- Chiral derivatizing agent
-
A chiral derivatizing agent (CDA) also known as a chiral resolving reagent, is a chiral auxiliary which can convert a mixture of enantiomers into diastereomers in order to analyse the quantities of each enantiomer present within the mix. In NMR spectroscopy these compounds are called chiral shift reagents.
History
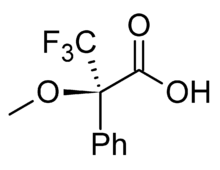
Since NMR spectroscopy has been available to chemists, there have been numerous studies on the applications of this technique. One of these noted the difference in the chemical shift (i.e. the distance between the peaks) of two diastereomers.[1] Conversely, two compounds that are enantiomers have the same NMR spectral properties. It was reasoned that if a mix of enantiomers could be converted into a mix of diastereomers by bonding them to another chemical that was itself chiral, it would be possible to distinguish this new mixture using NMR, and therefore learn about the original enantiomeric mixture. The first popular example of this technique was published in 1969 by Harry S. Mosher. The chiral agent is a single enantiomer of MTPA (α-methoxy-α-(trifluoromethyl)phenylacetic acid), also known as Mosher's acid.[2] The corresponding acid chloride is also known as Mosher's acid chloride, and the resultant diastereomeric esters are known as Mosher's esters. Another system is Pirkle's Alcohol developed in 1977.
Since then, other methods have been developed. At first, they were based on MTPA. Later, the principles were extended to phosphorus and boron systems. Research is still continuing on in this area and the limitations of CDA's as a valid test for enantiopurity are becoming fewer as more systems are designed.
Mosher's acid
Mosher's acid or acid chloride reacts easily with alcohols and amines to give esters and amides respectively. This method is able to determine the configuration of simple chiral amines and alcohols.[3] The reason racemization does not occur is because there is no α-hydrogen near the carboxyl group (therefore it cannot form an enol). This allows it to react with alcohol or amine to form an MTPA ester or amide respectively.
References
- ^ J. L. Mateos and D. J. Cram (1959). "Studies in Stereochemistry. XXXI. Conformation, Configuration and Physical Properties of Open-chain Diastereomers". J. Am. Chem. Soc. 81 (11): 2756–2762. doi:10.1021/ja01520a037.
- ^ J. A. Dale, D. L. Dull and H. S. Mosher (1969). "α-Methoxy-α-trifluoromethylphenylacetic acid, a versatile reagent for the determination of enantiomeric composition of alcohols and amines". J. Org. Chem. 34 (9): 2543–2549. doi:10.1021/jo01261a013.
- ^ D. Parker (1991). "NMR determination of enantiomeric purity". Chem. Rev. 91 (7): 1441–1457. doi:10.1021/cr00007a009.
Concepts in asymmetric synthesis Chirality types Chirality · Stereocenter · Planar chirality · Chiral ligand · Axial chirality · Supramolecular chirality · Inherent chiralityChiral molecules Stereoisomer · Enantiomer · Diastereomer · Meso compound · Enantiomeric excess · Diastereomeric excess ·Analysis Optical rotation · Chiral derivatizing agents · NMR spectroscopy of stereoisomers · Ultraviolet-visible spectroscopy of stereoisomersChiral resolution Recrystallization · Kinetic resolution · Chiral column chromatography · Diastereomeric recrystallizationReactions Categories:
Wikimedia Foundation. 2010.