- Zirconium tungstate
-
Zirconium(IV) tungstate 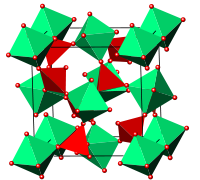 Other nameszirconium tungsten oxide
Other nameszirconium tungsten oxideIdentifiers CAS number 16853-74-0 Properties Molecular formula Zr(WO4)2 Molar mass 586.92 g/mol Appearance white powder Density 5.09 g/cm³, solid Solubility in water negligible Hazards MSDS MSDS EU classification not listed NFPA 704  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Zirconium tungstate (Zr(WO4)2) is a metal oxide with unusual properties. The phase formed at ambient pressure by reaction of ZrO2 and WO3 is a metastable cubic phase, which has negative thermal expansion characteristics, namely it shrinks over a wide range of temperatures when heated.[1] In contrast to most other ceramics exhibiting negative CTE (coefficient of thermal expansion), the CTE of ZrW2O8 is isotropic and has a large negative magnitude (average CTE of -7.2x10−6K−1) over a wide range of temperature (-273 °C to 777 °C).[2] A number of other phases are formed at high pressures.
Contents
Cubic phase
Cubic zirconium tungstate (alpha-ZrW2O8), one of the several known phases of zirconium tungstate (ZrW2O8) is perhaps one of the most studied materials to exhibit negative thermal expansion. It has been shown to contract continuously over a previously unprecedented temperature range of 0.3 to 1050 K (at higher temperatures the material decomposes). Since the structure is cubic, as described below, the thermal contraction is isotropic - equal in all directions. There is much ongoing research attempting to elucidate why the material exhibits such dramatic negative thermal expansion.
This phase is thermodynamically unstable at room temperature with respect to the binary oxides ZrO2 and WO3, but may be synthesised by heating stoichiometric quantities of these oxides together and then quenching the material by rapidly cooling it from approximately 900 °C to room temperature.
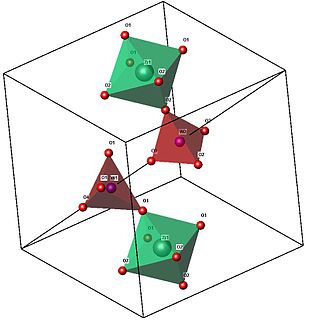
The structure of cubic zirconium tungstate consists of corner-sharing ZrO6 octahedral and WO4 tetrahedral structural units. Its unusual expansion properties are thought to be due to vibrational modes known as Rigid Unit Modes (RUMs), which involve the coupled rotation of the polyhedral units that make up the structure, and lead to contraction.
Detailed crystal structure
The arrangement of the groups in the structure of cubic ZrW2O8 is analogous to the simple NaCl structure, with ZrO6 octahedra at the Na sites, and W2O8 groups at the Cl sites. The unit cell consists of 44 atoms aligned in a primitive cubic Bravais lattice, with unit cell length 9.15462 Angstroms.
The ZrO6 octahedra are only slightly distorted from a regular conformation, and all oxygen sites in a given octahedron are related by symmetry. The W2O8 unit is made up of two crystallographically distinct WO4 tetrahedra, which are not formally bonded to each other. These two types of tetrahedra differ with respect to the W-O bond lengths and angles. The WO4 tetrahedra are distorted from a regular shape since one oxygen is unconstrained (an atom that is bonded only to the central tungsten (W) atom), and the three other oxygens are each bonded to a zirconium atom (i.e. the corner-sharing of polyhedra).
The structure has P213 space group symmetry at low temperatures. At higher temperatures, a centre of inversion is introduced by the disordering of the orientation of tungstate groups, and the space group above the phase transition temperature (~180C) is Pa
 .
.Octahedra and tetrahedra are linked together by sharing an oxygen atom. In the image, note the corner-touching between octahedra and tetrahedra; these are the location of the shared oxygen. The vertices of the tetrahedra and octahedra represent the oxygen, which are spread about the central zirconium and tungsten. Geometrically, the two shapes can "pivot" around these corner-sharing oxygens, without a distortion of the polyhedra themselves. This pivoting is what is thought to lead to the negative thermal expansion, as in certain low frequency normal modes this leads to the contracting 'RUMs' mentioned above.
High pressure forms
At high pressure, zirconium tungstate undergoes a series of phase transitions, first to an amorphous phase, and then to a U3O8-type phase, in which the zirconium and tungsten atoms are disordered.
References
- ^ Mary, T. A.; J. S. O. Evans, T. Vogt, A. W. Sleight (1996-04-05). "Negative Thermal Expansion from 0.3 to 1050 Kelvin in ZrW2O8". Science 272 (5258): 90–92. Bibcode 1996Sci...272...90M. doi:10.1126/science.272.5258.90. http://www.sciencemag.org/cgi/content/abstract/272/5258/90. Retrieved 2008-02-20.
- ^ Sleight, A.W. (1998). "Isotropic Negative Thermal Expansion". Ann. Rev. Mater. Sci. 28: 29–43. doi:10.1146/annurev.matsci.28.1.29.
External links
Zirconium compounds Categories:- Zirconium compounds
- Tungstates
- Inorganic compounds
Wikimedia Foundation. 2010.