- Lipoprotein-associated phospholipase A2
-
Phospholipase A2, group VII (platelet-activating factor acetylhydrolase, plasma) 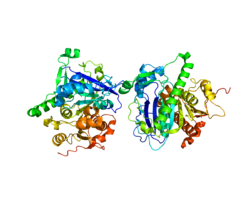
Rendering based on PDB 3D59.Available structures PDB 3D59, 3D5E, 3F96, 3F97, 3F98, 3F9C Identifiers Symbols PLA2G7; LDL-PLA2; LP-PLA2; PAFAH External IDs OMIM: 601690 MGI: 1351327 HomoloGene: 3725 GeneCards: PLA2G7 Gene EC number 3.1.1.47 Gene Ontology Molecular function • 1-alkyl-2-acetylglycerophosphocholine esterase activity
• phospholipid binding
• hydrolase activityCellular component • extracellular region
• extracellular spaceBiological process • lipid metabolic process
• inflammatory response
• lipid catabolic processSources: Amigo / QuickGO RNA expression pattern 
More reference expression data Orthologs Species Human Mouse Entrez 7941 27226 Ensembl ENSG00000146070 ENSMUSG00000023913 UniProt Q13093 Q3U1V7 RefSeq (mRNA) NM_001168357.1 NM_013737.5 RefSeq (protein) NP_001161829.1 NP_038765.2 Location (UCSC) Chr 6:
46.67 – 46.7 MbChr 17:
43.71 – 43.75 MbPubMed search [1] [2] Lipoprotein-associated phospholipase A2 (Lp-PLA2) also known as platelet-activating factor acetylhydrolase (PAF-AH) is a phospholipase A2 enzyme that in humans is encoded by the PLA2G7 gene.[1][2] Lp-PLA2 is a 45-kDa protein of 441 amino acids.[3]
Contents
Function
In the blood it travels mainly with low-density lipoprotein (LDL). Less than 20% is associated with high-density lipoprotein HDL. It is an enzyme produced by inflammatory cells and hydrolyzes oxidized phospholipids in LDL.
Lp-PLA2 is platelet-activating factor (PAF) acetylhydrolase (EC 3.1.1.47), a secreted enzyme that catalyzes the degradation of PAF to inactive products by hydrolysis of the acetyl group at the sn-2 position, producing the biologically inactive products LYSO-PAF and acetate.[4]
Clinical significance
Lp-PLA2 is involved in the development of atherosclerosis.[3] In human atherosclerotic lesions, 2 main sources of Lp-PLA2 can be identified, including that which is brought into the intima bound to LDL (from the circulation), and that which is synthesized de novo by plaque inflammatory cells (macrophages, T cells, mast cells)."
It is used as a marker for cardiac disease.[5]
A meta-analysis involving a total of 79,036 participants in 32 prospective studies found that Lp-PLA2 levels are positively correlated with increased risk of developing coronary heart disease and stroke.[6]
References
- ^ Tjoelker LW, Wilder C, Eberhardt C, Stafforini DM, Dietsch G, Schimpf B, Hooper S, Le Trong H, Cousens LS, Zimmerman GA (April 1995). "Anti-inflammatory properties of a platelet-activating factor acetylhydrolase". Nature 374 (6522): 549–53. doi:10.1038/374549a0. PMID 7700381.
- ^ Tew DG, Southan C, Rice SQ, Lawrence MP, Li H, Boyd HF, Moores K, Gloger IS, Macphee CH (April 1996). "Purification, properties, sequencing, and cloning of a lipoprotein-associated, serine-dependent phospholipase involved in the oxidative modification of low-density lipoproteins". Arterioscler. Thromb. Vasc. Biol. 16 (4): 591–9. doi:10.1161/01.ATV.16.4.591. PMID 8624782.
- ^ a b Zalewski A, Macphee C (May 2005). "Role of lipoprotein-associated phospholipase A2 in atherosclerosis: biology, epidemiology, and possible therapeutic target". Arterioscler. Thromb. Vasc. Biol. 25 (5): 923–31. doi:10.1161/01.ATV.0000160551.21962.a7. PMID 15731492.
- ^ "Entrez Gene: PLA2G7 phospholipase A2, group VII (platelet-activating factor acetylhydrolase, plasma)". http://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=7941.
- ^ Mohler ER, Ballantyne CM, Davidson MH, Hanefeld M, Ruilope LM, Johnson JL, Zalewski A (April 2008). "The effect of darapladib on plasma lipoprotein-associated phospholipase A2 activity and cardiovascular biomarkers in patients with stable coronary heart disease or coronary heart disease risk equivalent: the results of a multicenter, randomized, double-blind, placebo-controlled study". J. Am. Coll. Cardiol. 51 (17): 1632–41. doi:10.1016/j.jacc.2007.11.079. PMID 18436114.
- ^ The Lp-PLA2 Studies Collaboration (2010). "Lipoprotein-associated phospholipase A2 and risk of coronary disease, stroke, and mortality: collaborative analysis of 32 prospective studies". The Lancet 375 (9725): 1536–1544. doi:10.1016/S0140-6736(10)60319-4. Lay summary – BBC News.
See also
Further reading
- Schröder HC, Perovic S, Kavsan V, et al. (1998). "Mechanisms of prionSc- and HIV-1 gp120 induced neuronal cell death.". Neurotoxicology 19 (4–5): 683–8. PMID 9745929.
- Feng Y, Walsh CA (2001). "Protein-protein interactions, cytoskeletal regulation and neuronal migration". Nat. Rev. Neurosci. 2 (6): 408–16. doi:10.1038/35077559. PMID 11389474.
- Tjoelker LW, Eberhardt C, Unger J, et al. (1995). "Plasma platelet-activating factor acetylhydrolase is a secreted phospholipase A2 with a catalytic triad". J. Biol. Chem. 270 (43): 25481–7. doi:10.1074/jbc.270.43.25481. PMID 7592717.
- Stafforini DM, Satoh K, Atkinson DL, et al. (1996). "Platelet-activating factor acetylhydrolase deficiency. A missense mutation near the active site of an anti-inflammatory phospholipase". J. Clin. Invest. 97 (12): 2784–91. doi:10.1172/JCI118733. PMC 507371. PMID 8675689. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=507371.
- Yamada Y, Yokota M (1997). "Loss of activity of plasma platelet-activating factor acetylhydrolase due to a novel Gln281-->Arg mutation". Biochem. Biophys. Res. Commun. 236 (3): 772–5. doi:10.1006/bbrc.1997.7047. PMID 9245731.
- Mavoungou E, Georges-Courbot MC, Poaty-Mavoungou V, et al. (1997). "HIV and SIV envelope glycoproteins induce phospholipase A2 activation in human and macaque lymphocytes". J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 16 (1): 1–9. doi:10.1097/00042560-199709010-00001. PMID 9377118.
- Sapir T, Elbaum M, Reiner O (1998). "Reduction of microtubule catastrophe events by LIS1, platelet-activating factor acetylhydrolase subunit". EMBO J. 16 (23): 6977–84. doi:10.1093/emboj/16.23.6977. PMC 1170301. PMID 9384577. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1170301.
- Hiramoto M, Yoshida H, Imaizumi T, et al. (1998). "A mutation in plasma platelet-activating factor acetylhydrolase (Val279-->Phe) is a genetic risk factor for stroke". Stroke 28 (12): 2417–20. PMID 9412624.
- Yamada Y, Ichihara S, Fujimura T, Yokota M (1998). "Identification of the G994--> T missense in exon 9 of the plasma platelet-activating factor acetylhydrolase gene as an independent risk factor for coronary artery disease in Japanese men". Metab. Clin. Exp. 47 (2): 177–81. doi:10.1016/S0026-0495(98)90216-5. PMID 9472966.
- Yoshida H, Imaizumi T, Fujimoto K, et al. (1998). "A mutation in plasma platelet-activating factor acetylhydrolase (Val279Phe) is a genetic risk factor for cerebral hemorrhage but not for hypertension". Thromb. Haemost. 80 (3): 372–5. PMID 9759612.
- Lecointe N, Meerabux J, Ebihara M, et al. (1999). "Molecular analysis of an unstable genomic region at chromosome band 11q23 reveals a disruption of the gene encoding the alpha2 subunit of platelet-activating factor acetylhydrolase (Pafah1a2) in human lymphoma". Oncogene 18 (18): 2852–9. doi:10.1038/sj.onc.1202645. PMID 10362256.
- Kruse S, Mao XQ, Heinzmann A, et al. (2000). "The Ile198Thr and Ala379Val variants of plasmatic PAF-acetylhydrolase impair catalytical activities and are associated with atopy and asthma". Am. J. Hum. Genet. 66 (5): 1522–30. doi:10.1086/302901. PMC 1378003. PMID 10733466. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1378003.
- Howard KM, Olson MS (2000). "The expression and localization of plasma platelet-activating factor acetylhydrolase in endotoxemic rats". J. Biol. Chem. 275 (26): 19891–6. doi:10.1074/jbc.M001462200. PMID 10748027.
- Min JH, Wilder C, Aoki J, et al. (2001). "Platelet-activating factor acetylhydrolases: broad substrate specificity and lipoprotein binding does not modulate the catalytic properties of the plasma enzyme". Biochemistry 40 (15): 4539–49. doi:10.1021/bi002600g. PMID 11294621.
- Quarck R, De Geest B, Stengel D, et al. (2001). "Adenovirus-mediated gene transfer of human platelet-activating factor-acetylhydrolase prevents injury-induced neointima formation and reduces spontaneous atherosclerosis in apolipoprotein E-deficient mice". Circulation 103 (20): 2495–500. PMID 11369691.
- Unno N, Nakamura T, Mitsuoka H, et al. (2002). "Association of a G994 →T Missense Mutation in the Plasma Platelet-Activating Factor Acetylhydrolase Gene With Risk of Abdominal Aortic Aneurysm in Japanese". Ann. Surg. 235 (2): 297–302. doi:10.1097/00000658-200202000-00020. PMC 1422429. PMID 11807372. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1422429.
This enzyme-related article is a stub. You can help Wikipedia by expanding it.
