]
References
- ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, FL: CRC Press, pp. 4–56, ISBN 0-8493-0594-2
- ^ Copper(II) Tetrafluorborate, chemicalland21.com
Categories:- Inorganic compound stubs
- Copper compounds
- Tetrafluoroborates
Wikimedia Foundation. 2010.
